Global research landscape in pharmacotherapy for autism spectrum disorder core symptoms: a bibliometric study
Highlight box
Key findings
• Analysis of 109 publications [1993–2025] reveals a rising trend in autism spectrum disorder (ASD) pharmacotherapy research, centered on four clusters: repetitive behaviors, social behavior/neuropeptides, novel mechanisms, and adjunctive treatments.
• Research is shifting from traditional antipsychotics toward targeted neurobiological interventions and combination strategies.
What is known and what is new?
• Pharmacotherapy for ASD core symptoms remains challenging due to heterogeneity, with evidence often scattered across diverse drug classes and clinical trials.
• This study provides the first systematic bibliometric map of the field, quantifying the evolution from symptom-based management to mechanism-driven research and identifying emerging global trends.
What is the implication, and what should change now?
• There is a pressing need for more targeted and safe pharmacological options. Future efforts should prioritize multidisciplinary collaboration and focus on novel neurobiological targets to improve clinical outcomes.
Introduction
Autism spectrum disorder (ASD) is a complex neurodevelopmental disorder characterized by core symptoms such as social communication deficits, repetitive behaviors or restricted interests (1). ASD affects approximately over 28 million of the global population (2), with prevalence showing a significant upward trend over the past few decades (2,3). ASD not only severely impacts individual quality of life but also imposes substantial economic and psychological burdens on families and society (4). Consequently, identifying effective treatment methods, particularly pharmacotherapy targeting core symptoms, has become a focal point in current medical research.
The core symptoms of ASD form the foundation for diagnosis (5,6). Social communication deficits manifest as difficulties in social interaction, non-verbal communication impairments, and challenges in establishing and maintaining interpersonal relationships (1). Repetitive behaviors or restricted interests include repetitive motor patterns, insistence on routines or ritualized behaviors, and excessive focus on specific objects or topics with limited interest scope (1). The severity and presentation of these core symptoms vary significantly among individuals, posing substantial challenges for treatment planning (7).
Currently, ASD treatment primarily relies on non-pharmacological approaches. Crucially, early behavioral intervention is the only therapy currently shown to impact the core symptoms of ASD (8). Other supportive measures, which focus on broader functional improvement, include educational support (9) and rehabilitation training (9,10). However, as understanding of the neurobiological mechanisms underlying ASD deepens, the potential of pharmacotherapy to improve core symptoms is gaining increased attention. Existing pharmacotherapy mainly targets comorbid symptoms of ASD, such as attention-deficit/hyperactivity disorder (ADHD), anxiety, and depression (11,12). Treatments specifically addressing core symptoms remain largely in the exploratory phase. Emerging research suggests that the core symptoms of ASD are linked to complex neurobiological disruptions, including imbalances in excitatory (glutamate) and inhibitory [gamma-aminobutyric acid (GABA)] neurotransmission, as well as dysregulation of the serotonin and dopamine systems (13-15). Beyond simple neurotransmitter levels, abnormalities in synaptic plasticity, particularly involving SHANK proteins and neuropeptide signaling (e.g., oxytocin and vasopressin), are increasingly recognized as critical biological drivers of social and repetitive behavior deficits, offering precise targets for novel pharmacotherapeutic interventions.
Compared to traditional systematic reviews and meta-analyses, bibliometric analysis encompasses a broader range of research types, including clinical trials, case reports, and reviews, offering a more comprehensive overview of the field (16,17). Moreover, research on pharmacotherapy for ASD’s core symptoms is still in relatively early stages, with significant heterogeneity in research methodologies and evaluation standards, making direct meta-analysis challenging due to difficulties in data aggregation. Bibliometric methods do not rely on specific research outcome data but instead analyze external characteristics and citation relationships of literature to reveal knowledge structures, making them more suitable for assessing the developmental trajectory of this emerging research field.
In recent years, with the continuous development and refinement of medical research methodologies, an increasing number of scholars have focused on research into pharmacotherapy for ASD. From early case reports to large-scale randomized controlled trials, and from evaluations of single drug efficacy to explorations of combination therapy regimens, related research has shown rapid growth. However, due to the complexity of research designs, lack of uniform evaluation standards, and ethical considerations, findings in this field are dispersed across various journals and databases, lacking systematic integration and analysis.
This study employs bibliometric methods to systematically analyze the development and current status of research on pharmacotherapy for ASD’s core symptoms. By retrieving and analyzing relevant literature, we aim to explore the characteristics of this research field from multiple dimensions: (I) analyzing the temporal distribution and trend changes in publication quantity to understand the evolution of research activity; (II) identifying major research institutions, authors, and journals to explore academic collaboration networks and knowledge dissemination pathways; (III) revealing research hotspots and frontier directions through keyword co-occurrence analysis and thematic clustering; (IV) identifying influential literature and theoretical foundations based on citation analysis. The goal is to provide a panoramic understanding of pharmacotherapy research for ASD, identify research gaps and development opportunities, and offer references for future research directions. Additionally, the results of this study will assist clinicians, researchers, and policymakers in better understanding the development status of this field, promoting rational resource allocation and coordinated research development. We present this article in accordance with the BIBLIO reporting checklist (available at https://tp.amegroups.com/article/view/10.21037/tp-2025-1-936/rc).
Methods
Bibliometric methods have been widely applied in the field of ASD research. R (biblioshiny) was utilized for co-citation, co-word, co-authorship, and source citation analysis in this research. This paper aims to employ bibliometric research methods to describe literature sources, authors, documents, and keywords using various bibliometric techniques. To illustrate global collaboration in the field of pharmacotherapy for core symptoms of ASD, this study employs a network analysis framework to describe country collaboration, production, and citation patterns. The entire research process is conducted using the R package biblioshiny for statistical processing.
Data sources
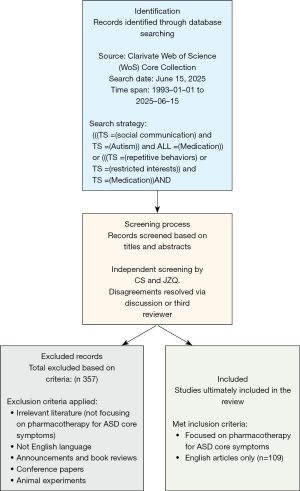
The comprehensive and reliable Clarivate Web of Science (WoS) Core Collection was utilized to retrieve all published research outcomes. The search strategy employed was: (((TS=(social communication*)) AND TS=(Autism*)) AND ALL=(Medication*) OR ((TS=(repetitive behaviors*) OR (TS=(restricted interests*)) AND TS=(Medication*)) AND ALL=(drug*)) with a time span of 19930101-202510615. This search was conducted on June 15, 2025. To enhance the accuracy of the investigation, C.S. and Z.J. independently screened the retrieved papers based on titles and abstracts under the supervision of C.S. to determine compliance with predetermined inclusion and exclusion criteria. Disagreements were resolved through internal discussion; if persistent, a third reviewer made the final decision. Inclusion criteria were: (I) articles must focus on pharmacotherapy for ASD core symptoms; (II) only English articles were eligible. Exclusion criteria were: (I) records related to announcements and book reviews were excluded; (II) conference papers were excluded; (III) animal experiments were excluded; (IV) review articles were included. A total of 466 records were retrieved, and after excluding irrelevant literature, books, and records in other languages, 109 references were ultimately included. The final inclusion of 109 articles reflects the highly specific focus of our study on medications targeting ASD core symptoms rather than more common comorbid symptoms, highlighting that this specific area of pharmacotherapy remains a specialized and emerging niche within the broader ASD research landscape (Figure 1).
Statistical analysis
In the analysis process, bibliometric techniques were employed to extract networks at different levels. Co-citation analysis, first introduced by American information scientist Henry Small, is a technique used to measure the degree of connection between documents. For example, if Document A cites both Document C and Document D, the number of documents that simultaneously cite both C and D is referred to as the co-citation strength. In this paper, the co-citation strength is 1, as only Document A cites both C and D. Co-word analysis typically originates from “author keywords”. If unavailable, notable terms can be extracted from the “article title” and “abstract”. Additionally, terms of interest can be extracted from the “article title”, “abstract”, and “full text” for analysis. Co-authorship networks are a common form of scientific collaboration networks, established through the collaborative relationships of paper authors. If two authors co-write one or more papers, a continuous relationship is formed between them. This network provides strong evidence for studying the structure of academic relationship networks.
Results
Trends in the literature
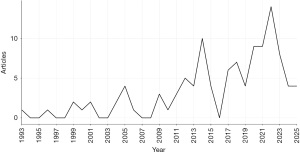
Based on the scientific outputs related to pharmacotherapy for ASD core symptoms from 1993 to 2025 (Figure 2) and bibliometric analysis (Table 1), the research has experienced rapid growth at an annual rate of 4.43%. As shown in Table 1, over the past decade, a total of 526 authors worldwide have contributed to 109 papers. Additionally, the values for international co-authorships and co-authors per document are notably high. This indicates a preference for collaboration among authors in this research field, with a significant proportion of international cooperation, aligning with the global trend in the field of ASD pharmacotherapy.
Table 1
| Main information | Results |
|---|---|
| Timespan, year | 1993–2025 |
| Sources (journals, books, etc.) | 91 |
| Documents | 109 |
| Annual growth rate, % | 4.43 |
| Document average age, years | 8.8 |
| Average citations per doc | 42.1 |
| References | 5,639 |
| Document contents | |
| Keywords plus (ID) | 407 |
| Author’s keywords (DE) | 264 |
| Authors | 526 |
| Authors of single-authored docs | 8 |
| Authors collaboration | |
| Single-authored docs | 8 |
| Co-authors per doc | 5.8 |
| International co-authorships, % | 22.02 |
| Document types | |
| Article | 58 |
| Review | 50 |
| Review; book chapter | 1 |
Data are presented as number unless otherwise indicated.
Distribution of literature sources
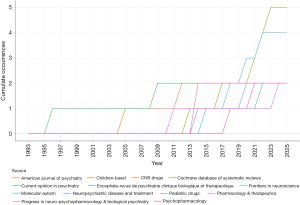
Regarding the publication dynamics of leading journals (Figure 3), most journals have shown a steady increase in cumulative publication volume. The French journal Encephale-Revue De Psychiatrie Clinique Biologique Et Therapeutique was a core journal in the field of pharmacotherapy for ASD core symptoms from 1993 to 2013. Since 2011, with the development of more medications targeting ASD core symptoms, journals focusing on clinical translational research, such as Molecular Autism, have seen an increase in publication volume. Additionally, journals primarily dedicated to reviews, such as the Cochrane Database of Systematic Reviews, have also experienced growth in publication volume.
In the source co-citation network, three clusters were identified (Figure 4), with Clusters 1, 2, and 3 being the primary sources of clustering. Clusters 1, 2, and 3 are interwoven and mixed together. Moreover, Cluster 3 demonstrates advantages in terms of betweenness centrality and the number of publications. The results indicate that more than half of the top ten high-output journals are located in Cluster 1, with Psychopharmacology and Progress in Neuro-Psychopharmacology & Biological Psychiatry leading the group.

Author-level analysis
Regarding the author-level analysis, the results first highlight the most relevant authors and several bibliometric indicators (Table 2), followed by the temporal evolution of authors’ publication outputs (Figure 5). The H-index presented here is derived specifically from the 109 included publications within this bibliometric analysis, representing the local impact of authors within this specialized research niche.
Table 2
| Ranking | Author | Number of paper | H-index | Total citation, n |
|---|---|---|---|---|
| 1 | Mcdougle CJ | 9 | 9 | 776 |
| 2 | Anagnostou E | 6 | 6 | 331 |
| 3 | Arango C | 5 | 5 | 214 |
| 4 | Vitiello B | 5 | 5 | 536 |
| 5 | Erickson CA | 4 | 4 | 130 |
| 6 | Hollander E | 5 | 4 | 203 |
| 7 | Mccracken JT | 4 | 4 | 467 |
| 8 | Scahill L | 4 | 4 | 499 |
| 9 | Alsayouf HA | 3 | 3 | 40 |
| 10 | Aman MG | 4 | 3 | 469 |

From Table 2, it is evident that the number of papers published by authors generally aligns with their ranking order. When comparing the H-index and total citation counts, the data show some fluctuations. Arango C ranks third in the top ten for H-index, with a total citation count of 214. Conversely, Vitiello B has a high citation count [536] and a higher H-index [5] with a publication count of 5, indicating that this author not only excels in quantity but also in the quality of their work.
In Figure 5, it is observed that Mcdougle CJ has been conducting research in the field of pharmacotherapy for ASD core symptoms since 2000, engaging in in-depth studies over a span of 20 years and producing high-quality research. Arango C began research in this area in 2020 and has recently produced a substantial amount of high-quality literature.
Analysis of international cooperation
By investigating national collaboration networks, both domestic and international cooperation, as well as publication output rates and citation networks, the analysis identified the countries with the highest article output based on the authors’ affiliations.
Through the analysis of inter-country collaboration networks, Figure 6 illustrates the collaboration network among the top 10 countries. The size of the nodes reflects the volume of collaboration, with larger nodes indicating more collaboration, while the thickness of the links represents the frequency of collaboration, with thicker links indicating higher collaboration frequency. Different colors represent clusters of major collaborating countries. The first tier of collaborating countries, led by the US, exhibits a high total citation count, with Australia and the US accounting for more than half of the total citations, as shown in Table 3. In terms of total citations and average annual citations, it was found that although Brazil has a relatively low total citation count, its average annual citation rate places it among the top three.

Table 3
| Country | Total citation, n | Average article citations |
|---|---|---|
| USA | 2,952 | 60.20 |
| Australia | 440 | 88.00 |
| Italy | 239 | 23.90 |
| U Arab Emirates | 178 | 44.50 |
| Canada | 169 | 42.20 |
| China | 150 | 25.00 |
| Germany | 108 | 36.00 |
| Iran | 67 | 11.20 |
| India | 61 | 30.50 |
| Brazil | 58 | 58.00 |
The analysis of corresponding authors’ countries revealed that research in the relevant field is predominantly conducted through international collaboration.
Keyword analysis
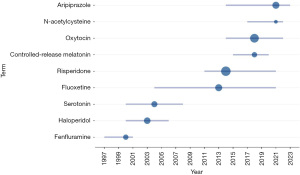
In the context of pharmacotherapy for ASD core symptoms, the most frequently used terms include risperidone, oxytocin, aripiprazole, fluoxetine, haloperidol, serotonin, controlled-release melatonin, fenfluramine, N-acetylcysteine (Figure 7). Trend analysis (Figure 8) indicates that research on topics such as fenfluramine, haloperidol, and serotonin has gradually decreased since 1997. In contrast, research interest in fluoxetine has remained steady since 2004. Additionally, oxytocin, controlled-release melatonin, aripiprazole, and N-acetylcysteine have emerged as popular drugs for treating ASD core symptoms in recent years.
Based on the co-word analysis (Figure 9), four clusters were identified, reflecting four major research areas. Since the co-occurrence network is derived from authors’ keywords, the analysis provides insights into the research domains that scholars are focusing on.

In Cluster 1, researchers primarily focus on the field of repetitive behavior. The results indicate that risperidone and fluoxetine are prominent drugs in the treatment of repetitive behaviors. Both medications indirectly mitigate repetitive behaviors in ASD by modulating the 5-hydroxytryptamine (5-HT)-related network. The difference lies in the fact that risperidone acts quickly and is more effective for motor-impulsive stereotypies, but it carries a significant metabolic/EPS burden. Fluoxetine is more targeted towards anxiety-compulsive stereotypies, with relatively mild side effects but smaller and delayed efficacy. Additionally, recent books, systematic reviews, and literature suggest that controlled-release melatonin is emerging as a new hotspot in the treatment of repetitive behaviors in autism.
Cluster 2 research focuses more on social behavior, including vasopressin and oxytocin. Vasopressin and oxytocin share “prosocial neuropeptide” micro-sites, similar brain region distribution and signaling mechanisms, a unified nasal delivery method, and good safety profiles in social function interventions for ASD.
Cluster 3 exhibits significant heterogeneity. While memantine is investigated for its potential to target core symptoms, aripiprazole and risperidone are primarily utilized for irritability in ASD—their only Food and Drug Administration (FDA)-approved indications in this population. Similarly, atomoxetine is primarily indicated for comorbid ADHD symptoms. Furthermore, the perceived efficacy of melatonin is more accurately attributed to its impact on sleep disorders common in ASD rather than core social deficits.
Cluster 4’s research themes focus more on adjunctive medications used in conjunction with relevant drug treatments.
The thematic terms were categorized into four types: basic, emerging or declining, niche, and motor themes (Figure 10). The representative labels for these categories are as follows: repetitive behavior and social behavior are core basic themes. Clozapine is identified as a motor theme (well-developed core research areas), while serotonin reuptake inhibitors represent a relatively niche theme (specialized topics with limited external connection). N-acetylcysteine, guanfacine, and memantine are identified as emerging themes with significant changes.
Discussion
Bibliometric analysis has revealed a significant trend of international collaboration in pharmacotherapy research for ASD core symptoms. Countries such as the USA, Australia, and Italy dominate this field. The USA leads in both total citation count and average annual citations, underscoring its global academic influence. The collaboration between Australia and the USA is particularly strong, with their combined citation count accounting for more than half of the total citations, indicating extensive and impactful cooperation in ASD drug treatment research. This international collaboration model facilitates knowledge sharing and dissemination, accelerates research progress, and advances the exploration of treatment strategies worldwide (18). However, despite the crucial role of international collaboration, challenges remain. Unequal distribution of research resources among countries is evident, with developed nations having significant advantages in funding, technology, and expertise, while developing countries face resource constraints. Cultural differences and language barriers may also affect the depth and breadth of international cooperation (19). Future efforts should focus on strengthening global collaboration mechanisms, promoting equitable resource allocation (e.g., through multilateral projects), and enhancing research capabilities in developing countries to achieve broader knowledge sharing and technology transfer (20,21).
Academic journals play a vital role in the dissemination of knowledge and academic exchange in pharmacotherapy research for ASD. The French journal Encephale-Revue De Psychiatrie Clinique Biologique Et Therapeutique was a core journal from 1993 to 2013, with a steady increase in publication volume, reflecting its leading position in early research. As research deepened and new drugs were developed, the journal landscape gradually changed. Since 2011, journals like Molecular Autism, focusing on clinical translational research, have seen a significant increase in publication volume, indicating a shift in research focus from basic theory to clinical application. Review journals such as the Cochrane Database of Systematic Reviews have also experienced growth, reflecting the increasing demand for systematic summaries and evaluations of existing research findings. These journals provide high-quality comprehensive analyses, offering important references for researchers and clinicians, and promoting the standardized development of the field.
The evolution of research themes and keywords in ASD drug treatment reflects the continuous exploration and optimization of treatment strategies for core symptoms by the academic community. Early research primarily focused on traditional drugs such as haloperidol and fenfluramine. These drugs alleviate autism symptoms, particularly repetitive behaviors and anxiety-related comorbid symptoms, by modulating dopamine and serotonin systems (22-24). Haloperidol, as a typical antipsychotic, shows efficacy in controlling aggression and self-injurious behavior, but its significant side effects, including extrapyramidal symptoms (EPS) and metabolic syndrome, limit its long-term use (25). Fenfluramine, which improves behavioral issues by increasing serotonin levels, has been banned in many countries due to potential cardiovascular risks (26,27). In recent years, research hotspots have gradually shifted to novel drugs and treatment methods. Oxytocin has gained widespread attention for its potential to improve social behavior. As a neuropeptide, oxytocin enhances social interaction and emotional recognition abilities, with clinical trials indicating some improvement in social deficits for certain autism patients (22,28,29). However, its efficacy varies among individuals, and further research is needed to determine long-term safety and optimal dosage. Controlled-release melatonin has become a research hotspot due to its significant effects on improving sleep disorders (30,31). Autism patients often experience sleep issues, and controlled-release melatonin effectively improves sleep quality by regulating circadian rhythms, thereby indirectly alleviating other core symptoms. Its advantage lies in having fewer side effects, making it suitable for long-term use. Aripiprazole, a partial dopamine agonist, shows good efficacy in controlling irritability and aggressive behavior, with relatively mild side effects (32). N-acetylcysteine, through antioxidant and anti-inflammatory effects, shows potential in improving repetitive behaviors and emotional regulation, but its efficacy requires validation through large-scale clinical trials (33,34). Future research should focus on individualized treatment and combination drug strategies. For example, combining oxytocin and controlled-release melatonin may enhance overall efficacy through multi-target action (35). Additionally, the development of new drugs based on neurobiological mechanisms will be an important direction, such as drugs targeting the glutamate system, GABA system, and immune modulation. Recent clinical trials have explored arbaclofen (36) (targeting the GABA system), sulforaphane (37), and bumetanide (38) for their potential to impact core symptoms, though findings remain mixed. Research should also emphasize long-term follow-up to assess drug safety and tolerability, providing more reliable evidence for clinical practice.
The development trends in ASD drug treatment can be analyzed from three aspects: drug types, mechanisms of action, and research methods (39). Early research mainly focused on antipsychotics and antidepressants, which alleviate symptoms by modulating dopamine and serotonin systems. With a deeper understanding of the neurobiological mechanisms of ASD, research focus has gradually shifted to more targeted novel drugs. For example, neuropeptide drugs (such as oxytocin) and antioxidants (such as N-acetylcysteine) have become research focal points due to their unique mechanisms of action and better safety profiles. Research is gradually evolving from single-target to multi-target combination therapy. The combined use of glutamate inhibitors (such as memantine) and partial dopamine agonists (such as aripiprazole) may enhance efficacy through synergistic effects. Precision medicine strategies based on genetic and molecular levels are also becoming research hotspots. Drug development targeting specific gene mutations (such as SHANK3 or FMR1) offers new possibilities for individualized treatment (40,41). Future research should focus on individualized treatment, combination drug strategies, novel drug development, long-term follow-up studies, and interdisciplinary collaboration. Develop personalized treatment plans based on patients’ genotypes, phenotypes, and biomarkers. Explore the combined use of multi-target drugs to enhance efficacy and reduce side effects. Drug development targeting the glutamate system, GABA system, and immune modulation provides more options for ASD treatment. Assess the long-term efficacy and safety of drugs to provide more reliable evidence for clinical practice. Furthermore, given that the target population primarily involves children and adolescents, future research must prioritize ethical transparency and long-term safety monitoring. Regulatory frameworks should be standardized globally to ensure that emerging pharmacotherapies undergo rigorous pediatric-specific clinical trials before widespread adoption. Integrate research findings from neuroscience, genetics, and immunology to drive innovative development in ASD drug treatment.
Despite significant progress in ASD drug treatment research, numerous challenges remain. The core symptoms of ASD exhibit high heterogeneity, with significant differences in symptom presentation and severity among patients, complicating the formulation and implementation of treatment plans. Existing drugs primarily target comorbid symptoms, with limited efficacy on core symptoms, and some drugs have significant side effects, restricting their clinical application. The heterogeneity of research methods and lack of unified evaluation standards also affect the reliability and comparability of research results. Differences in evaluation tools and indicators used in different studies lead to difficulties in data aggregation and analysis. Future efforts should focus on standardizing research methods and establishing unified evaluation standards to enhance the comparability and reproducibility of research results. Special attention is needed to protect the rights of child and adolescent participants, and the long-term safety and efficacy of medications require further evaluation. Future research should emphasize the long-term efficacy and safety of drugs through long-term follow-up studies. In summary, while ASD drug treatment research has made significant progress, it still faces numerous challenges. Future efforts should focus on strengthening international collaboration, optimizing research methods and evaluation standards, and promoting the development and application of novel drugs to achieve more effective individualized treatment strategies and improve the quality of life for individuals with ASD.
There are several key strengths in this study. It utilized the authoritative WoS Core Collection and employed a rigorous manual screening process by independent reviewers to ensure the high relevance and accuracy of the included literature. Additionally, the use of biblioshiny allowed for a comprehensive visualization of the field’s evolution over three decades. However, limitations must be acknowledged. Firstly, the analysis relied solely on the WoS Core Collection. While WoS is a premier database for bibliometric analysis, its exclusion of other databases like Scopus or PubMed might lead to coverage bias and potentially omit relevant studies. This restriction may lead to an underestimation of scientific output from non-English-speaking nations, particularly in regions where ASD research and clinical trials are rapidly expanding in local languages. Relying solely on a single database could miss unique records indexed elsewhere. Furthermore, the use of the term ’social communication' in our search strategy may have limited the retrieval of older pharmacotherapeutic literature published before this became a standardized diagnostic term. Older studies focusing on separate social and language domains might be underrepresented. Citation metrics inherently favor older publications, potentially underrepresenting the impact of recent studies. Finally, this analysis focuses on research trends based on metadata rather than evaluating the clinical quality of the evidence. It is important to note that bibliometric prominence—such as high citation counts or keyword frequency—reflects academic attention and research trends rather than the clinical safety, methodological quality, or therapeutic effectiveness of the medications discussed. Consequently, the findings of this study should be interpreted as a map of the research landscape rather than a direct guide for clinical decision-making or an endorsement of specific pharmacological interventions.
Conclusions
In conclusion, while significant strides have been made in understanding the potential for pharmacotherapy in ASD, substantial challenges persist. Bibliometric analysis confirms the field’s dynamism and international engagement but also reveals the need for greater standardization, deeper investment in novel mechanisms. Addressing the heterogeneity of ASD and developing treatments with demonstrable efficacy and improved safety profiles should be central to future research priorities. Continued global collaboration and multidisciplinary efforts are vital to translate current scientific insights into tangible improvements in the lives of individuals affected by ASD.
Acknowledgments
None.
Footnote
Reporting Checklist: The authors have completed the BIBLIO reporting checklist. Available at https://tp.amegroups.com/article/view/10.21037/tp-2025-1-936/rc
Peer Review File: Available at https://tp.amegroups.com/article/view/10.21037/tp-2025-1-936/prf
Funding: None.
Conflicts of Interest: All authors have completed the ICMJE uniform disclosure form (available at https://tp.amegroups.com/article/view/10.21037/tp-2025-1-936/coif). The authors have no conflicts of interest to declare.
Ethical Statement: The authors are accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Open Access Statement: This is an Open Access article distributed in accordance with the Creative Commons Attribution-NonCommercial-NoDerivs 4.0 International License (CC BY-NC-ND 4.0), which permits the non-commercial replication and distribution of the article with the strict proviso that no changes or edits are made and the original work is properly cited (including links to both the formal publication through the relevant DOI and the license). See: https://creativecommons.org/licenses/by-nc-nd/4.0/.
References
- American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders: DSM-5. 5th ed. Washington, DC: American Psychiatric Association; 2013.
- Solmi M, Song M, Yon DK, et al. Incidence, prevalence, and global burden of autism spectrum disorder from 1990 to 2019 across 204 countries. Mol Psychiatry 2022;27:4172-80. [Crossref] [PubMed]
- The burden of mental disorders across the states of India: the Global Burden of Disease Study 1990-2017. Lancet Psychiatry 2020;7:148-61. [Crossref] [PubMed]
- The global epidemiology and health burden of the autism spectrum: findings from the Global Burden of Disease Study 2021. Lancet Psychiatry 2025;12:111-21. [Crossref] [PubMed]
- Maenner MJ, Rice CE, Arneson CL, et al. Potential impact of DSM-5 criteria on autism spectrum disorder prevalence estimates. JAMA Psychiatry 2014;71:292-300. [Crossref] [PubMed]
- DSM-5: diagnosis of mental disorders. Lancet 2010;376:390.
- Sharma SR, Gonda X, Tarazi FI. Autism Spectrum Disorder: Classification, diagnosis and therapy. Pharmacol Ther 2018;190:91-104. [Crossref] [PubMed]
- Wood JJ, Kendall PC, Wood KS, et al. Cognitive Behavioral Treatments for Anxiety in Children With Autism Spectrum Disorder: A Randomized Clinical Trial. JAMA Psychiatry 2020;77:474-83. [Crossref] [PubMed]
- Morsa M, De Andrade V, Alcaraz C, et al. A scoping review of education and training interventions in Autism Spectrum Disorder. Patient Educ Couns 2022;105:2850-9. [Crossref] [PubMed]
- Özkan E, Belhan Çelik S, Yaran M, et al. Joint Attention-Based Occupational Therapy Intervention in Preschoolers With Autism Spectrum Disorder: A Randomized Controlled Trial. Am J Occup Ther 2023;77:7702205090. [Crossref] [PubMed]
- Ray LA, Meredith LR, Kiluk BD, et al. Combined Pharmacotherapy and Cognitive Behavioral Therapy for Adults With Alcohol or Substance Use Disorders: A Systematic Review and Meta-analysis. JAMA Netw Open 2020;3:e208279. [Crossref] [PubMed]
- Rodrigues R, Lai MC, Beswick A, et al. Practitioner Review: Pharmacological treatment of attention-deficit/hyperactivity disorder symptoms in children and youth with autism spectrum disorder: a systematic review and meta-analysis. J Child Psychol Psychiatry 2021;62:680-700. [Crossref] [PubMed]
- Man KK, Tong HH, Wong LY, et al. Exposure to selective serotonin reuptake inhibitors during pregnancy and risk of autism spectrum disorder in children: a systematic review and meta-analysis of observational studies. Neurosci Biobehav Rev 2015;49:82-9. [Crossref] [PubMed]
- Cabana-Domínguez J, Torrico B, Reif A, et al. Comprehensive exploration of the genetic contribution of the dopaminergic and serotonergic pathways to psychiatric disorders. Transl Psychiatry 2022;12:11. [Crossref] [PubMed]
- Mahdavi M, Kheirollahi M, Riahi R, et al. Meta-Analysis of the Association between GABA Receptor Polymorphisms and Autism Spectrum Disorder (ASD). J Mol Neurosci 2018;65:1-9. [Crossref] [PubMed]
- Mejia C, Wu M, Zhang Y, et al. Exploring Topics in Bibliometric Research Through Citation Networks and Semantic Analysis. Front Res Metr Anal 2021;6:742311. [Crossref] [PubMed]
- Glynatsi NE, Knight VA. A bibliometric study of research topics, collaboration, and centrality in the iterated prisoner's dilemma. Humanities and Social Sciences Communications 2021;8:45. [Crossref]
- Chen Y, Du X, Zhang X, et al. Research trends of inflammation in autism spectrum disorders: a bibliometric analysis. Front Immunol 2025;16:1534660. [Crossref] [PubMed]
- Masri AT, Nasir AK, Irshaid AG, et al. Autism services in low-resource areas. Neurosciences (Riyadh) 2023;28:116-22. [Crossref] [PubMed]
- Liu Q, Hsieh WY, Chen G. A systematic review and meta-analysis of parent-mediated intervention for children and adolescents with autism spectrum disorder in mainland China, Hong Kong, and Taiwan. Autism 2020;24:1960-79. [Crossref] [PubMed]
- Sandbank M, Bottema-Beutel K, Crowley LaPoint S, et al. Autism intervention meta-analysis of early childhood studies (Project AIM): updated systematic review and secondary analysis. BMJ 2023;383:e076733. [Crossref] [PubMed]
- Siafis S, Çıray O, Wu H, et al. Pharmacological and dietary-supplement treatments for autism spectrum disorder: a systematic review and network meta-analysis. Mol Autism 2022;13:10. [Crossref] [PubMed]
- Rajapakse T, Pringsheim T. Pharmacotherapeutics of Tourette syndrome and stereotypies in autism. Semin Pediatr Neurol 2010;17:254-60. [Crossref] [PubMed]
- Reddihough DS, Marraffa C, Mouti A, et al. Effect of Fluoxetine on Obsessive-Compulsive Behaviors in Children and Adolescents With Autism Spectrum Disorders: A Randomized Clinical Trial. JAMA 2019;322:1561-9. [Crossref] [PubMed]
- Beach SR, Gross AF, Hartney KE, et al. Intravenous haloperidol: A systematic review of side effects and recommendations for clinical use. Gen Hosp Psychiatry 2020;67:42-50. [Crossref] [PubMed]
- Schoonjans AS, Marchau F, Paelinck BP, et al. Cardiovascular safety of low-dose fenfluramine in Dravet syndrome: a review of its benefit-risk profile in a new patient population. Curr Med Res Opin 2017;33:1773-81. [Crossref] [PubMed]
- Seferian A, Chaumais MC, Savale L, et al. Drugs induced pulmonary arterial hypertension. Presse Med 2013;42:e303-10. [Crossref] [PubMed]
- Josselsohn A, Zhao Y, Espinoza D, et al. Oxytocin in neurodevelopmental disorders: Autism spectrum disorder and Prader-Willi syndrome. Pharmacol Ther 2024;264:108734. [Crossref] [PubMed]
- Sgritta M, Dooling SW, Buffington SA, et al. Mechanisms Underlying Microbial-Mediated Changes in Social Behavior in Mouse Models of Autism Spectrum Disorder. Neuron 2019;101:246-259.e6. [Crossref] [PubMed]
- Xiong M, Li F, Liu Z, et al. Efficacy of Melatonin for Insomnia in Children with Autism Spectrum Disorder: A Meta-analysis. Neuropediatrics 2023;54:167-73. [Crossref] [PubMed]
- Bruni O, Biggio G, Malorgio E, et al. Insomnia in children affected by autism spectrum disorder: The role of melatonin in treatment. Sleep Med 2024;119:511-7. [Crossref] [PubMed]
- Hirsch LE, Pringsheim T. Aripiprazole for autism spectrum disorders (ASD). Cochrane Database Syst Rev 2016;2016:CD009043. [Crossref] [PubMed]
- Lee TM, Lee KM, Lee CY, et al. Effectiveness of N-acetylcysteine in autism spectrum disorders: A meta-analysis of randomized controlled trials. Aust N Z J Psychiatry 2021;55:196-206. [Crossref] [PubMed]
- Wink LK, Adams R, Wang Z, et al. A randomized placebo-controlled pilot study of N-acetylcysteine in youth with autism spectrum disorder. Mol Autism 2016;7:26. [Crossref] [PubMed]
- Caddeo A, Trampetti R, Messina G, et al. A neuroendocrine therapeutic approach with the pineal hormone melatonin, cannabidiol and oxytocin (MCO regimen) in the treatment of the autism spectrum disorders. J Immun Allergy 2020;1:1-7.
- Huang Q, Velthuis H, Pereira AC, et al. Exploratory evidence for differences in GABAergic regulation of auditory processing in autism spectrum disorder. Transl Psychiatry 2023;13:320. [Crossref] [PubMed]
- Magner M, Thorová K, Župová V, et al. Sulforaphane Treatment in Children with Autism: A Prospective Randomized Double-Blind Study. Nutrients 2023;15:718. [Crossref] [PubMed]
- Sprengers JJ, van Andel DM, Zuithoff NPA, et al. Bumetanide for Core Symptoms of Autism Spectrum Disorder (BAMBI): A Single Center, Double-Blinded, Participant-Randomized, Placebo-Controlled, Phase-2 Superiority Trial. J Am Acad Child Adolesc Psychiatry 2021;60:865-76. [Crossref] [PubMed]
- Schubart JR, Camacho F, Leslie D. Psychotropic medication trends among children and adolescents with autism spectrum disorder in the Medicaid program. Autism 2014;18:631-7. [Crossref] [PubMed]
- Hagerman R, Au J, Hagerman P. FMR1 premutation and full mutation molecular mechanisms related to autism. J Neurodev Disord 2011;3:211-24. [Crossref] [PubMed]
- Cochoy DM, Kolevzon A, Kajiwara Y, et al. Phenotypic and functional analysis of SHANK3 stop mutations identified in individuals with ASD and/or ID. Mol Autism 2015;6:23. [Crossref] [PubMed]