Relationship between gut microbiota establishment patterns and the risk of necrotizing enterocolitis in preterm infants: a retrospective cohort study
Highlight box
Key findings
• This longitudinal cohort study demonstrates that preterm infants who develop necrotizing enterocolitis (NEC) exhibit early and persistent gut dysbiosis, characterized by reduced microbial diversity, decreased Bifidobacterium and Lactobacillus, and overgrowth of potential pathogens such as Enterobacteriaceae and Klebsiella. These alterations are detectable as early as day 3 of life and worsen over time. A lower Shannon index was identified as an independent risk factor, while higher diversity was protective. Additional independent risk factors included low birth weight, feeding intolerance, and prolonged or broad-spectrum antibiotic use.
What is known and what is new?
• NEC is strongly associated with intestinal immaturity and microbial imbalance in preterm infants. Reduced diversity and pathogen dominance have been reported prior to disease onset.
• This study provides longitudinal evidence in a Chinese cohort, showing that dysbiosis begins very early (day 3) and progresses dynamically. It identifies the Shannon index as a potential early biomarker for NEC risk and highlights the impact of antibiotic exposure and feeding practices on microbiota development.
What is the implication, and what should change now?
• Early microbiota monitoring should be integrated into clinical care for preterm infants, particularly using diversity indices such as the Shannon index. Antibiotic use should be carefully limited in duration and spectrum. Strategies to promote breastfeeding, optimize feeding tolerance, and consider targeted probiotic supplementation should be strengthened. These measures may help prevent NEC and improve outcomes in this high-risk population.
Introduction
Necrotizing enterocolitis (NEC) is one of the most severe gastrointestinal emergencies in the neonatal period, primarily affecting preterm and low birth weight infants. Statistics indicate that the incidence of NEC in very low birth weight infants (<1,500 g) reaches 5–10%, with a mortality rate of 20–30% (1,2). With advances in neonatal intensive care technology, although the survival rate of preterm infants (PIs) has significantly improved, the incidence of NEC has not decreased markedly and remains a major cause of mortality and long-term neurodevelopmental impairment in PIs (3). The pathogenesis of NEC is complex, involving multiple factors such as immature intestinal immune system, impaired intestinal mucosal barrier function, and insufficient intestinal blood perfusion. However, increasing evidence in recent years indicates that gut dysbiosis plays a key role in the development of NEC (4). Under normal circumstances, the establishment of the neonatal gut microbiota is a dynamic succession process, gradually evolving from a sterile state at birth into a complex microbial ecosystem. Due to characteristics such as low gestational age, immature immune function, and prolonged hospitalization, the gut microbiota establishment in PIs exhibits significant differences compared to full-term infants (5,6). Research has found that PIs demonstrate reduced gut microbiota diversity, delayed colonization of beneficial bacteria such as Bifidobacterium and Lactobacillus, and overgrowth of potential pathogenic bacteria such as Enterobacteriaceae (7). This dysbiosis may serve as a significant prerequisite for the development of NEC.
The application of 16S rRNA high-throughput sequencing enables a more comprehensive and accurate understanding of the composition and dynamic changes of the gut microbiota. By analyzing microbiota diversity indices such as the Shannon index and Simpson index, along with the relative abundances of various bacterial genera, the degree of dysbiosis can be quantitatively assessed (8,9). However, longitudinal studies on the relationship between the gut microbiota establishment patterns and the risk of NEC in PIs remain relatively limited, particularly with insufficient research data available for PIs in China.
In this context, the present study employed a retrospective cohort design to systematically analyze the gut microbiota characteristics of PIs at different time points after birth, explore the relationship between microbiota establishment patterns and the occurrence of NEC, and identify clinical factors influencing microbiota establishment, with the goal of providing scientific evidence for the early prevention and intervention of NEC. We present this article in accordance with the STROBE reporting checklist (available at https://tp.amegroups.com/article/view/10.21037/tp-2025-aw-713/rc).
Methods
Study design
This retrospective cohort study was conducted in the Neonatal Intensive Care Unit (NICU) of a tertiary hospital from January 2023 to December 2024. The study protocol was approved by the ethics committee of Wenzhou People’s Hospital. The study was conducted in accordance with the Declaration of Helsinki and its subsequent amendments. Since this retrospective study did not involve any additional interventions, the requirement for informed consent was waived by the hospital’s ethics committee. All data were anonymized to strictly protect the privacy of pediatric patients.
Study population
Inclusion criteria:
- PIs with gestational age <37 weeks;
- Birth weight <2,500 g;
- Hospitalization duration ≥21 days;
- Complete gut microbiota testing data (at 3, 7, 14, and 21 days after birth);
- Complete clinical records.
Exclusion criteria:
- Congenital digestive tract malformations;
- Chromosomal abnormalities or inherited metabolic disorders;
- Death within 72 hours of birth;
- Severe congenital heart disease;
- Maternal use of immunosuppressive agents during pregnancy;
- Transfer to another hospital or discontinuation of treatment.
Diagnostic criteria for NEC
Diagnosis of NEC was based on the modified Bell staging criteria (10): stage I (suspected NEC): nonspecific symptoms such as abdominal distension, gastric stasis, and bloody stools; stage II (confirmed NEC): stage I symptoms plus imaging changes such as pneumatosis intestinalis and portal venous gas; stage III (progressive NEC): stage II manifestations plus complications such as intestinal perforation, peritonitis, and shock. In this study, patients with Bell stage ≥ II were defined as the NEC group. In addition, 18 patients with Bell stage I (suspected NEC) were included for supplementary subgroup analysis. These infants were not included in the primary comparison between the NEC group and the control group (CG).
Follow-up time
The observation window period of this study was from the birth of the infants to 21 days after birth. The average follow-up time of the NEC group ranged from 7 to 21 days, and the onset time of NEC occurred between 3 and 18 days after birth. All included infants completed the 21-day observation period or reached a clear clinical outcome (NEC diagnosis, death, or discharge).
Sample size calculation
According to literature reports, the incidence of NEC in very low birth weight infants is approximately 5–10%. In this study, it was anticipated that the difference in Shannon index between the NEC group and the CG would be clinically significant. With α =0.05 and β =0.20 (test power 1-β =0.80), a two-tailed test was employed. Based on preliminary pre-experimental data, the expected mean Shannon index for the CG was 1.87±0.52, while that for the NEC group was 1.23±0.45, with a difference of 0.64 between the two groups. The sample size for each group was calculated using the following formula for comparing two independent sample means:
Where σ is the pooled standard deviation and δ is the difference between the two group means.
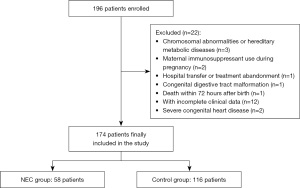
Calculations indicated that each group required approximately 52 patients. Accounting for a 20% dropout rate, 65 patients were required per group, for a total sample size of 130 patients. The study ultimately included 174 patients (58 in the NEC group and 116 in the CG), meeting the statistical requirements. The CG was selected from PIs who were hospitalized during the same period, met the inclusion criteria, had complete gut microbiota monitoring data, and did not develop NEC during hospitalization. They were selected using a random number table at a 1:2 ratio and were matched to the NEC group by admission time. The specific screening process is shown in Figure 1.
Sample collection and processing
Fecal sample collection
Fresh fecal samples were collected at 3, 7, 14, and 21 days after birth. Each sample weighed ≥200 mg and was collected using sterile fecal collection tubes. Samples were immediately stored at −80 ℃ pending analysis. All fecal samples from both the NEC group and CG were collected, processed, and analyzed using identical protocols to ensure comparability of assessment methods.
DNA extraction and bioinformatics analysis
Total fecal DNA was extracted using the MolPure® Stool DNA Kit (Yeasen, China), and DNA concentration and purity were assessed using a NanoDrop 2000 spectrophotometer (Thermo Fisher Scientific, USA). The V3-V4 region of the 16S rRNA gene was amplified by polymerase chain reaction (PCR) using the primers 341F (5'-CCTAYGGGRBGCASCAG-3') and 806R (5'-GGACTACNNGGGTATCTAAT-3'). The primer sequences were removed before data analysis. High-throughput sequencing was performed using the Illumina MiSeq PE300 platform. Rw data were quality-filtered using fastp (v0.23.2) with the following parameters: a quality threshold of Q20, a sliding window size of 4 bp, and a minimum sequence length of 200 bp. Each sample yielded an average of 45,632±8,247 raw reads, and an average of 38,456±7,123 valid sequences were retained after quality control. Further analysis was conducted using the QIIME2 (v2023.5) platform. The DADA2 algorithm was used for sequence denoising and amplicon sequence variant (ASV) generation, yielding a total of 2,847 ASVs. Taxonomic annotation was performed using a Naive Bayes classifier based on the Silva (v138) reference database. At the genus level, approximately 8.3% of the sequences were unclassified; at the species level, approximately 34.6% of the sequences were unclassified. Alpha diversity indices (Shannon index, Simpson index) were calculated. Beta diversity was calculated using Bray-Curtis distance, and visualization was performed using principal coordinate analysis (PCoA). The relative abundance at each taxonomic level (phylum, class, order, family, and genus) was statistically analyzed and visualized using R software (v4.2.1).
The Shannon index and Simpson index are commonly used indicators for assessing microbiome alpha diversity. The Shannon index comprehensively reflects species richness and evenness, typically ranging from 0 to 5. Higher values indicate greater microbial diversity and a more even distribution of species. The Simpson index, which reflects the dominance of microbiome, was calculated in the 1-D form in this study. Values range from 0 to 1, with values closer to 1 indicating a more even distribution of species and greater microbial diversity.
Clinical data collection
Clinical data were collected, including the patient’s basic information (gestational age, birth weight, sex, mode of delivery, Apgar score), feeding status (feeding method, initiation time, time to achieve full enteral feeding, feeding intolerance), and treatment measures (mechanical ventilation, antibiotic use, probiotic use).
Feeding intolerance was defined as meeting any of the following criteria: (I) gastric residuals >50% of the previous feeding volume or >3 mL/kg; (II) abdominal distension (increase in abdominal circumference >2 cm); (III) vomiting (≥2 times/day); (IV) presence of bile in gastric contents. In this study, feeding intolerance events were recorded cumulatively before NEC diagnosis in the NEC group or before discharge in the CG. Events on the day of NEC onset and thereafter were excluded.
Antibiotic use: for PIs with high-risk factors for infection (premature rupture of membranes >18 hours, maternal fever before delivery, meconium-stained amniotic fluid, etc.), ampicillin combined with gentamicin was empirically used after birth. The regimen was adjusted based on blood culture results and clinical presentation. Broad-spectrum antibiotics were defined as third-generation cephalosporins (cefotaxime, ceftriaxone), carbapenems (meropenem, imipenem), or β-lactam/β-lactamase inhibitor combinations (piperacillin/tazobactam).
Probiotic use: prophylactic probiotics (Bifidobacterium triple live bacteria powder, 1 g twice daily) were administered to eligible PIs (birth weight >1,000 g, no severe infection, and initiation of enteral feeding) until discharge. Probiotics were used in 23 cases (39.66%) in the NEC group and 67 cases (57.76%) in the CG.
Statistical analysis
Data analysis was performed using Statistical Package for the Social Sciences (SPSS) 26.0. All continuous data were tested for normality using the Shapiro-Wilk test and conformed to normal distribution, which were presented as mean ± standard deviation. Intergroup comparisons were conducted using the independent-samples t-tests. Repeated-measures analysis of variance was applied for comparisons at multiple time points, and Bonferroni correction was used for multiple comparisons between groups. Categorical data were expressed as number of cases (percentages) and were compared using the chi-square test or Fisher’s exact test. Univariate logistic regression analysis was performed to assess the association between each variable and the occurrence of NEC. Variables with P<0.10 in the univariate analysis were included as candidate variables in the multivariate analysis. Multivariate logistic regression was conducted using the Forward LR, with an inclusion criterion of P<0.05 and an exclusion criterion of P>0.10. The goodness of fit of the model was evaluated using the Hosmer-Lemeshow test. A two-sided P<0.05 was considered statistically significant.
Results
Comparison of basic characteristics between the two groups
This study included a total of 174 PIs, comprising 58 cases in the NEC group and 116 cases in the CG. Among the NEC cases, 28 were classified as stage II and 30 as stage III, while an additional 18 cases with Bell stage I were included only in the subgroup analysis. The NEC group exhibited significantly lower gestational age, birth weight, and antibiotic use within 3 days of birth compared to the CG (P<0.05), but the differences were small and should be interpreted with caution. The NEC group also showed lower 5-minute Apgar scores and a higher proportion of mechanical ventilation (P<0.05). No statistically significant differences were observed between the two groups in sex ratio, mode of delivery, antenatal hormone use, and coinfections (P>0.05). Among the 58 infants in the NEC group, the median age at onset (day of diagnosis) was 9 days [interquartile range (IQR), 6–13 days], with a range of 3 to 18 days. Regarding the distribution of onset time, 18 infants (31.03%) were diagnosed within 3–7 days after birth, 32 infants (55.17%) within 8–14 days, and 8 infants (13.79%) within 15–18 days. Further analyses were performed to compare the microbiota characteristics between early-onset (≤7 days, n=18) and late-onset (>7 days, n=40) NEC cases. On the third day after birth, the Shannon index was slightly lower in early-onset NEC infants (1.08±0.38) compared with those in late-onset NEC infants (1.30±0.48), though the difference was not statistically significant (P=0.09). No significant differences were observed in the composition of major bacterial genera between the two groups (P>0.05), which may be related to the smaller sample size of early-onset cases (Table 1).
Table 1
| Characteristics | NEC group (n=58) | Control group (n=116) | t/c2 | P |
|---|---|---|---|---|
| Gestational age (weeks) | 29.34±2.18 | 30.12±2.45 | 2.057 | 0.04 |
| Birth weight (g) | 1,245.67±285.34 | 1,398.45±312.56 | 3.124 | 0.002 |
| Male | 32 (55.17) | 58 (50.00) | 0.408 | 0.52 |
| Caesarean section | 41 (70.69) | 75 (64.66) | 0.623 | 0.43 |
| 5-minute Apgar scores | 7.82±1.24 | 8.36±0.98 | 3.084 | 0.002 |
| Antenatal hormone use | 38 (65.52) | 82 (70.69) | 0.478 | 0.49 |
| Mechanical ventilation | 45 (77.59) | 68 (58.62) | 5.982 | 0.01 |
| Antibiotic use within 3 days of birth | 48 (82.76) | 72 (62.07) | 7.234 | 0.007 |
| Coinfections | 23 (39.66) | 35 (30.17) | 0.698 | 0.21 |
Data are presented as number (%) or mean ± standard deviation. NEC, necrotizing enterocolitis.
Dynamic changes in gut microbiota diversity
Longitudinal analysis revealed that gut microbiota diversity in the CG gradually increased with age, whereas diversity in the NEC group showed slow growth. At 3, 7, 14, and 21 days after birth, the NEC group exhibited both significantly lower Shannon index and Simpson index than the CG (P<0.05), with the difference further increased by 21 days after birth (Figure 2).

Analysis of gut microbiota composition in the two groups at 3 days after birth
On the third day after birth, the NEC group exhibited significantly lower relative abundance of Firmicutes, significantly higher relative abundance of Proteobacteria, and significantly lower relative abundance of Actinobacteria compared with the CG (P<0.05). At 3 days after birth, intergroup comparisons at the genus level revealed that the NEC group had significantly lower relative abundances of Bifidobacterium and Lactobacillus, but higher relative abundances of Enterobacteriaceae, Klebsiella, Staphylococcus, and Fusobacterium compared to the CG (P<0.05) (Figure 3).

Effects of feeding methods on bacterial genera in two groups of pediatric patients
The proportion of patients primarily breastfed in the NEC group was significantly lower than that in the CG (P<0.05). The NEC group exhibited a longer initiation time for feeding, a longer time to achieve full enteral feeding, a higher incidence of feeding intolerance, and a lower proportion of probiotic use compared to the CG (P<0.05) (Figure 4).

Analysis of antibiotic use and microbiota relationship
The NEC group exhibited a significantly higher proportion of infants receiving antibiotics, a longer duration of antibiotic use, and a higher proportion of broad-spectrum antibiotic use compared to the CG (P<0.05) (Table 2). Further comparative analysis revealed that after antibiotic use, the Shannon index decreased in both groups compared to baseline (3 days after birth) (P<0.05). After antibiotic use, the NEC group had lower Shannon index than the CG (P<0.05) (Figure 5).
Table 2
| The use of antibiotics | NEC group (n=58) | Control group (n=116) | t/χ2 | P |
|---|---|---|---|---|
| Antibiotic use | 52 (89.66) | 78 (67.24) | 9.856 | 0.002 |
| Duration of use (days) | 12.67±4.89 | 6.34±3.12 | 9.876 | <0.001 |
| Broad-spectrum antibiotics | 48 (82.76) | 62 (53.45) | 13.567 | <0.001 |
Data are presented as number (%) or mean ± standard deviation. NEC, necrotizing enterocolitis.

Subgroup analysis of microbiota characteristics in PIs with different Bell stages
To further explore the relationship between disease severity and changes in gut microbiota, this study additionally included 18 PIs with Bell stage I for subgroup analysis. Based on Bell staging, included patients were categorized into 18 cases in stage I, 28 cases in stage II, and 30 cases in stage III. With increasing disease severity, both the Shannon index and Simpson index progressively decreased (P<0.05). The abundances of Bifidobacterium and Lactobacillus decreased, while those of Enterobacteriaceae and Klebsiella increased (P<0.05) (Figure 6).

Analysis of risk factors for NEC
Univariate analysis was conducted to identify risk factors for NEC, with the occurrence of NEC as the dependent variable and factors such as gestational age and birth weight as independent variables. Our results indicated that gestational age, birth weight, 5-minute Apgar score, mechanical ventilation, absence of breastfeeding as the primary feeding method, initiation time for feeds, feeding intolerance, absence of probiotic use, prolonged antibiotic use, low Shannon and Simpson indices were associated with the occurrence of NEC (using P<0.10 as the screening criterion for inclusion in multivariate analysis) (Table 3). Further analysis using multivariate logistic regression revealed that birth weight, feeding intolerance, duration of antibiotic use, broad-spectrum antibiotic use, and Shannon index were independent risk factors for the occurrence of NEC (P<0.05) (Table 4).
Table 3
| Variable | NEC group (n=58) | Control group (n=116) | t/c2 | P |
|---|---|---|---|---|
| Gestational age (weeks) | 29.34±2.18 | 30.12±2.45 | 2.057 | 0.04 |
| Birth weight (g) | 1,245.67±285.34 | 1,398.45±312.56 | 3.124 | 0.002 |
| Male | 32 (55.17) | 58 (50.00) | 0.408 | 0.52 |
| Caesarean section | 41 (70.69) | 75 (64.66) | 0.623 | 0.43 |
| 5-minute Apgar score | 7.82±1.24 | 8.36±0.98 | 3.084 | 0.002 |
| Antenatal hormone use | 38 (65.52) | 82 (70.69) | 0.478 | 0.49 |
| Mechanical ventilation | 45 (77.59) | 68 (58.62) | 5.982 | 0.01 |
| Breastfeeding as the primary feeding method | 15 (25.86) | 68 (58.62) | 5.438 | 0.02 |
| Initiation time for feeding (d) | 4.82±2.13 | 3.15±1.89 | 4.865 | <0.001 |
| Feeding intolerance | 35 (60.34) | 28 (24.14) | 19.867 | <0.001 |
| Probiotic use | 23 (39.66) | 67 (57.76) | 4.862 | 0.03 |
| Antibiotic use | 52 (89.66) | 78 (67.24) | 9.856 | 0.002 |
| Duration of antibiotic use (d) | 12.67±4.89 | 6.34±3.12 | 9.876 | <0.001 |
| Broad-spectrum antibiotic use | 48 (82.76) | 62 (53.45) | 13.567 | <0.001 |
| Shannon index (3d after birth) | 1.23±0.45 | 1.87±0.52 | 8.124 | <0.001 |
| Simpson index (3d after birth) | 0.42±0.18 | 0.68±0.23 | 7.689 | <0.001 |
Data are presented as number (%) or mean ± standard deviation. NEC, necrotizing enterocolitis.
Table 4
| Variable | β | SE | Wald χ2 | OR value | 95% CI | P value |
|---|---|---|---|---|---|---|
| Gestational age (weeks) | −0.089 | 0.078 | 1.298 | 0.915 | 0.784–1.068 | 0.26 |
| Birth weight (g) | −0.002 | 0.001 | 6.842 | 0.998 | 0.996–0.999 | 0.009 |
| 5-minute Apgar score | −0.234 | 0.156 | 2.256 | 0.791 | 0.583–1.075 | 0.13 |
| Mechanical ventilation | 0.445 | 0.378 | 1.384 | 1.560 | 0.744–3.270 | 0.24 |
| Breastfeeding as the primary feeding method | −0.398 | 0.345 | 1.332 | 0.672 | 0.342–1.320 | 0.25 |
| Feeding intolerance | 1.156 | 0.367 | 9.920 | 3.174 | 1.546–6.517 | 0.002 |
| Probiotic use | −0.287 | 0.352 | 0.665 | 0.751 | 0.377–1.496 | 0.42 |
| Duration of antibiotic use (d) | 0.189 | 0.045 | 17.642 | 1.208 | 1.106–1.320 | <0.001 |
| Broad-spectrum antibiotic use | 1.098 | 0.418 | 6.896 | 2.999 | 1.323–6.800 | 0.009 |
| Shannon index (3d after birth) | −1.287 | 0.456 | 7.974 | 0.276 | 0.113–0.674 | 0.005 |
CI, confidence interval; NEC, necrotizing enterocolitis; OR, odds ratio; SE, standard error.
Discussion
This study conducted a longitudinal cohort analysis of 174 PIs and found that pediatric patients in the NEC group exhibited significantly reduced gut microbiota diversity, delayed colonization of beneficial bacteria (Bifidobacterium and Lactobacillus), and alongside overgrowth of potential pathogens (Escherichia and Klebsiella). This finding aligns with previous studies (11,12), suggesting that gut dysbiosis plays a central role in the pathogenesis of NEC. Furthermore, the findings indicated that gut dysbiosis in PIs emerged as early as 3 days after birth, with differences progressively increasing over time. This suggests that the microbiological basis for NEC may be established in the early stages of life. The Shannon index, as a key indicator of microbiota diversity, was identified as an independent protective factor for NEC in multivariate analysis, suggesting its potential as a biomarker for NEC risk assessment.
Comparison of gut microbiota characteristics with previous studies
In the present study, PIs in the NEC group exhibited distinct gut dysbiosis, which is consistent with findings from multiple previous studies conducted both domestically and internationally. Previous research (13) reported that NEC infants exhibited Proteobacteria overgrowth and reduced microbial diversity before disease onset, which is in agreement with our results. A meta-analysis including multiple studies (14) demonstrated that increased relative abundance of Proteobacteria and decreased abundance of Firmicutes represent consistent findings in the intestines of infants with NEC. At the genus level, the relative abundance of Bifidobacterium was significantly lower in the NEC group compared with the CG, which is consistent with previous reports of delayed colonization of Bifidobacterium in infants with NEC (15). Notably, the relative abundance of Klebsiella in the NEC group was higher than that reported in most previous international studies. This discrepancy may be attributed to regional differences, variations in antibiotic use patterns, or differences in the timing of sample collection. To our knowledge, this study is the first to systematically analyze gut microbiota dynamics at multiple postnatal time points in a Chinese PI cohort. It revealed that gut microbiota differences between the NEC group and CG were already present by postnatal day 3 and expanded over time. This finding provides important evidence for the early prediction of NEC.
Effects of antibiotic use on the microbiota
This study found that the duration of antibiotic use was an independent risk factor for the occurrence of NEC, with broad-spectrum antibiotics posing an even higher risk. This is consistent with other research. Previous studies indicate that antibiotics disrupt the normal microecological balance by non-selectively killing intestinal bacteria, leading to a reduction in beneficial bacteria and the selective growth of resistant strains (16,17). The results of this study indicated that the Shannon index decreased significantly in both groups of pediatric patients after antibiotic use, but the decrease was more pronounced in the NEC group, suggesting that the gut microbiota of pediatric patients with NEC is more susceptible to antibiotic perturbation. These findings have certain clinical significance. It is recommended that in clinical practice, the indications for antibiotic use in PIs should be strictly followed, the duration of antibiotic treatment should be minimized, and unnecessary use of broad-spectrum antibiotics should be avoided; for pediatric patients who require antibiotic treatment, concurrent probiotic supplementation should be considered to maintain intestinal microecological balance (18,19).
Protective effect of feeding methods
Breastfeeding demonstrated a protective effect in univariate analysis. However, it did not reach statistical significance in multivariate analysis, which may be related to factors such as the sample size and source of cases in this study. Previous studies indicate that breast milk contains abundant bioactive substances such as oligosaccharides, immunoglobulins, and lactoferrin, which promote the growth of beneficial bacteria like bifidobacteria and inhibit the colonization of pathogenic bacteria (20-22). This plays a positive role in reducing the incidence of NEC. In this study, the breastfeeding rate in the NEC group was only 48.28%, significantly lower than 67.24% in the CG. This finding also suggests that breastfeeding education and support should be strengthened. Additionally, this study found that feeding intolerance was an independent risk factor for NEC, reflecting the interplay between intestinal immaturity and gut dysbiosis. The aforementioned phenomena may arise because feeding intolerance can easily lead to inadequate intestinal nutrient supply, thereby impairing intestinal mucosal development and barrier function. Concurrently, it may prolong the duration of parenteral nutrition in pediatric patients, ultimately increasing the risk of infection (8,23,24).
Potential value of probiotic supplementation
This study also found that probiotic use may offer potential protective value against NEC. Although the use of probiotics did not show an independent protective effect in the multivariate analysis of this study, the rate of probiotic use among pediatric patients in the NEC group was indeed lower than that in the CG. In this cohort study, the Shannon index at 14 days after birth was significantly higher in infants receiving probiotics (2.12±0.65) compared to those not receiving probiotics (1.78±0.58) (P=0.001). It suggests that probiotic supplementation may help maintain gut microbiota diversity. Evidence from previous studies (25-27) indicates that prophylactic use of probiotics may help reduce the incidence of NEC in PIs. The absence of statistical significance in this study may be attributed to the small sample size. Further large-scale prospective studies are needed to further investigate the protective effects of probiotics.
Early warning and intervention strategies
Based on the findings of this study, we propose the following strategies for NEC prevention and control: (I) establish a risk assessment system based on gut microbiota monitoring, with enhanced surveillance for high-risk pediatric patients exhibiting a Shannon index persistently below 1.5. Notably, approximately 10% of infants in the CG had a Shannon index below 1.5. Although they did not develop NEC, this suggests that these infants may be at higher risk of NEC and require close monitoring; (II) employ strategies to reduce unnecessary antibiotic use; (III) strengthen breastfeeding support and establish a milk bank to ensure PIs have access to breast milk; (IV) administer probiotics prophylactically to high-risk pediatric patients, preferably using a combination of Bifidobacterium and Lactobacillus; (V) improve feeding strategies by implementing standardized feeding protocols and promptly identifying and managing feeding and tolerances.
Limitations
Although this study provides certain recommendations for reducing the incidence of NEC in PIs, it also presents several limitations. For instance, the retrospective study design may introduce selection bias and information bias; the relatively small sample size limited the statistical power of subgroup analyses; only the gut microbiota changes within up to 21 days after birth were observed; the maternal factors were insufficiently assessed; and analyses of microbiome beta diversity were absent. These limitations warrant further refinement in subsequent studies.
To address the aforementioned limitations, future research will focus on conducting multicenter prospective cohort studies to expand sample sizes and extend follow-up periods. Randomized controlled trials of probiotic interventions will be conducted to clarify their protective effects. Additionally, efforts will be made to investigate the mechanisms of microbiota-host interactions, especially the impact of microbiota on intestinal immune development.
Conclusions
The pattern of the gut microbiota establishment in PIs is closely associated with the occurrence of NEC, which is characterized by reduced microbiota diversity, decreased beneficial bacteria, and increased potential 319 pathogens. Birth weight, feeding intolerance, and antibiotic use are significant risk factors for NEC, while a higher Shannon index offers protective effects. Clinically, attention should be paid for monitoring the gut microbiota of PIs, judicious use of antibiotics, promotion of breastfeeding, and timely supplementation with probiotics to reduce the risk of NEC and improve the prognosis for PIs.
Acknowledgments
None.
Footnote
Reporting Checklist: The authors have completed the STROBE reporting checklist. Available at https://tp.amegroups.com/article/view/10.21037/tp-2025-aw-713/rc
Data Sharing Statement: Available at https://tp.amegroups.com/article/view/10.21037/tp-2025-aw-713/dss
Peer Review File: Available at https://tp.amegroups.com/article/view/10.21037/tp-2025-aw-713/prf
Funding: This work was supported by
Conflicts of Interest: All authors have completed the ICMJE uniform disclosure form (available at https://tp.amegroups.com/article/view/10.21037/tp-2025-aw-713/coif). All authors reports that this work was supported by project titled “Neonatal gut microbiota and bacterial inflammation, Regulatory effects of Bifidobacterium longum and its application in zebrafish model”. The authors have no other conflicts of interest to declare.
Ethical Statement: The authors are accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved. The study protocol was approved by the ethics committee of Wenzhou People’s Hospital. The study was conducted in accordance with the Declaration of Helsinki and its subsequent amendments. Since this retrospective study did not involve any additional interventions, the requirement for informed consent was waived by the hospital’s ethics committee.
Open Access Statement: This is an Open Access article distributed in accordance with the Creative Commons Attribution-NonCommercial-NoDerivs 4.0 International License (CC BY-NC-ND 4.0), which permits the non-commercial replication and distribution of the article with the strict proviso that no changes or edits are made and the original work is properly cited (including links to both the formal publication through the relevant DOI and the license). See: https://creativecommons.org/licenses/by-nc-nd/4.0/.
References
- Hu X, Liang H, Li F, et al. Necrotizing enterocolitis: current understanding of the prevention and management. Pediatr Surg Int 2024;40:32. [Crossref] [PubMed]
- Roberts AG, Younge N, Greenberg RG. Neonatal Necrotizing Enterocolitis: An Update on Pathophysiology, Treatment, and Prevention. Paediatr Drugs 2024;26:259-75. [Crossref] [PubMed]
- Cai X, Liebe HL, Golubkova A, et al. A Review of the Diagnosis and Treatment of Necrotizing Enterocolitis. Curr Pediatr Rev 2023;19:285-95. [Crossref] [PubMed]
- Neu J. Prevention of Necrotizing Enterocolitis. Clin Perinatol 2022;49:195-206. [Crossref] [PubMed]
- Monzon N, Kasahara EM, Gunasekaran A, et al. Impact of neonatal nutrition on necrotizing enterocolitis. Semin Pediatr Surg 2023;32:151305. [Crossref] [PubMed]
- Martin CA, Markel TA. Preface: Necrotizing enterocolitis. Semin Pediatr Surg 2023;32:151303. [Crossref] [PubMed]
- Kelleher ST, McMahon CJ, James A. Necrotizing Enterocolitis in Children with Congenital Heart Disease: A Literature Review. Pediatr Cardiol 2021;42:1688-99. [Crossref] [PubMed]
- David P, Claud EC. Necrotizing Enterocolitis and the Preterm Infant Microbiome. Adv Exp Med Biol 2024;1449:29-41. [Crossref] [PubMed]
- Maheshwari A, Traub TM, Garg PM, et al. Necrotizing Enterocolitis: Clinical Features, Histopathological Characteristics, and Genetic Associations. Curr Pediatr Rev 2022;18:210-25. [Crossref] [PubMed]
- Kiu R, Darby EM, Alcon-Giner C, et al. Impact of early life antibiotic and probiotic treatment on gut microbiome and resistome of very-low-birth-weight preterm infants. Nat Commun 2025;16:7569. [Crossref] [PubMed]
- Thänert R, Keen EC, Dantas G, et al. Necrotizing Enterocolitis and the Microbiome: Current Status and Future Directions. J Infect Dis 2021;223:S257-63. [Crossref] [PubMed]
- Sowden M, van Weissenbruch MM, Bulabula ANH, et al. Effect of a Multi-Strain Probiotic on the Incidence and Severity of Necrotizing Enterocolitis and Feeding Intolerances in Preterm Neonates. Nutrients 2022;14:3305. [Crossref] [PubMed]
- Wang Y, Jiang K, Xia Q, et al. Exploration of pathogenic microorganism within the small intestine of necrotizing enterocolitis. World J Pediatr 2024;20:165-72. [Crossref] [PubMed]
- Mani S, Hazra S, Hagan J, et al. Viral Infections and Neonatal Necrotizing Enterocolitis: A Meta-analysis. Pediatrics 2023;152:e2022060876. [Crossref] [PubMed]
- Samarra A, Cabrera-Rubio R, Martínez-Costa C, et al. The role of Bifidobacterium genus in modulating the neonate microbiota: implications for antibiotic resistance acquisition in early life. Gut Microbes 2024;16:2357176. [Crossref] [PubMed]
- Pace E, Yanowitz TD, Waltz P, et al. Antibiotic therapy and necrotizing enterocolitis. Semin Pediatr Surg 2023;32:151308. [Crossref] [PubMed]
- Cuna A, Morowitz MJ, Sampath V. Early antibiotics and risk for necrotizing enterocolitis in premature infants: A narrative review. Front Pediatr 2023;11:1112812. [Crossref] [PubMed]
- Li F, Hooi SL, Choo YM, et al. Progression of gut microbiome in preterm infants during the first three months. Sci Rep 2025;15:12104. [Crossref] [PubMed]
- Gill EM, Jung K, Qvist N, et al. Antibiotics in the medical and surgical treatment of necrotizing enterocolitis. A systematic review. BMC Pediatr 2022;22:66. [Crossref] [PubMed]
- Thai JD, Gregory KE. Bioactive Factors in Human Breast Milk Attenuate Intestinal Inflammation during Early Life. Nutrients 2020;12:581. [Crossref] [PubMed]
- Serce Pehlevan O, Benzer D, Gursoy T, et al. Synbiotics use for preventing sepsis and necrotizing enterocolitis in very low birth weight neonates: a randomized controlled trial. Clin Exp Pediatr 2020;63:226-31. [Crossref] [PubMed]
- Ma LY, Liu QQ, Wu L, et al. Effect of electroacupuncture at Zusanli (ST36) and Zhongwan (CV12) on intestinal nutritional feeding intolerance in patients with severe acute pancreatitis. Zhen Ci Yan Jiu 2021;46:312-7. [PubMed]
- Sun JK, Nie S, Chen YM, et al. Effects of permissive hypocaloric vs standard enteral feeding on gastrointestinal function and outcomes in sepsis. World J Gastroenterol 2021;27:4900-12. [Crossref] [PubMed]
- Beharry KD, Latkowska M, Valencia AM, et al. Factors Influencing Neonatal Gut Microbiome and Health with a Focus on Necrotizing Enterocolitis. Microorganisms 2023;11:2528. [Crossref] [PubMed]
- Razak A, Patel RM, Gautham KS. Use of Probiotics to Prevent Necrotizing Enterocolitis: Evidence to Clinical Practice. JAMA Pediatr 2021;175:773-4. [Crossref] [PubMed]
- Calvo LN, Greenberg RG, Gray KD. Safety and Effectiveness of Probiotics in Preterm Infants with Necrotizing Enterocolitis. Neoreviews 2024;25:e193-206. [Crossref]
- Nolan LS, Rimer JM, Good M. The Role of Human Milk Oligosaccharides and Probiotics on the Neonatal Microbiome and Risk of Necrotizing Enterocolitis: A Narrative Review. Nutrients 2020;12:3052. [Crossref] [PubMed]