Concurrent chemoradiotherapy with temozolomide in newly pediatric diffuse intrinsic pontine glioma: clinical outcomes and safety analysis
Highlight box
Key findings
• Diffuse intrinsic pontine glioma (DIPG) remains a devastating pediatric brainstem malignancy with uniformly poor prognosis. This study evaluated concurrent temozolomide with radiotherapy in 24 patients, demonstrating temporary 88% disease control at initial post-radiotherapy assessment (4±1 weeks post-treatment) and median survival of 12 months.
• Treatment was feasible with manageable hematologic toxicity and no treatment-related mortality. H3K27M mutations characterized the majority of cases, reflecting typical molecular landscape.
What is known and what is new?
• Standard radiotherapy yields 9–11 months median survival with 20–40% radiological response rates (defined as complete or partial response) in DIPG.
• This study demonstrates temporary disease control of 88% (50% partial response, 38% stable disease) at initial post-radiotherapy assessment, compared to the 20–40% response rates reported with radiotherapy alone. However, when comparing our 50% partial response rate to historical series, the improvement is modest and within the upper range of previously reported outcomes. The findings establish feasibility of concurrent temozolomide with manageable toxicity profiles.
What is the implication, and what should change now?
• These retrospective findings are hypothesis-generating and suggest that concurrent chemoradiotherapy warrants further investigation in prospective randomized controlled trials before any recommendations can be made regarding standard clinical practice. The modest improvements in disease control must be weighed against the treatment burden, and healthcare costs.
• Future priorities include molecular profiling for personalized approaches, combination strategies with targeted agents, and standardized international protocols to accelerate therapeutic development for this fatal malignancy.
Introduction
Background
Diffuse intrinsic pontine glioma (DIPG) accounts for 10–15% of pediatric central nervous system malignancies and represents the leading cause of brain tumor mortality in children (1). These tumors infiltrate the brainstem, precluding gross total resection due to critical anatomical constraints and the high risk of permanent neurological sequelae (2). The infiltrative nature of these lesions, combined with their location within eloquent brain structures, creates unique therapeutic challenges that distinguish DIPG from other pediatric brain tumors.
Peak incidence occurs at 6–7 years, with most cases diagnosed between ages 5–9 years (3). Clinical presentation typically includes progressive cranial nerve palsies, ataxia, and pyramidal tract dysfunction, often developing over weeks to months. The rapidity of symptom progression frequently necessitates urgent therapeutic intervention, though treatment options remain limited.
Molecular characterization has identified histone H3K27M mutations in approximately 70–80% of DIPGs, conferring particularly aggressive behavior and poor prognosis (4,5). This mutation disrupts normal chromatin regulation and has emerged as both a prognostic biomarker and potential therapeutic target. Additional molecular alterations, including TP53 mutations and PDGFRA amplifications, further define the biological landscape of these tumors.
Despite decades of clinical investigation, standard therapy remains focal radiotherapy, providing transient symptom relief but minimal survival benefit (6). Historical median survival ranges from 9–11 months, with fewer than 10% of patients surviving beyond two years. The blood-brain barrier presents additional challenges for systemic therapy delivery, limiting the effectiveness of many chemotherapeutic agents.
Temozolomide, an oral alkylating agent with favorable blood-brain barrier penetration, has demonstrated efficacy in adult glioblastoma and select pediatric brain tumors (7,8). Its dual mechanism of action includes direct cytotoxic effects through DNA methylation and radiosensitizing properties that enhance radiation-induced tumor cell death (9). The rationale for concurrent administration with radiation therapy stems from preclinical studies demonstrating synergistic antitumor effects.
Objective
We report our institutional experience treating newly diagnosed pediatric DIPG patients with concurrent chemoradiotherapy followed by adjuvant temozolomide, analyzing response rates, survival outcomes, and comprehensive safety profiles. We present this article in accordance with the STROBE reporting checklist (available at https://tp.amegroups.com/article/view/10.21037/tp-2025-1-942/rc).
Methods
Study population
This retrospective cohort included pediatric patients with newly diagnosed DIPG treated at the Pediatric Oncology Unit, Fondazione Policlinico Universitario Agostino Gemelli IRCCS from 2006 to 2020. Eligibility criteria were: age <18 years; radiographically confirmed DIPG based on characteristic magnetic resonance imaging (MRI) findings; adequate performance status to complete planned radiotherapy course; adequate organ function (hemoglobin >8 g/dL, absolute neutrophil count >1,500/mm3, platelets >100,000/mm3, AST/ALT <2.5× upper limit normal, creatinine <1.5× normal); Karnofsky/Lansky performance score ≥50. Patients with prior chemotherapy, radiotherapy, or significant comorbidities precluding treatment were excluded.
Diagnosis was established through multidisciplinary review incorporating clinical presentation, neuroimaging findings, and histological confirmation when safely obtainable. MRI characteristics supporting DIPG diagnosis included T2-hyperintense lesions involving >50% of the pons with accompanying clinical symptoms.
Treatment protocol
All patients received focal external beam radiotherapy with concurrent temozolomide according to standardized institutional protocols. Radiation planning utilized computed tomography (CT)-MRI fusion with clinical target volume (CTV1) defined as gross tumor plus margin that evolved over the study period. Early cases [2006–2012] utilized margins of 1.5–3.0 cm, reflecting institutional practices at that time. Following publication of contemporary guidelines and increased understanding of DIPG infiltration patterns, later cases [2013–2020] adopted more conformal margins of 1.0–1.5 cm. Margins were adjusted for anatomical boundaries including the tentorium cerebelli, skull base, and cervicomedullary junction. The tentorium was not routinely used as a hard boundary (clipped), but CTV margins were modified to respect this anatomical structure when appropriate. Planning target volume incorporated additional 3–5 mm margins for setup uncertainties and organ motion.
Sequential boost technique delivered 45 Gy to CTV1, then 59.4 Gy to reduced volume (CTV2) encompassing gross disease plus 1.0–1.5 cm margin. Treatment was administered in 1.8 Gy fractions over approximately 6.5 weeks using linear accelerator-based techniques with appropriate beam arrangements to minimize dose to critical structures. All patients received a single course of radiotherapy as described above, except for 2 patients who received salvage re-irradiation.
Concurrent temozolomide was administered orally at 75 mg/m2 daily throughout radiotherapy, including weekends and holidays. Dose modifications were implemented for grade ≥3 hematological toxicity or grade ≥2 non-hematological toxicity. After a mandatory 4±1 weeks rest interval, adjuvant therapy consisted of temozolomide 200 mg/m2 daily for 5 consecutive days every 28 days for up to 13 cycles or until progression/unacceptable toxicity.
Response assessment and follow-up
Tumor response was assessed by contrast-enhanced MRI at 4±1 weeks post-radiotherapy completion, then every 3 cycles during adjuvant therapy, and upon clinical suspicion of progression. Standard neuroimaging response criteria defined complete response (CR), partial response (PR), stable disease (SD), and progressive disease (PD). Response assessment incorporated both T2-fluid-attenuated inversion recovery (FLAIR) and contrast-enhanced T1-weighted sequences.
Clinical evaluations included neurological examination, performance status assessment, and toxicity grading at each visit. Quality of life measures were documented using age-appropriate validated instruments when feasible.
Statistical analysis
Survival analysis utilized Kaplan-Meier methodology with log-rank tests for group comparisons. Progression-free survival (PFS) was defined as time from diagnosis to documented radiological progression or death from any cause. Overall survival (OS) was calculated from diagnosis to death or last follow-up. Patients alive at analysis were censored at last contact date.
Univariate and multivariate analyses examined potential prognostic factors including age, sex, biopsy status, molecular markers, and treatment response. Statistical significance was set at P<0.05. All analyses were performed using appropriate statistical software with institutional review board approval.
Safety evaluation
Adverse events were graded per Common Terminology Criteria for Adverse Events (CTCAE) v5.0. Toxicity assessment occurred weekly during concurrent therapy, then at each cycle during adjuvant. Dose reductions of 25% were implemented for treatment delays >7 days due to toxicity. Treatment discontinuation criteria included grade 4 non-hematological toxicity or recurrent grade 3 hematological toxicity despite dose reduction.
All patients received trimethoprim-sulfamethoxazole prophylaxis against Pneumocystis carinii pneumonia during treatment. Supportive care included antiemetics, growth factor support when indicated, and aggressive management of treatment-related complications.
Ethical considerations
The study was conducted in accordance with the Declaration of Helsinki and its subsequent amendments. The study was approved by Università Cattolica del Sacro Cuore Institutional Review Board (approval No. DIPUSVSP-25-06-2164) and individual consent for this retrospective analysis was waived.
Results
Twenty-four patients were analyzed with comprehensive baseline characteristics detailed in Table 1. Median age was 7 years with broad age distribution reflecting typical DIPG demographics. Female predominance (71%) was observed, consistent with some published series. Tissue confirmation was obtained in 19 patients (79%) through stereotactic biopsy procedures, with H3K27M mutation identified in 14 cases (74%) among tested specimens.
Table 1
| Characteristic | Value |
|---|---|
| Age at enrollment (years) | |
| Median | 7 |
| range | 3–16 |
| Sex, n | |
| Male | 7 |
| Female | 17 |
| Biopsy performed, n/N [%] | 19/24 [79] |
| H3K27M mutation, n/N [%] | 14/19 [74] |
| Metastases at diagnosis | 0 |
| Time from diagnosis to RT start (days) | |
| Median | 24 |
| Range | 13–62 |
| Duration of RT (weeks) | |
| Median | 6.9 |
RT, radiotherapy.
No patients presented with metastatic disease at diagnosis, though 3 patients (13%) demonstrated multifocal brainstem involvement. Baseline performance status was generally favorable, with 83% of patients maintaining Karnofsky/Lansky scores ≥70. Median time from symptom onset to diagnosis was 6 weeks (range, 2–16 weeks), reflecting typical clinical presentation patterns.
Treatment delivery was generally successful with 92% of patients completing planned radiotherapy without significant interruptions. Median radiation therapy duration was 47 days (range, 42–56 days). Concurrent temozolomide was administered at full dose in 21 patients (88%), with dose reductions required in 3 patients due to early hematological toxicity.
Treatment response
Post-radiotherapy response assessment revealed encouraging initial disease control rates. PR was achieved in 12 patients (50%), SD in 9 patients (38%), and PD in 3 patients (12%), yielding an overall disease control rate of 88%. No CRs were observed, consistent with the infiltrative nature of DIPG. This disease control was observed at the first post-treatment imaging assessment (4±1 weeks after radiotherapy completion) and does not reflect long-term control, as all patients ultimately experienced disease progression.
Response duration varied considerably, with responding patients maintaining benefit for median 7 months (range, 3–18 months). Patients achieving PR demonstrated superior subsequent survival compared to those with SD (median 14.5 vs. 9.2 months, P=0.041).
Time to progression showed significant heterogeneity, with 25% of patients progressing within 6 months and 10% maintaining disease control beyond 18 months. Patterns of progression included local recurrence (75%), distant central nervous system (CNS) spread (15%), and combined local/distant failure (10%).
Survival outcomes
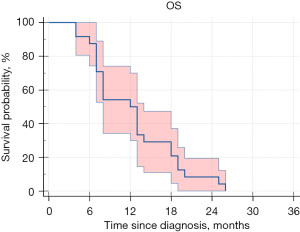
Median OS was 12.0 months [95% confidence interval (CI): 7.0–14.0 months], comparing favorably with historical controls receiving radiotherapy alone. OS probabilities were 54% at 12 months, 28% at 18 months, and 8% at 24 months. Three patients (13%) achieved long-term survival exceeding 18 months, with one patient surviving 28 months before ultimate progression (Figure 1).
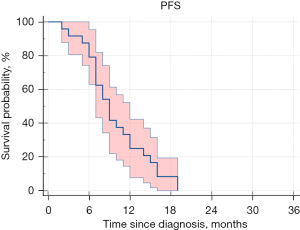
Median PFS was 9.0 months (95% CI: 7.0–12.0 months), with corresponding rates of 25% at 12 months, 4% at 18 months, and 0% at 24 months. All patients had experienced disease progression by 24-month follow-up (Figure 2). Subgroup analysis revealed trends toward improved outcomes in younger patients (≤6 years) compared to older children (median OS 14.2 vs. 10.1 months, P=0.18), though statistical significance was not achieved given limited sample size. H3K27M mutation status did not significantly impact survival outcomes in this cohort.
Safety profile and treatment tolerance
Safety analysis revealed distinct toxicity patterns between concurrent and adjuvant phases (Table 2). During concurrent therapy, grade 2 emesis represented the most frequent adverse event, affecting 8 patients (33%). Acute radiation-related effects included mild dermatitis (25%) and fatigue (42%), both generally manageable with supportive care measures.
Table 2
| Toxicity type | Phase | Grade | No. of patients | Percentage (%) |
|---|---|---|---|---|
| Vomiting | RT + concurrent TMZ | II | 8 | 33 |
| Leucopenia | Adjuvant TMZ | III–IV | 4 | 17 |
| Thrombocytopenia | Adjuvant TMZ | III–IV | 6 | 25 |
| Anemia | Adjuvant TMZ | III–IV | 0 | 0 |
| Serious infections | Overall treatment | Any grade | 0 | 0 |
RT, radiotherapy; TMZ, temozolomide.
Adjuvant phase toxicity was dominated by hematological effects, reflecting temozolomide’s myelosuppressive properties. Grade 3–4 leucopenia occurred in 4 patients (17%), necessitating dose reductions and treatment delays. Thrombocytopenia of similar severity affected 6 patients (25%), requiring platelet transfusions in 2 cases. No instances of grade 3–4 anemia were observed.
Secondary complications included opportunistic infections in 2 patients despite prophylaxis, both responding to appropriate antimicrobial therapy without treatment discontinuation. No treatment-related endocrinopathies and deaths occurred, and quality of life parameters remained stable during active therapy in most patients.
Treatment discontinuation due to toxicity occurred in 3 patients (13%), all due to recurrent grade 3 hematological effects during adjuvant therapy. Dose reductions were implemented in 8 patients (33%), primarily for hematological toxicity. Despite these modifications, treatment tolerance was generally acceptable with appropriate monitoring and supportive care interventions.
Discussion
This single-institution experience demonstrates that concurrent chemoradiotherapy achieves meaningful initial response rates in pediatric DIPG, with 88% of patients experiencing disease control following primary treatment. The 50% PR rate compares favorably with historical radiotherapy-alone series reporting 20–40% response rates (10,11). This improvement suggests potential synergistic effects between radiation therapy and temozolomide, possibly mediated through radiosensitization mechanisms and independent cytotoxic activity.
The observed response duration of 7 months aligns with expected treatment benefits, though substantial variability exists among individual patients. Factors contributing to this heterogeneity likely include molecular characteristics, tumor location, baseline performance status, and individual response to treatments.
Our median OS of 12 months aligns closely with contemporary reports ranging from 9–15 months for DIPG patients receiving multimodal therapy (12,13). The 12-month OS rate of 54% compares favorably to many published series, though direct comparisons remain challenging due to patient selection variations, treatment protocol differences, and institutional factors affecting outcome reporting.
The identification of long-term survivors (>18 months) in 13% of our cohort provides cautious optimism, though these patients ultimately succumbed to disease progression. This observation underscores the need for improved adjuvant strategies and novel approaches targeting mechanisms of therapeutic resistance.
The high prevalence of H3K27M mutations (74%) reflects typical DIPG molecular characteristics and confirms appropriate patient selection. This alteration associates with particularly aggressive biological behavior and resistance to conventional therapies, potentially explaining limited long-term survival despite initial treatment responses (14,15). The lack of significant survival differences between H3K27M-positive and negative cases may reflect small sample size limitations or indicate that treatment effectiveness transcends specific molecular subtypes.
Future investigations should incorporate comprehensive molecular profiling to identify predictive biomarkers and guide personalized treatment approaches. Emerging therapeutic targets include EZH2 inhibitors, histone deacetylase modulators, and immunotherapeutic approaches specifically designed for H3K27M-mutant tumors (16,17).
Safety analysis revealed manageable toxicity profiles consistent with temozolomide’s established adverse event spectrum. The predominance of hematological toxicity during adjuvant therapy reflects expected drug effects and was successfully managed through dose modifications and supportive care measures. The absence of treatment-related mortality demonstrates the feasibility of this approach in the pediatric population. Quality of life considerations remain paramount in DIPG management given limited curative potential. Our experience suggests that concurrent chemoradiotherapy provides meaningful symptom control without excessive toxicity burden, supporting its continued use while novel approaches are developed.
Our findings should be compared with other temozolomide-based approaches in DIPG. Broniscer et al. evaluated adjuvant temozolomide following radiotherapy (without concurrent administration) in 26 children with DIPG, reporting median OS of 8.7 months and 1-year OS of 37.5%. They concluded that adjuvant temozolomide after radiotherapy did not improve survival compared to historical controls (18). In contrast, our study utilized concurrent temozolomide during radiotherapy followed by adjuvant therapy, achieving a median OS of 12.0 months and 1-year OS of 54%. While direct comparison is limited by differences in study design and patient populations, our concurrent approach may offer modest improvements, though these must be interpreted cautiously given the retrospective nature of our study. However, the benefit of adding temozolomide to radiotherapy in DIPG remains still controversial. Izzuddeen et al. conducted a randomized controlled trial comparing conventional radiotherapy alone (60 Gy/30 fractions) versus hypofractionated radiotherapy (39 Gy/13 fractions) with concurrent and adjuvant temozolomide in 35 patients. They found no significant survival difference between arms (median OS 11 vs. 12 months, P=0.208), with 28% of patients in the chemoradiotherapy arm developing grade 3–4 hematological toxicity (19). Our study, utilizing conventional fractionation (59.4 Gy) with concurrent and adjuvant temozolomide, achieved median OS of 12.0 months, similar to both arms of the Izzuddeen study. These comparable outcomes across different treatment approaches raise important questions about whether the addition of temozolomide provides meaningful survival benefit beyond radiotherapy alone, particularly when considering the additional toxicity burden.
It is important to acknowledge that the benefit of adding temozolomide to radiotherapy in DIPG remains highly controversial. Multiple studies have failed to demonstrate statistically significant survival improvements with combination therapy compared to radiotherapy alone.
The Children’s Oncology Group study ACNS0126 prospectively evaluated concurrent and adjuvant temozolomide in 63 children with DIPG, reporting median OS of 9.6 months and 1-year EFS of only 14%, which was actually inferior to their historical control (21.9%, P=0.96). The authors concluded that temozolomide was not more effective than previously reported regimens (13).
Furthermore, Zaghloul et al. in their large prospective randomized trial of 253 DIPG patients treated with radiotherapy alone (three different fractionation schedules) achieved median OS of 8.75 months and PFS of 7.5 months, with 1-year OS rates of 29.2%. These results were achieved without the hematological toxicity, treatment burden, and healthcare costs associated with systemic chemotherapy (20).
These findings collectively suggest that the addition of temozolomide might not provide substantial survival benefit. Our study, being retrospective and lacking a concurrent control group receiving radiotherapy alone, cannot definitively resolve this controversy.
Several limitations merit acknowledgment in interpreting our results. The retrospective design introduces potential selection bias and limits comprehensive toxicity assessment. Small sample size precludes definitive conclusions regarding optimal patient selection criteria or treatment modifications. In addition, almost all our patients didn’t receive re-irradiation at progression. Janssens et al. reported improved OS in 31 children with DIPG who received re-irradiation at first progression (median OS 13.7 months from diagnosis) compared to matched controls who did not (median OS 10.3 months), despite similar PFS. The potential survival benefit of selective re-irradiation may have influenced outcomes in contemporary series, and the absence of this salvage option in our cohort may affect direct comparisons with other published studies (21). The lack of a contemporaneous control group receiving radiotherapy alone prevents definitive attribution of survival benefits to temozolomide addition. However, ethical considerations preclude randomized studies withholding potentially beneficial therapy in this universally fatal disease.
A critical consideration in evaluating any DIPG treatment is the balance between potential survival benefit and treatment-related morbidity, particularly in a disease with such uniformly poor prognosis. In our cohort, 42% of patients experienced grade 3–4 hematological toxicity, requiring dose modifications (33%), treatment delays, and in some cases blood product support and antimicrobial therapy. Three patients (13%) discontinued treatment due to recurrent grade 3 hematological toxicity. For children with such limited life expectancy (median survival 12 months), the question arises whether modest or uncertain survival improvements justify the additional toxicity burden, healthcare costs, frequent hospital visits, and emotional stress for patients and families. This risk-benefit consideration must be carefully discussed with families when making treatment decisions, presenting both the limited potential benefits and substantial toxicity risks.
Future therapeutic developments must incorporate novel biological insights, including precision medicine approaches targeting specific molecular alterations (22-25). Combination strategies utilizing immunotherapy, targeted agents, and novel drug delivery systems warrant priority investigation. Enhanced understanding of the tumor microenvironment and mechanisms of therapeutic resistance will guide next-generation treatment protocols.
Conclusions
Concurrent chemoradiotherapy with temozolomide produces encouraging initial response rates in pediatric DIPG with acceptable toxicity profiles suitable for the pediatric population. However, these retrospective findings require validation in prospective randomized trials before definitive treatment recommendations can be made. The substantial hematological toxicity observed, and treatment burden must be carefully weighed against the uncertain survival benefits when counseling families about treatment options.
Given the conflicting evidence in the literature regarding the benefit of adding chemotherapy to radiotherapy, and considering the substantial toxicity and treatment burden, the role of concurrent chemoradiotherapy in DIPG management remains uncertain and requires further clarification through well-designed prospective studies.
The observed disease control rate of 88% and median survival of 12 months represent meaningful improvements over historical controls, though long-term outcomes remain substantially limited.
These results support continued investigation of temozolomide-based approaches while emphasizing urgent need for innovative treatment strategies incorporating novel biological targets and enhanced therapeutic delivery methods. The identification of molecular biomarkers and development of personalized treatment approaches represent critical priorities for improving outcomes in this devastating pediatric malignancy.
Optimal DIPG management requires coordinated multidisciplinary expertise encompassing neuro-oncology, radiation oncology, neurosurgery, and comprehensive supportive care services. While current therapies provide limited survival benefit, they offer important symptom palliation and quality-of-life improvements for patients and families confronting this challenging diagnosis. Continued collaborative research efforts remain essential for developing curative approaches for future DIPG patients.
Acknowledgments
The authors thank “Fondazione per l’Oncologia Pediatrica” for their dedicated patient care and scientific support.
Footnote
Reporting Checklist: The authors have completed the STROBE reporting checklist. Available at https://tp.amegroups.com/article/view/10.21037/tp-2025-1-942/rc
Data Sharing Statement: Available at https://tp.amegroups.com/article/view/10.21037/tp-2025-1-942/dss
Peer Review File: Available at https://tp.amegroups.com/article/view/10.21037/tp-2025-1-942/prf
Funding: None.
Conflicts of Interest: All authors have completed the ICMJE uniform disclosure form (available at https://tp.amegroups.com/article/view/10.21037/tp-2025-1-942/coif). The authors have no conflicts of interest to declare.
Ethical Statement: The authors are accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved. The study was conducted in accordance with the Declaration of Helsinki and its subsequent amendments. The study was approved by the Università Cattolica del Sacro Cuore Institutional Review Board (approval No. DIPUSVSP-25-06-2164) and individual consent for this retrospective analysis was waived.
Open Access Statement: This is an Open Access article distributed in accordance with the Creative Commons Attribution-NonCommercial-NoDerivs 4.0 International License (CC BY-NC-ND 4.0), which permits the non-commercial replication and distribution of the article with the strict proviso that no changes or edits are made and the original work is properly cited (including links to both the formal publication through the relevant DOI and the license). See: https://creativecommons.org/licenses/by-nc-nd/4.0/.
References
- Ostrom QT, Patil N, Cioffi G, et al. CBTRUS Statistical Report: Primary Brain and Other Central Nervous System Tumors Diagnosed in the United States in 2013-2017. Neuro Oncol 2020;22:iv1-iv96. [Crossref] [PubMed]
- Grimm SA, Chamberlain MC. Brainstem glioma: a review. Curr Neurol Neurosci Rep 2013;13:346. [Crossref] [PubMed]
- Hoffman LM, Veldhuijzen van Zanten SEM, Colditz N, et al. Clinical, Radiologic, Pathologic, and Molecular Characteristics of Long-Term Survivors of Diffuse Intrinsic Pontine Glioma (DIPG): A Collaborative Report From the International and European Society for Pediatric Oncology DIPG Registries. J Clin Oncol 2018;36:1963-72. [Crossref] [PubMed]
- Schwartzentruber J, Korshunov A, Liu XY, et al. Driver mutations in histone H3.3 and chromatin remodelling genes in paediatric glioblastoma. Nature 2012;482:226-31. [Crossref] [PubMed]
- Khuong-Quang DA, Buczkowicz P, Rakopoulos P, et al. K27M mutation in histone H3.3 defines clinically and biologically distinct subgroups of pediatric diffuse intrinsic pontine gliomas. Acta Neuropathol 2012;124:439-47. [Crossref] [PubMed]
- Janssens GO, Jansen MH, Lauwers SJ, et al. Hypofractionation vs conventional radiation therapy for newly diagnosed diffuse intrinsic pontine glioma: a matched-cohort analysis. Int J Radiat Oncol Biol Phys 2013;85:315-20. [Crossref] [PubMed]
- Stupp R, Mason WP, van den Bent MJ, et al. Radiotherapy plus concomitant and adjuvant temozolomide for glioblastoma. N Engl J Med 2005;352:987-96. [Crossref] [PubMed]
- Cohen KJ, Pollack IF, Zhou T, et al. Temozolomide in the treatment of high-grade gliomas in children: a report from the Children's Oncology Group. Neuro Oncol 2011;13:317-23. [Crossref] [PubMed]
- van Nifterik KA, van den Berg J, van der Meide WF, et al. Absence of the MGMT protein as well as methylation of the MGMT promoter predict the sensitivity for temozolomide. Br J Cancer 2010;103:29-35. [Crossref] [PubMed]
- Hargrave D, Bartels U, Bouffet E. Diffuse brainstem glioma in children: critical review of clinical trials. Lancet Oncol 2006;7:241-8. [Crossref] [PubMed]
- Jansen MH, Veldhuijzen van Zanten SE, Sanchez Aliaga E, et al. Survival prediction model of children with diffuse intrinsic pontine glioma based on clinical and radiological criteria. Neuro Oncol 2015;17:160-6. [Crossref] [PubMed]
- Bailey S, Howman A, Wheatley K, et al. Diffuse intrinsic pontine glioma treated with prolonged temozolomide and radiotherapy--results of a United Kingdom phase II trial (CNS 2007 04). Eur J Cancer 2013;49:3856-62. [Crossref] [PubMed]
- Cohen KJ, Heideman RL, Zhou T, et al. Temozolomide in the treatment of children with newly diagnosed diffuse intrinsic pontine gliomas: a report from the Children's Oncology Group. Neuro Oncol 2011;13:410-6. [Crossref] [PubMed]
- Castel D, Philippe C, Calmon R, et al. Histone H3F3A and HIST1H3B K27M mutations define two subgroups of diffuse intrinsic pontine gliomas with different prognosis and phenotypes. Acta Neuropathol 2015;130:815-27. [Crossref] [PubMed]
- Mackay A, Burford A, Carvalho D, et al. Integrated Molecular Meta-Analysis of 1,000 Pediatric High-Grade and Diffuse Intrinsic Pontine Glioma. Cancer Cell 2017;32:520-537.e5. [Crossref] [PubMed]
- Mohammad F, Weissmann S, Leblanc B, et al. EZH2 is a potential therapeutic target for H3K27M-mutant pediatric gliomas. Nat Med 2017;23:483-92. [Crossref] [PubMed]
- Hashizume R, Andor N, Ihara Y, et al. Pharmacologic inhibition of histone demethylation as a therapy for pediatric brainstem glioma. Nat Med 2014;20:1394-6. [Crossref] [PubMed]
- Broniscer A, Iacono L, Chintagumpala M, et al. Role of temozolomide after radiotherapy for newly diagnosed diffuse brainstem glioma in children: results of a multiinstitutional study (SJHG-98). Cancer 2005;103:133-9. [Crossref] [PubMed]
- Izzuddeen Y, Gupta S, Haresh KP, et al. Hypofractionated radiotherapy with temozolomide in diffuse intrinsic pontine gliomas: a randomized controlled trial. J Neurooncol 2020;146:91-5. [Crossref] [PubMed]
- Zaghloul MS, Nasr A, Tolba M, et al. Hypofractionated Radiation Therapy For Diffuse Intrinsic Pontine Glioma: A Noninferiority Randomized Study Including 253 Children. Int J Radiat Oncol Biol Phys 2022;113:360-8. [Crossref] [PubMed]
- Janssens GO, Gandola L, Bolle S, et al. Survival benefit for patients with diffuse intrinsic pontine glioma (DIPG) undergoing re-irradiation at first progression: A matched-cohort analysis on behalf of the SIOP-E-HGG/DIPG working group. Eur J Cancer 2017;73:38-47. [Crossref] [PubMed]
- Kaminskiy Y, Degtyarev V, Stepanov A, et al. CAR T cell therapy for central nervous system solid tumors: current progress and future directions. Front Immunol 2025;16:1600403. [Crossref] [PubMed]
- Perrone MG, Ruggiero A, Centonze A, et al. Diffuse Intrinsic Pontine Glioma (DIPG): Breakthrough and Clinical Perspective. Curr Med Chem 2021;28:3287-317. [Crossref] [PubMed]
- Meco D, Attinà G, Mastrangelo S, et al. Emerging Perspectives on the Antiparasitic Mebendazole as a Repurposed Drug for the Treatment of Brain Cancers. Int J Mol Sci 2023;24:1334. [Crossref] [PubMed]
- Weisbrod LJ, Thiraviyam A, Vengoji R, et al. Diffuse intrinsic pontine glioma (DIPG): A review of current and emerging treatment strategies. Cancer Lett 2024;590:216876. [Crossref] [PubMed]