Living donor liver transplantation in a pediatric patient with mitochondrial DNA depletion syndrome caused by two novel POLG1 mutations: a case report and literature review
Highlight box
Key findings
• An 11-month-old male with mitochondrial DNA depletion syndrome (mtDDS) due to two novel heterozygous POLG1 mutations (695G>A, 1735C>T; autosomal recessive) presented with liver failure/hypoglycemia without neurological symptoms (atypical for mtDDS).
• Liver biopsy confirmed mitochondrial dysfunction [respiratory chain complex (RCC) I/III activity 32%/28% of controls]; living donor liver transplantation (LDLT) from the father achieved full recovery with 6-month normal liver function/growth and no hypoglycemia recurrence.
What is known and what is new?
• mtDDS (caused by POLG1 mutations) typically involves neuro-hepatic/myopathic manifestations
• The identification of two pathogenic POLG1 mutations and an atypical mtDDS phenotype with isolated liver dysfunction.
What is the implication, and what should change now?
• Clinicians should suspect mtDDS in pediatric unexplained liver failure/hypoglycemia (even without neurological signs) and perform whole exome sequencing (WES) for genetic diagnosis.
• LDLT is a viable curative option for such patients; long-term follow-up is critical to monitor for late neurological complications of POLG1 mutations.
Introduction
Mitochondrial DNA depletion syndrome (mtDDS) is a group of rare genetic disorders characterized by a significant reduction in mitochondrial DNA (mtDNA) content, predominantly caused by mutations in genes encoding nuclear factors responsible for mtDNA maintenance, including POLG1. These disorders manifest with a diverse range of clinical symptoms, including myopathy, hepatopathy, and neurological deficits, complicating timely diagnosis and treatment (1,2). The prevalence of mtDDS is low, yet it poses significant clinical challenges, particularly in pediatric populations where early identification and intervention are crucial for improving outcomes (3). In this report, we present a unique case of an 11-month-old male infant diagnosed with mtDDS due to POLG1 mutations, who exhibited liver failure and recurrent hypoglycemia. The clinical presentation included elevated liver enzymes, jaundice, vomiting, and poor feeding, symptoms that are often nonspecific and can mimic other common pediatric illnesses (4,5). This highlights the diagnostic complexities associated with mtDDS, where the need for genetic testing, including whole exome sequencing (WES), becomes paramount for confirming the diagnosis and guiding appropriate management strategies (2,6).
Significantly, this case identifies two previously unreported mutations in the POLG1 gene (695G>A and 1735C>T) that contribute to the understanding of genetic variations linked to mtDDS. Notably, the absence of neurological manifestations in this patient represents a departure from typical presentations, underscoring the clinical heterogeneity of mtDDS (1). Furthermore, the successful outcome following liver transplantation not only illustrates the potential for considerable improvements in quality of life but also emphasizes the essential role of timely genetic testing in managing unexplained liver failure in infants (7). This case underscores the significance of recognizing mtDDSs in pediatric patients presenting with liver dysfunction and metabolic crises. The findings advocate for heightened awareness among clinicians regarding the potential genetic underpinnings of such cases, which can lead to early diagnosis and intervention, ultimately improving clinical outcomes. In summary, this report contributes valuable insights into the clinical spectrum of mtDDS, emphasizing the importance of genetic diagnosis and the potential for successful therapeutic interventions. We present this article in accordance with the CARE reporting checklist (available at https://tp.amegroups.com/article/view/10.21037/tp-2025-1-883/rc).
Case presentation
Chief complaint
An 11-month-old male infant exhibited symptoms of liver failure and recurrent hypoglycemia.
History of past illness
He was born uneventfully at a gestational age of 39 weeks and 3 days with a birth weight of 3,257 g. The patient initially presented with vomiting, primarily postprandial, which occurred approximately three to four times per day. The vomitus predominantly consisted of gastric contents with occasional mucus, and there was no evidence of blood. The infant also displayed mild abdominal distension without diarrhea. Approximately 4 months prior to admission, the infant experienced vomiting without significant provocation. This was followed by the onset of scleral icterus (total bilirubin, 56.32 µmol/L; direct bilirubin, 34.61 µmol/L) 3 months prior to admission, which was overlooked by the family. Then the patient underwent multiple examinations, including imaging, virus, serum amino acid, serum carnitine, urine organic acid and genetic testing for citrin deficiency. However, no abnormal findings were identified, and ambulatory follow-up was continued. Two months prior to admission, after receiving the measles, mumps, and rubella vaccine, the patient developed a fever with a maximum temperature of 38.9 ℃, which resolved with symptomatic treatment without chills or seizures. Upon further examination, the infant presented with listlessness and poor appetite, alongside reduced urine output. The patient was referred to a local hospital, where laboratory tests revealed significantly elevated liver enzymes: alanine aminotransferase at 2,500 U/L and aspartate aminotransferase at 814 U/L, and total bilirubin of 69.5 µmol/L, with direct bilirubin at 55.6 µmol/L. Additional laboratory results indicated hypoglycemia with blood glucose levels dropping to 1.7 mmol/L and elevated lactic acid levels at 6.81 mmol/L. Imaging studies and further assessments suggested hepatic enlargement and increased echogenicity on abdominal ultrasound, with multiple laboratory findings indicating liver dysfunction and glucose metabolic disorders.
Personal and family history
Both the patient’s father and mother are of Han ethnicity, with no known ancestry from other ethnic groups, and there was no hereditary history of congenital metabolic disorders in his family members.
History of present illness
He was referred to our center because of progressive impairment of liver function at 10 months of age. Examinations on admission revealed the following: body length, 69 cm (3% on age, Z score −1.7); body weight, 7,439 g (3% on age, Z score −1.95); jaundice, hepatosplenomegaly; physical examination revealed a liver span of 2 cm below the right costal margin with a moderate consistency; no superficial venous dilatation, palmar erythema, or other stigmata of chronic liver disease were observed; laboratory examination results presented in Table 1, on admission column; ultrasound and computed tomography (CT) showed liver diffuse lesions, portal hypertension, splenomegaly, ascites. As part of the metabolic and pre-transplant evaluation, ophthalmologic examination including fundoscopy and echocardiography were performed, revealing no significant abnormalities. A formal pre-transplant neurological assessment, including developmental evaluation, electroencephalogram (EEG), and brain magnetic resonance imaging (MRI), was conducted and showed no evidence of neurological involvement at that time.
Table 1
| Examination items | On admission | Before transplantation | On discharge | Normal range |
|---|---|---|---|---|
| ALT (U/L) | 894 | 189 | 15 | 8–71 |
| AST (U/L) | 2352.7 | 687.1 | 31.6 | 21–80 |
| GGT (U/L) | 71 | 74 | 10 | 6–31 |
| Total bilirubin (μmol/L) | 151.67 | 97.03 | 21.31 | 3.42–21.00 |
| Direct bilirubin (μmol/L) | 89.77 | 55.27 | 14.93 | 0.00–6.84 |
| INR | 2.28 | 1.40 | 1.11 | 0.8–1.2 |
| pH | 7.357 | 7.342 | 7.416 | 7.35–7.45 |
| pCO2 (mmHg) | 33.5 | 36.7 | 39.4 | 35–45 |
| Lactate (mmol/L) | 4.5 | 3.1 | 0.9 | 0.7–2.0 |
| Ammonia (μmol/L) | 67 | 69 | 56 | 0–72 |
| Urea nitrogen (mmol/L) | 1.32 | 2.23 | 1.78 | 1.1–5.9 |
ALT, alanine aminotransferase; AST, aspartate aminotransferase; GGT, gamma-glutamyl transpeptidase; INR, international normalized ratio; pCO2, partial pressure of carbon dioxide; pH, potential of hydrogen.
The patient was managed in the intensive care unit with a comprehensive treatment plan, including liver-protective agents such as glycyrrhizin and glutathione, vitamin B6 for gastrointestinal symptoms relief, and antibiotics for infection prophylaxis. Intermittent hemodiafiltration and plasma exchange were required to maintain water balance and clotting function, and continuous glucose supplementation was given since hypoglycemia was easily triggered. Additionally, vitamin K1 was administered to improve coagulopathy, and ursodeoxycholic acid was used for liver support. Daily monitoring of blood glucose levels showed fluctuations between 4–8 mmol/L, with the lowest recorded level at 2.6 mmol/L. After treatment, the patient’s temperature returned to normal, jaundice subsided, and significant improvements were observed in metabolic and liver function parameters compared with the patient’s condition upon admission.
Pathological and genetic diagnosis
The liver biopsy specimens showed diffuse hepatocyte swelling with ballooning degeneration. Approximately 50% of hepatocytes exhibited bullae and microvesicular steatosis, accompanied by intrahepatic cholestasis and punctate necrosis. The portal tracts demonstrated fibrous tissue proliferation and bile duct hyperplasia, with predominant lymphocyte and neutrophil infiltration. Occasional eosinophilic cell infiltration was observed, along with interstitial inflammation and bile plugs. Early-stage pseudolobule formation was noted (Figure 1). Histopathological examination (electron microscopy) revealed mild hepatocyte swelling without glycogen accumulation. Mitochondria were sparsely distributed and mildly swollen, with numerous small lipid vacuoles present. Some mitochondria showed significant enlargement, elongation, and an abnormal ring-shaped morphology. The mitochondrial cristae appeared dense, and the matrix density was increased. No special crystals or inclusions were observed. This pathological result suggests that the patient may have a mitochondrial disease, which may be hereditary.

Respiratory chain complex (RCC) activities were measured in frozen liver tissue obtained via biopsy. Spectrophotometric assays were used to determine the activities of complexes I, II, III, and IV. Activities were normalized to citrate synthase activity to account for mitochondrial content. Age-matched pediatric controls (n=10) with normal liver histology were used to establish reference ranges. Biochemical analysis of liver tissue further confirmed mitochondrial dysfunction: RCC I and III activities were reduced to 32% and 28% of age-matched controls, respectively, while complex II (nuclear-encoded) activity remained normal—supporting a mtDNA-dependent defect (Table 2).
Table 2
| RCC I | RCC II | RCC III | RCC IV | CS | |
|---|---|---|---|---|---|
| Ratio to control (%) | 10.3 | 11.8 | 14.9 | 122.2 | 111.7 |
| Ratio to CS (%) | 8.3 | 9.5 | 11.9 | 103.4 | – |
CS, citrate synthase; RCC, respiratory chain complex.
Furthermore, a WES was performed using next-generation sequencing. Genetic testing was performed to investigate mtDDS-related genes: first excluding mutations in Deoxyguanosine Kinase (DGUOK) and Mitochondrial Inner Membrane Protein Mpv17 (MPV17) (common causes of hepatocerebral mtDDS), then analyzing POLG1 via Sanger sequencing. This confirmed the presence of two previously unreported heterozygous mutations: 695G>A (chr15:87873472, predicting p.Arg232His) and 1735C>T (chr15:89868895, predicting p.Arg579Trp). Parental testing revealed the father was a carrier of 695G>A and the mother of 1735C>T, confirming autosomal recessive inheritance (Figure 2). These mutations were absent in healthy controls, and in silico analysis (PolyPhen-2, SIFT) predicted both to be pathogenic. Interspecies alignment showed the affected amino acids (Arg232, Arg579) are conserved across mammals (human, macaca, rat, mouse) and Drosophila, further supporting their functional importance. These results contributed to the diagnosis of mtDDS with liver failure.

Treatment
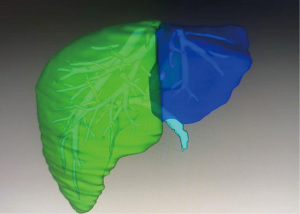
Given the unknown etiology of the liver lesion, we conducted a liver biopsy following adequate safety protocols. Liver biopsy showed early-stage pseudolobule formation with marked fibrosis and marked hepatocyte steatosis (50%) along with giant cell transformation, indicating that liver involvement was considered irreversible. Following stabilization (Table 1), the patient underwent living donor liver transplantation (LDLT) 17 days after admission, using a left lateral segment graft from the patient’s father (Figure 3). The operation time was 5 h 23 min. The graft weighed 196 g. The graft-to-recipient weight ratio was 2.65%. The cold ischemic time was 75 min and the warm ischemic time was 4 min (Figure 4). The procedure was uneventful, with postoperative management including immunosuppressive therapy and nutritional support. The postoperative course was uneventful, and liver function improved smoothly. The high preoperative serum lactate levels also improved quickly after LDLT. All procedures performed in this study were in accordance with the ethical standards of the institutional and/or national research committee(s) and with the Declaration of Helsinki and its subsequent amendments. Written informed consent was obtained from the patient’s legal guardian for the publication of this case report and accompanying images. A copy of the written consent is available for review by the editorial office of this journal.

Outcomes
The patient was discharged 21 days after surgery. Follow-up to 6 months post-transplant, the patient demonstrated normal growth and development, with no recurrence of liver function abnormalities or episodes of hypoglycemia (Table 1). Liver biopsy of the graft showed that no significant inflammation or rejection in the managed area, no abnormalities in the bile ducts, and no liver fibrosis (Figure 5).

Discussion
The case of a POLG1 mutation leading to mtDDS presents unique diagnostic challenges, particularly when clinical manifestations deviate from the typical neurological symptoms often associated with this condition. In this instance, the patient exhibited recurrent hypoglycemia and liver failure without neurological impairment, a variation not extensively documented in existing literature. Mazziotta et al. reported a case of a 3-month-old girl died from severe metabolic acidosis and developing liver dysfunction in 1992, which was similar to this case (8). However, due to the limitation of the time, the pathogenic gene was not identified. Bortot et al. found two mutations (1868T>G and 2263A>G) which had similar manifestations of the disease, and the patient died from liver failure as well (9). In 2023, Kadohisa et al. reported a case of children with myocerebrohepatopathy spectrum (MCHS) caused by homozygous mutation of POLG gene who received live liver transplantation with good outcome (10). The decision to proceed with liver transplantation in a patient with POLG mutations requires careful consideration of the risk of post-transplant neurological deterioration, a hallmark of the Alpers-Huttenlocher spectrum. In this case, the multidisciplinary transplant team extensively reviewed the patient’s condition. The absence of pre-existing neurological symptoms, normal developmental assessment, EEG, and brain MRI provided reassurance regarding the immediate neurological risk. Furthermore, the patient’s progressive liver failure posed an imminent threat to survival, with no alternative curative therapy available. The decision was made after thorough counseling of the family regarding the potential risks and benefits, including the possibility of future neurological decline. Previous studies have demonstrated that POLG1 mutations can result in a spectrum of clinical manifestations including Alpers-Huttenlocher syndrome and progressive external ophthalmoplegia, which are commonly associated with significant neurological deficits (11,12). The identification of novel mutations such as 695G>A and 1735C>T in this case not only expands the known mutation spectrum associated with mtDDS but also emphasizes the necessity for comprehensive genetic testing in atypical presentations, as similar cases have highlighted the importance of early genetic diagnosis to inform management strategies (13,14).
Moreover, the successful intervention of liver transplantation in this patient underscores the potential for surgical options in improving outcomes for individuals with POLG1-related mtDDS, particularly when liver dysfunction is prominent (15,16). The interplay between genetic mutations and clinical phenotype variation necessitates a personalized approach to management, as seen in this case where the absence of neurological symptoms deviates from traditional presentations (17,18). Thus, this case not only contributes to the understanding of POLG1 mutations but also advocates for heightened awareness and thorough investigation of metabolic disorders in pediatric patients presenting with liver failure and hypoglycemia.
The diagnostic challenges presented by POLG1 mutations, particularly in cases of mtDDS, underscore the necessity for clinicians to adopt a nuanced approach when evaluating patients with atypical clinical manifestations. The current case, which highlights recurrent hypoglycemia and liver failure without neurological impairment, expands upon previously documented clinical phenotypes associated with POLG1 mutations, which often include significant neurological deficits such as those seen in Alpers syndrome (11). Given the extensive variability in clinical presentations linked to POLG1 mutations, it becomes imperative for healthcare providers to maintain a high index of suspicion for mtDDS in patients exhibiting unexplained liver dysfunction and metabolic disturbances, as early genetic testing can significantly inform treatment strategies (13,17). Furthermore, the integration of advanced genomic tools such as WES can facilitate the identification of novel mutations, thereby enriching the understanding of the mutation spectrum and enhancing diagnostic accuracy (19,20).
In addition to the diagnostic complexities, therapeutic options for managing POLG1-related mtDDS, particularly in cases of liver involvement, warrant discussion. The successful outcome of liver transplantation in this patient illustrates the potential benefits of surgical intervention in improving quality of life for individuals with severe liver dysfunction attributable to POLG1 mutations (15,21). Moreover, unlike the findings of Kadohisa et al., this patient achieved a favorable clinical outcome as a heterozygote (10). This case reinforces the importance of a personalized management approach that considers the specific genetic and clinical characteristics of each patient (22). While our patient demonstrated a favorable early post-transplant course, the follow-up period remains short. Longer-term monitoring is essential to evaluate the risk of neurological deterioration and other potential late complications associated with POLG mutations. As the literature continues to evolve, it is imperative for clinicians to remain informed about the diverse manifestations of POLG1-related disorders and the implications for both diagnosis and treatment, emphasizing the need for ongoing education and awareness within the medical community regarding mitochondrial diseases (6,22,23).
Conclusions
In conclusion, the presented case illustrates the complexities surrounding the diagnosis and management of mtDDS due to POLG1 mutations, particularly when clinical manifestations diverge from the classical neurological deficits typically associated with this condition. The unique presentation of recurrent hypoglycemia and liver failure without neurological impairment emphasizes the necessity for clinicians to maintain a high index of suspicion for mtDDS in pediatric patients exhibiting unexplained liver dysfunction. This case underscores the importance of comprehensive genetic testing, including WES, to identify novel mutations such as 695G>A and 1735C>T, thereby enhancing diagnostic accuracy and informing treatment decisions. The successful outcome following liver transplantation not only highlights the potential for surgical intervention to significantly improve the quality of life in these patients but also advocates for a personalized approach to management, taking into account the specific genetic and clinical characteristics of each case. While our patient demonstrated a favorable early post-transplant course, the follow-up period remains short. Longer-term monitoring is essential to evaluate the risk of neurological deterioration and other potential late complications associated with POLG mutations.
After our detailed explanations, the patient’s legal guardian obtained a full understanding of the patient’s condition, acknowledged liver transplantation as an appropriate therapeutic modality, and proactively completed all preparations for the LDLT surgery. The guardian expressed satisfaction with the patient’s postoperative recovery and has been actively cooperating with the follow-up arrangements.
Reflecting on the lessons learned from this case, it becomes evident that early recognition and intervention are paramount in optimizing outcomes for children with mtDDS. While this report contributes to the expanding knowledge of POLG1-related disorders, it also acknowledges the limitations inherent in single-case studies, including the specificity of findings and the need for longitudinal follow-up to better understand the long-term implications of such mutations. Consequently, this case serves as a clarion call for heightened awareness among healthcare providers about the diverse manifestations of mitochondrial diseases, fostering an environment conducive to early diagnosis, timely intervention, and improved patient outcomes.
Acknowledgments
First and foremost, the authors would like to express sincere gratitude to the head of the General Surgery Department and Ethic Committee at Beijing Friendship Hospital for approving the submission of this retrospective case report. We also extend our thanks to the medical team involved in the diagnosis and treatment of the pediatric patient, whose professional efforts laid the foundation for this case analysis. Additionally, we appreciate the support from the Laboratory and Imaging Department for providing essential clinical data. Finally, we would like to thank the patient’s family for their trust and consent to share the relevant medical information for academic exchange.
Footnote
Reporting Checklist: The authors have completed the CARE reporting checklist. Available at https://tp.amegroups.com/article/view/10.21037/tp-2025-1-883/rc
Peer Review File: Available at https://tp.amegroups.com/article/view/10.21037/tp-2025-1-883/prf
Funding: None.
Conflicts of Interest: All authors have completed the ICMJE uniform disclosure form (available at https://tp.amegroups.com/article/view/10.21037/tp-2025-1-883/coif). The authors have no conflicts of interest to declare.
Ethical Statement: The authors are accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved. All procedures performed in this study were in accordance with the ethical standards of the institutional and/or national research committee(s) and with the Declaration of Helsinki and its subsequent amendments. Written informed consent was obtained from the patient’s legal guardian for the publication of this case report and accompanying images. A copy of the written consent is available for review by the editorial office of this journal.
Open Access Statement: This is an Open Access article distributed in accordance with the Creative Commons Attribution-NonCommercial-NoDerivs 4.0 International License (CC BY-NC-ND 4.0), which permits the non-commercial replication and distribution of the article with the strict proviso that no changes or edits are made and the original work is properly cited (including links to both the formal publication through the relevant DOI and the license). See: https://creativecommons.org/licenses/by-nc-nd/4.0/.
References
- Basel D. Mitochondrial DNA Depletion Syndromes. Clin Perinatol 2020;47:123-41. [Crossref] [PubMed]
- Huang CC, Hsu CH. Mitochondrial disease and mitochondrial DNA depletion syndromes. Acta Neurol Taiwan 2009;18:287-95.
- Nogueira C, Almeida LS, Nesti C, et al. Syndromes associated with mitochon-drial DNA depletion. Ital J Pediatr 2014;40:34. [Crossref] [PubMed]
- Filiano JJ, Goldenthal MJ, Mamourian AC, et al. Mitochondrial DNA depletion in Leigh syndrome. Pediatr Neurol 2002;26:239-42. [Crossref] [PubMed]
- Rahman S, Poulton J. Diagnosis of mitochondrial DNA depletion syndromes. Arch Dis Child 2009;94:3-5. [Crossref] [PubMed]
- Burusnukul P, de los Reyes EC. Phenotypic variations in 3 children with POLG1 mutations. J Child Neurol 2009;24:482-6. [Crossref] [PubMed]
- Bijarnia-Mahay S, Mohan N, Goyal D, et al. Mitochondrial DNA depletion syn-drome causing liver failure. Indian Pediatr 2014;51:666-8. [Crossref] [PubMed]
- Mazziotta MR, Ricci E, Bertini E, et al. Fatal infantile liver failure associated with mitochondrial DNA depletion. J Pediatr 1992;121:896-901. [Crossref] [PubMed]
- Bortot B, Barbi E, Biffi S, et al. Two novel POLG mutations causing hepatic mi-tochondrial DNA depletion with recurrent hypoketotic hypoglycaemia and fatal liver dysfunction. Dig Liver Dis 2009;41:494-9. [Crossref] [PubMed]
- Kadohisa M, Okamoto T, Yamamoto M, et al. Living donor liver transplantation for myocerebrohepatopathy spectrum due to POLG mutations. Pediatr Transplant 2024;28:e14659. [Crossref] [PubMed]
- Bulst S, Abicht A, Holinski-Feder E, et al. In vitro supplementation with dAMP/dGMP leads to partial restoration of mtDNA levels in mitochondrial de-pletion syndromes. Hum Mol Genet 2009;18:1590-9. [Crossref] [PubMed]
- Milone M, Massie R. Polymerase gamma 1 mutations: clinical correlations. Neurologist 2010;16:84-91. [Crossref] [PubMed]
- Lamperti C, Zeviani M. Encephalomyopathies caused by abnormal nucle-ar-mitochondrial intergenomic cross-talk. Acta Myol 2009;28:2-11.
- Poulton J, Hirano M, Spinazzola A, et al. Collated mutations in mitochondrial DNA (mtDNA) depletion syndrome (excluding the mitochondrial gamma poly-merase, POLG1). Biochim Biophys Acta 2009;1792:1109-12. [Crossref] [PubMed]
- Stewart JD, Tennant S, Powell H, et al. Novel POLG1 mutations associated with neuromuscular and liver phenotypes in adults and children. J Med Genet 2009;46:209-14. [Crossref] [PubMed]
- Spinazzola A, Zeviani M. Disorders of nuclear-mitochondrial intergenomic sig-naling. Gene 2005;354:162-8. [Crossref] [PubMed]
- Kim J, Kang E, Kim Y, et al. MPV17 mutations in patients with hepatocerebral mitochondrial DNA depletion syndrome. Mol Genet Metab Rep 2016;8:74-6. [Crossref] [PubMed]
- Ashley N, O'Rourke A, Smith C, et al. Depletion of mitochondrial DNA in fibro-blast cultures from patients with POLG1 mutations is a consequence of catalytic mutations. Hum Mol Genet 2008;17:2496-506. [Crossref] [PubMed]
- Wang Y, Dantas TJ, Lalor P, et al. Promoter hijack reveals pericentrin functions in mitosis and the DNA damage response. Cell Cycle 2013;12:635-46. [Crossref] [PubMed]
- Finsterer J. Clinico-Genetic Spectrum of POLG1 Mutation Carriers from India. J Mol Neurosci 2022;72:45-7. [Crossref] [PubMed]
- Ylönen S, Ylikotila P, Siitonen A, et al. Variations of mitochondrial DNA poly-merase γ in patients with Parkinson's disease. J Neurol 2013;260:3144-9. [Crossref] [PubMed]
- Kollberg G, Moslemi AR, Darin N, et al. POLG1 mutations associated with pro-gressive encephalopathy in childhood. J Neuropathol Exp Neurol 2006;65:758-68. [Crossref] [PubMed]
- Deschauer M, Tennant S, Rokicka A, et al. MELAS associated with mutations in the POLG1 gene. Neurology 2007;68:1741-2. [Crossref] [PubMed]