Risk factors for complications after laparoscopic surgery in children with congenital choledochal cysts
Highlight box
Key findings
• This study identified cyst wall thickness >5 mm, chronic cholecystitis, Todani type I, elevated C-reactive protein (CRP), and elevated direct bilirubin (DB) as independent risk factors for postoperative complications following laparoscopic choledochal cyst excision in children.
• A nomogram incorporating these factors was developed and validated for individualized risk stratification.
What is known and what is new?
• Laparoscopic Roux-en-Y hepaticojejunostomy is the standard treatment for pediatric choledochal cysts. Postoperative complications remain a significant concern, and factors such as cyst wall thickness and Todani classification have been anecdotally linked to outcomes.
• This study provides a multivariable, quantitative risk model that integrates routinely available preoperative clinical and imaging variables. This is the first study to develop and internally validate a nomogram for preoperative stratification of complication risk in children undergoing laparoscopic surgery for congenital choledochal cysts.
What is the implication, and what should change now?
• Preoperative assessment should systematically include measurement of cyst wall thickness, precise Todani classification, and evaluation of inflammatory (CRP) and obstructive (DB) markers to improve surgical outcomes and quality of life in children with choledochal cysts.
• The predictive model requires prospective external validation in independent cohorts. Future research should aim to refine the model by incorporating intraoperative variables and exploring its impact on long-term clinical outcomes.
Introduction
Background
Congenital choledochal cyst (CCC), also known as congenital choledochal dilatation (CBD), is a rare congenital biliary malformation characterized by cystic or fusiform dilation of the bile ducts (1). This condition exhibits a distinct geographic distribution, being predominantly prevalent in Asian populations, with an estimated incidence of 1 in 1,000 live births. In contrast, the incidence in Western populations is significantly lower, ranging from 1 in 100,000 to 1 in 150,000 live births, and it demonstrates a female predominance (2,3). The most widely used classification system of CCC is the Todani classification, which divides choledochal cysts into five types. Among these, type I cysts, characterized by cystic or fusiform dilation of the extrahepatic bile ducts, account for approximately 80% of cases (4).
Rationale and knowledge gap
Pancreaticobiliary maljunction (PBM) and congenital biliary stricture constitute the primary etiological factors for choledochal cysts (5). Clinically, choledochal cysts typically present with a triad of symptoms: recurrent abdominal pain, painless jaundice, and abdominal masses (6). Complete cyst excision with biliary reconstruction remains the optimal curative intervention (7). While laparoscopic techniques have emerged as the primary minimally invasive approach for pediatric choledochal cyst management (8,9), these procedures demand advanced surgical expertise and meticulous preoperative planning. Operative challenges are compounded by limited visualization, anatomical variations in the hepatic ductal system, and cyst wall inflammation, particularly prevalent in children (10), which collectively elevate complication risks. Complications are stratified temporally: early (hemorrhage, biliary leakage, pancreatic fistula, ileus, surgical site infection) and late (hepaticojejunostomy stricture, cholangitis, adhesive bowel obstruction, reoperation) (11), which affects the life quality of patients with choledochal cyst.
Emerging evidence identifies neonatal presentation, PBM, Todani type, and inflammatory severity as key risk factors of post-excision complications (12,13). Nevertheless, validated predictive models remain underdeveloped.
Objective
This study aims to identify independent predictors of complications following CCC excision in pediatric patients and to develop a clinically applicable risk-stratification model. The model is intended to assist in preoperative risk assessment and to guide individualized management strategies, ultimately aiming to improve surgical outcomes and quality of life in this population. We present this article in accordance with the STROBE reporting checklist (available at https://tp.amegroups.com/article/view/10.21037/tp-2025-1-948/rc).
Methods
Study population
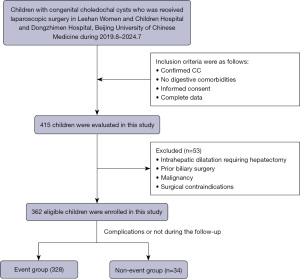
A total of 362 pediatric patients who underwent laparoscopic Roux-en-Y hepaticojejunostomy for choledochal cysts between August 2019 and July 2024 were retrospectively enrolled from two medical centers: Leshan Women and Children Hospital (200 children) and Dongzhimen Hospital, Beijing University of Chinese Medicine (162 children) (Figure 1). Study endpoints depended on the presence or absence of defined complications based on ≥6 months of postoperative follow-up.
Inclusion criteria comprised: (I) confirmed choledochal cyst diagnosis; (II) absence of concomitant digestive system comorbidities or malformations; (III) parental provision of written informed consent; (IV) availability of complete clinical, laboratory, and imaging data.
Exclusion criteria included: (I) intrahepatic biliary dilatation necessitating radical hepatic resection; (II) history of prior biliary surgery; (III) malignant transformation of the cyst or biliary tract; (IV) surgical contraindications; (V) death during follow-up.
The study was conducted in accordance with the Declaration of Helsinki and its subsequent amendments. The study was approved by the Ethics Committee of Leshan Women and Children Hospital (No. Lsfu250236) and informed consent was obtained from all patients’ legal guardians. Dongzhimen Hospital, Beijing University of Chinese Medicine was also informed and agreed the study.
Data collection
Demographic and clinical data were extracted by trained researchers using standardized questionnaires, physical examination records, and a thorough review of electronic medical records. The collected variables included:
Demographic and operative variables: sex, age at surgery, clinical symptoms at presentation, and operative time (minutes).
Imaging-based variables:
- Cyst diameter: the maximal transverse diameter measured on preoperative imaging (cm).
- Cyst wall thickness: the thickest portion of the cyst wall was measured on preoperative ultrasound, computed tomography (CT), or magnetic resonance imaging (MRI) (mm). For analytical purposes, a threshold of >5 mm was used to define cyst wall thickening. This cutoff was selected based on prior clinical observations and literature suggesting that wall thickness >4–5 mm is associated with localized inflammation, fibrosis, and increased surgical complexity (14-16).
- Todani classification: determined according to the widely accepted Todani system (1977).
- Chronic cholecystitis: diagnosed based on preoperative imaging features (e.g., wall thickening, enhancement) and/or confirmed by intraoperative inspection and postoperative histopathology.
Laboratory variables: all blood tests were performed preoperatively. The following markers were recorded with their respective units and reference ranges: C-reactive protein (CRP, mg/L; normal <5–10 mg/L); white blood cell count (WBC, ×109/L; normal 4×109–10×109/L); alanine aminotransferase (ALT, U/L; normal <40 U/L); direct bilirubin (DB, µmol/L; normal 0–8 µmol/L); total bilirubin (TB, µmol/L; normal 2–20 µmol/L).
All imaging and laboratory data were reviewed independently by two clinicians; discrepancies were resolved by consensus or by a third senior reviewer.
Postoperative outcomes
The primary endpoint was postoperative complications, including mid-to-long-term morbidity and mortality. Children dead during the follow-up was excluded. Complications were categorized temporally as early (occurring within 30 days postoperatively: e.g., biliary leakage, pancreatic fistula, surgical site infection) or late (occurring after 6 months: e.g., anastomotic stricture, choledocholithiasis, chronic pancreatitis). Event was defined as the absence of complication events during follow-up.
Follow-up
Patients underwent follow-up assessments every 6 months via outpatient visits and telephone interviews to monitor long-term complications. The median follow-up duration for all enrolled patients was 3.4 years (range, 1–5 years).
The laparoscopic Roux-en-Y hepaticojejunostomy procedure
The devices used were SRORZ pediatric laparoscopic devices (SYZBA-2010142, 7220BA/26005BAK). Following supine positioning and standard anesthesia, a 0.5-cm umbilical incision established CO2 pneumoperitoneum (6–8 mmHg). Five-millimeter trocars were placed: umbilical (optical port), bilateral subcostal (operating ports), and epigastric (auxiliary port). Teres ligament suspension facilitated hepatic retraction and cyst exposure. Intraoperative cholangiography (30 mL contrast) under C-arm guidance defined cyst anatomy and pancreaticobiliary junction. Electrocautery dissection preceded cholecystectomy and common bile duct transection distal to the cyst. The cyst was resected to the common hepatic duct confluence. Extracorporeal jejunal preparation included: 15-cm Roux limb division, 35-cm hepatic jejunostomy via linear stapler, and anti-reflux seromuscular fixation. Intracorporeal Roux-en-Y reconstruction featured retrocolic passage and continuous knotless hepaticojejunostomy, with final drainage in the lesser sac (17).
Statistical analysis
All analyses were performed using R software, version 3.0. Categorical variables were summarized as frequencies and percentages and compared using the chi‑square test. Continuous variables were assessed for normality with the Shapiro-Wilk test. Normally distributed data are presented as mean ± standard deviation and compared using the independent t‑test; non‑normally distributed data are expressed as median [interquartile range (IQR)] and compared using the Wilcoxon rank‑sum test. Univariable and multivariable logistic regression were employed to identify risk factors for postoperative complications. All continuous variables (including CRP and DB) were entered into the regression models as continuous predictors; no predefined cut‑off values were used for dichotomization. Categorical predictors (e.g., cyst wall thickness >5 mm, Todani type I, chronic cholecystitis) were included as binary variables. Results are reported as odds ratios (ORs) with 95% confidence intervals (CIs). Event‑free survival was analyzed using Cox proportional hazards regression. Variables with a two‑sided P<0.05 in the multivariable model were incorporated into a nomogram for individualized risk prediction.
Results
General characteristics
A total of 362 patients undergoing laparoscopic surgery between August 2019 and July 2024 were enrolled in this study. The derivation of the final study cohort is detailed in Figure 1. Among them, 34 (9.39%) patients were categorized into the event group, while 328 (90.61%) patients comprised the non-event group. Baseline demographic, clinical, and laboratory characteristics of the cohort are summarized in Table 1.
Table 1
| Variable | Non-event (n=328) | Event (n=34) | P |
|---|---|---|---|
| Age (years) | 3.4 (1.62–4.83) | 1.5 (1.15–3.72) | 0.01 |
| Male | 92 (28.0) | 10 (29.4) | 0.58 |
| Operation time (min) | 208.1±28.0 | 214.5±32.8 | 0.07 |
| Clinical symptoms (yes) | 118 (36.0) | 16 (47.1) | 0.03 |
| Thickness of cyst wall (mm) | <0.001 | ||
| >5 | 85 (25.9) | 22 (64.7) | |
| ≤5 | 243 (74.1) | 12 (35.3) | |
| Chronic cholecystitis | 103 (31.4) | 27 (79.4) | 0.008 |
| Diameter of cyst (cm) | 3.1 (1.9–4.2) | 3.7 (2.4–5.3) | 0.04 |
| Todani classification | <0.001 | ||
| Type IVb | 186 (56.7) | 3 (8.8) | |
| Type I | 142 (43.3) | 31 (91.2) | |
| CRP (mg/L) | 1.49 (0.58–4.36) | 7.05 (5.69–9.32) | <0.001 |
| WBC (×109/L) | 6.80 (5.24–9.50) | 7.24 (6.29–14.38) | 0.057 |
| ALT (U/L) | 27.52 (25.34–29.50) | 38.83 (32.67–44.58) | 0.06 |
| DB (µmol/L) | 2.64 (1.32–3.79) | 3.75 (2.43–6.52) | 0.03 |
| TB (µmol/L) | 11.20 (9.06–13.24) | 13.80 (9.52–16.58) | 0.61 |
Categorical data are presented as number (percentage); continuous data are presented as mean ± standard deviation if normally distributed, or as median (interquartile range). P value <0.05 was considered statistically significant. ALT, alanine aminotransferase; CRP, C‑reactive protein; DB, direct bilirubin; TB, total bilirubin; WBC, white blood cell count.
Compared with the non-event group, the event group demonstrated significantly younger age, higher incidence of clinical symptoms, greater cyst wall thickness (mm), increased prevalence of chronic cholecystitis, larger cyst diameter, higher proportion of Todani type I classification, elevated CRP levels, and elevated DB levels. However, no statistically significant differences were observed between the groups regarding gender distribution, operative time, WBC, ALT, or TB.
Prevalences of different type of complications
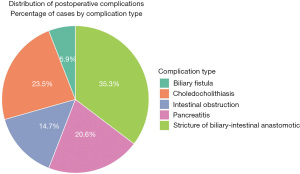
Postoperative complication rates following laparoscopic Roux-en-Y hepaticojejunostomy are detailed in Figure 2, demonstrating the following distribution: stricture of biliary-intestinal anastomotic (35.3%), choledocholithiasis (23.5%), pancreatitis (20.6%), intestinal obstruction (14.7%), and biliary fistula (5.9%).
Risk factors for postoperative complications after laparoscopic surgery for CCCs based on logistic regression analysis
Both univariate and multivariate logistic regression analyses identified five significant risk factors for complications following laparoscopic Roux-en-Y hepaticojejunostomy in children with choledochal cysts: cyst wall thickness >5 mm, chronic cholecystitis, Todani type I classification, elevated CRP and elevated DB (Table 2 and Figure 3). Notably, the multivariate regression-based nomogram demonstrated that cyst wall thickness >5 mm, Todani type I classification, and DB level exerted the greatest impact on complication risk (Figure 4).
Table 2
| Variables | Univariable | Multivariable | |||
|---|---|---|---|---|---|
| OR (95% CI) | P value | OR (95% CI) | Adjusted P value | ||
| Age (years) | 1.72 (0.37–3.21) | 0.01 | 0.97 (0.25–3.49) | 0.78 | |
| Male | 1.35 (0.62–3.38) | 0.60 | |||
| Operation time (min) | 1.19 (0.48–2.37) | 0.62 | |||
| Clinical symptoms (yes) | 2.34 (1.45–3.46) | 0.052 | |||
| Thickness of cyst wall (>5 mm) | 5.69 (2.49–7.30) | <0.001 | 4.23 (1.26–6.75) | <0.001 | |
| Chronic cholecystitis (with) | 7.31 (2.06–10.39) | <0.001 | 6.42 (3.39–9.24) | <0.001 | |
| Diameter of cyst (cm) | 1.24 (0.65–2.74) | 0.55 | |||
| Todani classification (type I) | 8.27 (3.12–12.68) | <0.001 | 6.13 (2.74–10.38) | 0.002 | |
| CRP (mg/L) | 2.23 (0.86–3.65) | 0.03 | 1.52 (1.12–2.16) | 0.04 | |
| WBC (×109/L) | 1.58 (0.74–3.52) | 0.87 | |||
| ALT (U/L) | 1.02 (0.42–2.14) | 0.70 | |||
| DB (µmol/L) | 2.42 (0.78–4.59) | 0.03 | 2.05 (1.23–4.17) | 0.04 | |
| TB (µmol/L) | 3.29 (0.53–5.36) | 0.89 | |||
P value (or adjusted P value) <0.05 was considered statistically significant. ALT, alanine aminotransferase; CI, confidence interval; CRP, C‑reactive protein; DB, direct bilirubin; OR, odds ratio; TB, total bilirubin; WBC, white blood cell count.


Risk factors for different type of complications after laparoscopic surgery for CCCs based on logistic regression analysis
Following the identification of key risk factors for complications, we conducted subgroup analyses to evaluate their impact on specific complication categories: biliary-enteric anastomotic stricture (Figure 5A), choledocholithiasis (Figure 5B), and pancreatitis (Figure 5C). Our analysis revealed that cyst wall thickness >5 mm, chronic cholecystitis, Todani type I classification, and elevated CRP levels significantly influenced all three complication subtypes (Table 3 and Figure 5A-5C). In contrast, elevated DB levels did not demonstrate statistical significance in the biliary-enteric anastomotic stricture and pancreatitis subgroups.

Table 3
| Variables | Stricture of biliary-intestinal anastomotic | Choledocholithiasis | Pancreatitis | |||||
|---|---|---|---|---|---|---|---|---|
| OR (95% CI) | P value | OR (95% CI) | P value | OR (95% CI) | P value | |||
| Thickness of cyst wall (>5 mm) | 4.46 (2.35–6.91) | <0.001 | 3.52 (2.24–7.13) | <0.001 | 5.43 (2.63–7.44) | <0.001 | ||
| Chronic cholecystitis | 5.13 (1.47–7.62) | <0.001 | 3.62 (1.96–5.42) | <0.001 | 3.38 (1.24–6.49) | <0.001 | ||
| Todani classification (type I) | 8.37 (3.54–10.45) | <0.001 | 7.82 (4.15–11.39) | <0.001 | 6.62 (3.41–10.24) | <0.001 | ||
| CRP (mg/L) | 3.26 (1.15–5.48) | 0.01 | 2.48 (1.69–4.01) | 0.04 | 2.04 (1.17–5.12) | 0.03 | ||
| DB (µmol/L) | 1.21 (0.83–4.15) | 0.07 | 2.41 (1.84–5.37) | 0.02 | 1.49 (1.02–4.58) | 0.06 | ||
P value (or adjusted P value) <0.05 was considered statistically significant. CI, confidence interval; CRP, C‑reactive protein; DB, direct bilirubin; OR, odds ratio.
Event-free survival curves for complications after laparoscopic surgery according to the thickness of cyst wall
The above results demonstrated that cyst wall thickness serves as a significant predictor for complications in choledochal cysts. A univariable Cox regression was subsequently constructed to evaluate this association. As shown in Figure 6, the main event-free survival curve and its magnified inset (which focuses on the early-to-mid follow-up phase to clarify curve separation) demonstrate that patients with a cyst wall thickness >5 mm had a significantly higher risk of complications compared to those with a thickness ≤5 mm (P=0.04).

Discussion
Key findings
This study identified several independent risk factors for postoperative complications following laparoscopic choledochal cyst excision in children. A cyst wall thickness >5 mm, the presence of Todani type I anatomy, and elevated preoperative levels of DB and CRP were significantly associated with an increased risk of complications in multivariable analysis. Furthermore, a younger age at surgery and the presence of chronic cholecystitis were also significant predictors in univariable analysis.
Strengths and limitations
A key strength of this study is the development of a multivariable model that synthesizes several readily available preoperative clinical and imaging variables into a risk stratification framework, which holds potential for clinical translation. The findings are reinforced by the cohort’s size and the consistency of identified risk factors with established pathological mechanisms of choledochal cyst disease.
Several limitations must be acknowledged. The retrospective, two-center design introduces potential selection and information biases. The model was developed and validated internally; therefore, its generalizability requires confirmation through external validation in independent, preferably prospective, cohorts. The sample size, while substantial for a rare condition, may have limited the power to detect other significant predictors, such as cyst diameter. Furthermore, the >5 mm cutoff, though supported by existing evidence, remains arbitrary and requires external validation. Future models may treat cyst wall thickness as a continuous variable or use data-driven cutoffs for greater precision. Finally, we could not account for several technically relevant intraoperative variables (e.g., surgical difficulty scores, precise distance of the choledochal-cyst confluence, and anatomical variations of the hepatic vasculature), which may influence outcomes.
Comparison with similar research
In pediatric populations, CCCs characteristically manifest as abdominal pain, palpable mass, and/or jaundice. Prompt surgical intervention upon diagnosis is essential to prevent recurrent cholangitis, which may precipitate severe sequelae including hepatic cirrhosis, cyst carcinogenesis, rupture, or other life-threatening complications. Laparoscopic cyst excision with Roux-en-Y hepaticojejunostomy has been established as the standard therapeutic approach in pediatric surgery. Notwithstanding its widespread adoption, technical challenges persist due to restricted operative visualization, anatomical variations of the hepatic ductal system, and cyst wall inflammation—particularly in pediatric anatomy. Thus, comprehensive risk factor identification remains paramount for optimizing clinical outcomes in children with CCC.
Our study found that 34 patients (9.39%) with complications versus 328 (90.61%) without complications, aligning with existing literature. Zhuansun et al. (11) retrospectively analyzed 155 pediatric choledochal cyst patients (2014–2017), reporting complication rates of 9.5% (4/42) in 3D laparoscopic and 15.0% (17/113) in 2D laparoscopic groups. Similarly, Lin et al. (14) documented a 10.9% (17/156) complication rate among 156 children undergoing cyst resection with Roux-en-Y hepaticojejunostomy (2016–2022). Furthermore, we found the predominant complications were Stricture of biliary-intestinal anastomotic (35.3%), choledocholithiasis (23.5%), pancreatitis (20.6%),which was few reported by other studies.
To investigate the risk factors of complication, pediatric characteristic analysis was conducted and revealed children with complications exhibited significantly younger age, increased cyst wall thickness (>5 mm), chronic cholecystitis, greater Todani type I prevalence, and elevated CRP/DB levels. Moreover, regression analysis confirmed these attributes as key risk factors, with cyst wall thickness >5 mm, Todani type I, and DB level demonstrating the most significant impact. Furthermore, cox regression found that children with cyst wall thickness >5 mm had a significantly higher risk of complications.
Explanations of findings
Age constitutes a significant risk factor, as demonstrated by its statistical significance in univariable regression analysis. This association may be attributable to the heightened vulnerability of pediatric patients to biliary obstruction, spontaneous perforation, and hepatic cirrhosis in the natural course of the disease, coupled with cyst-related infections and adhesion formation that impair growth and development (18,19). However, age was not an independent predictor in the multivariable model, indicating that its effect may be explained by other factors such as cyst wall thickness or chronic cholecystitis. Thus, while younger children often present with more advanced disease, the direct contribution of age to complication risk appears modest when these preoperative variables are considered. Consequently, early surgical intervention represents a clinical imperative for choledochal cysts in infants and young children.
Regression analysis identified cystic wall thickening of the common bile duct as a significant risk factor for postoperative complications. This association may be attributed to inflammation-induced neovascularization and elevated capillary permeability (20). Mechanistically, biliary stenosis or structural anomalies trigger recurrent chronic inflammation at the pancreatobiliary junction. This inflammatory process induces both severe peri-tissue adhesions—increasing surgical difficulty—and smooth muscle fibroplasia within the cystic wall, thereby exacerbating biliary stricture (21). Consequently, the degree of cyst wall thickening serves as an indicator of local inflammatory severity, providing critical guidance for optimizing surgical planning and improving prognostic outcomes. In this study, cyst wall thickness was selected as the primary variable for Cox regression analysis based on the following considerations: First, cyst wall thickness >5 mm emerged as the strongest and most consistent imaging-based predictor of complications in both univariable and multivariable logistic regression analyses. Second, this parameter is readily measurable on routine preoperative imaging and directly reflects the degree of local inflammation and fibrosis, which is closely associated with surgical difficulty and prognosis. Finally, its pathophysiological basis is closely linked to the timing of complication development, making it well-suited for analyzing time-to-event outcomes. In contrast, cyst diameter did not demonstrate independent predictive value in this study and was therefore not included as a core variable in the survival analysis; this finding warrants further validation in larger cohorts.
Elevated DB constitutes a key risk factor for postoperative complications in CCCs through three synergistic mechanisms: (I) serving as a biomarker of biliary obstruction (e.g., biliary atresia) or hepatocellular transport dysfunction, indicating underlying stenosis or inflammation (22); (II) directly increasing cholangitis risk (23); and (III) reducing intraoperative hepatic tolerance while delaying postoperative functional recovery. Notably, while DB emerged as an independent predictor in our overall multivariate model, subgroup analysis revealed its association was strongest for choledocholithiasis. This may be attributed to the direct role of DB in promoting biliary stasis, sludge formation, and lithogenic bile composition—key pathophysiological processes in stone development. In contrast, its association with biliary-enteric anastomotic stricture and pancreatitis was less pronounced, suggesting that these complications are more strongly driven by local fibrotic changes, surgical factors, or PBM rather than bilirubin levels alone.
Preoperative chronic cholangitis constitutes a significant risk factor for postoperative complications due to recurrent inflammation-induced fibrotic changes in the common bile duct, which increases surgical complexity during reoperation and compromises bile-intestinal anastomotic integrity (24). Furthermore, elevated serum CRP levels serve as an independent risk predictor, directly correlating with cholangitis severity and reflecting underlying infectious processes (25).
This study identifies Todani type I as a significant risk factor for pediatric choledochal cysts. Type Ic cysts demonstrate dual pathological characteristics: association with mild biliary stenosis (26) and abnormal pancreatobiliary junctions that elevate postoperative pancreatitis and biliary fistula risks (27,28). Mechanistically, intraductal protein plugs—prevalent in type I variants—promote bile reflux and pancreatitis. Consequently, multifactorial analysis correlating Todani subtypes with post-Roux-en-Y complications is essential for developing individualized treatment strategies to optimize pediatric outcomes.
Implications and actions needed
The findings have direct clinical implications. Preoperative measurement of cyst wall thickness on imaging, precise Todani classification, and assessment of inflammatory (CRP) and obstructive (DB) markers should be integrated into routine surgical planning. The developed risk model can help surgeons identify high-risk patients preoperatively, allowing for optimized surgical scheduling, enhanced patient counseling, and possibly guiding the allocation of surgical expertise or the consideration of alternative techniques (e.g., robotic assistance) in complex cases.
Future research should focus on the prospective external validation and refinement of this predictive model. Incorporating intraoperative variables (e.g., adhesion scores, operative time) and longer-term follow-up data into future models could enhance their predictive accuracy. Ultimately, a validated, easy-to-use risk score could facilitate personalized surgical management and improve outcomes for children undergoing choledochal cyst excision.
Conclusions
Cyst wall thickness >5 mm, chronic cholecystitis, Todani type I, elevated CRP, and elevated DB were selected as factors to assess the prognosis of children with choledochal cysts, providing reference significance of the prognosis evaluation and treatment for children with choledochal cysts.
Acknowledgments
None.
Footnote
Reporting Checklist: The authors have completed the STROBE reporting checklist. Available at https://tp.amegroups.com/article/view/10.21037/tp-2025-1-948/rc
Data Sharing Statement: Available at https://tp.amegroups.com/article/view/10.21037/tp-2025-1-948/dss
Peer Review File: Available at https://tp.amegroups.com/article/view/10.21037/tp-2025-1-948/prf
Funding: None.
Conflicts of Interest: All authors have completed the ICMJE uniform disclosure form (available at https://tp.amegroups.com/article/view/10.21037/tp-2025-1-948/coif). The authors have no conflicts of interest to declare.
Ethical Statement: The authors are accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved. The study was conducted in accordance with the Declaration of Helsinki and its subsequent amendments. The study was approved by the Ethics Committee of Leshan Women and Children Hospital (No. Lsfu250236) and informed consent was obtained from all patients’ legal guardians. Dongzhimen Hospital, Beijing University of Chinese Medicine was also informed and agreed the study.
Open Access Statement: This is an Open Access article distributed in accordance with the Creative Commons Attribution-NonCommercial-NoDerivs 4.0 International License (CC BY-NC-ND 4.0), which permits the non-commercial replication and distribution of the article with the strict proviso that no changes or edits are made and the original work is properly cited (including links to both the formal publication through the relevant DOI and the license). See: https://creativecommons.org/licenses/by-nc-nd/4.0/.
References
- Soares KC, Arnaoutakis DJ, Kamel I, et al. Choledochal cysts: presentation, clinical differentiation, and management. J Am Coll Surg 2014;219:1167-80. [Crossref] [PubMed]
- Olbourne NA. Choledochal cysts. A review of the cystic anomalies of the biliary tree. Ann R Coll Surg Engl 1975;56:26-32. [PubMed]
- Kim HJ, Kim MH, Lee SK, et al. Normal structure, variations, and anomalies of the pancreaticobiliary ducts of Koreans: a nationwide cooperative prospective study. Gastrointest Endosc 2002;55:889-96. [Crossref] [PubMed]
- Todani T, Watanabe Y, Fujii T, et al. Anomalous arrangement of the pancreatobiliary ductal system in patients with a choledochal cyst. Am J Surg 1984;147:672-6. [Crossref] [PubMed]
- Kemmotsu H, Mouri T, Muraji T. Congenital stenosis of the hepatic duct at the porta hepatis in children with choledochal cyst. J Pediatr Surg 2009;44:512-6. [Crossref] [PubMed]
- Xiao J, Chen M, Hong T, et al. Surgical Management and Prognosis of Congenital Choledochal Cysts in Adults: A Single Asian Center Cohort of 69 Cases. J Oncol 2022;2022:9930710. [Crossref] [PubMed]
- Ishibashi T, Kasahara K, Yasuda Y, et al. Malignant change in the biliary tract after excision of choledochal cyst. Br J Surg 1997;84:1687-91. [PubMed]
- Liuming H, Hongwu Z, Gang L, et al. The effect of laparoscopic excision vs open excision in children with choledochal cyst: a midterm follow-up study. J Pediatr Surg 2011;46:662-5. [Crossref] [PubMed]
- Diao M, Li L, Cheng W. Laparoscopic versus Open Roux-en-Y hepatojejunostomy for children with choledochal cysts: intermediate-term follow-up results. Surg Endosc 2011;25:1567-73. [Crossref] [PubMed]
- Urushihara N, Hamada Y, Kamisawa T, et al. Classification of pancreaticobiliary maljunction and clinical features in children. J Hepatobiliary Pancreat Sci 2017;24:449-55. [Crossref] [PubMed]
- Zhuansun D, Jiao C, Meng X, et al. A Study of Three-Dimensional Versus Two-Dimensional Laparoscopic Surgery in Resection of Congenital Choledochal Cyst of Children and Jejunum Roux-en-Y Anastomosis. J Laparoendosc Adv Surg Tech A 2020;30:344-9. [Crossref] [PubMed]
- Koshinaga T, Inoue M, Ohashi K, et al. Persistent biliary dilatation and stenosis in postoperative congenital choledochal cyst. J Hepatobiliary Pancreat Sci 2011;18:47-52. [Crossref] [PubMed]
- Tang ST, Yang Y, Wang Y, et al. Laparoscopic choledochal cyst excision, hepaticojejunostomy, and extracorporeal Roux-en-Y anastomosis: a technical skill and intermediate-term report in 62 cases. Surg Endosc 2011;25:416-22. [Crossref] [PubMed]
- Lin Y, Xu X, Chen S, et al. Construction of nomogram based on clinical factors for the risk prediction of postoperative complications in children with choledochal cyst. Front Pediatr 2024;12:1372514. [Crossref] [PubMed]
- Cochran ED, Lazow SP, Kim AG, et al. The in-utero diagnosis of choledochal cyst: can postnatal imaging predict benefit from early surgical intervention? J Matern Fetal Neonatal Med 2022;35:1070-4. [Crossref] [PubMed]
- Guan X, Li J, Wang Z, et al. Timing of operation in children with a prenatal diagnosis of choledochal cyst: A single-center retrospective study. J Hepatobiliary Pancreat Sci 2022;29:1308-15. [Crossref] [PubMed]
- Ure BM, Schier F, Schmidt AI, et al. Laparoscopic resection of congenital choledochal cyst, choledochojejunostomy, and extraabdominal Roux-en-Y anastomosis. Surg Endosc 2005;19:1055-7. [Crossref] [PubMed]
- Ishibashi H, Shimada M, Kamisawa T, et al. Japanese clinical practice guidelines for congenital biliary dilatation. J Hepatobiliary Pancreat Sci 2017;24:1-16. [Crossref] [PubMed]
- Diao M, Li L, Cheng W. Congenital biliary dilatation may consist of 2 disease entities. J Pediatr Surg 2011;46:1503-9. [Crossref] [PubMed]
- Kamisawa T, Ando H, Suyama M, et al. Japanese clinical practice guidelines for pancreaticobiliary maljunction. J Gastroenterol 2012;47:731-59. [Crossref] [PubMed]
- Hwang HS, Kim MJ, Lee SS, et al. Smooth Muscle Distribution Patterns of Choledochal Cysts and Their Implications for Pathogenesis and Postoperative Complications. Am J Clin Pathol 2020;153:760-71. [Crossref] [PubMed]
- Harpavat S, Finegold MJ, Karpen SJ. Patients with biliary atresia have elevated direct/conjugated bilirubin levels shortly after birth. Pediatrics 2011;128:e1428-33. [Crossref] [PubMed]
- Yan L, Yan L. Relationship between hepatic function recovery and postoperative cholangitis in neonates undergoing hepaticojejunostomy for biliary atresia. Am J Transl Res 2024;16:3822-31. [Crossref] [PubMed]
- Diao M, Li L, Cheng W. Recurrence of biliary tract obstructions after primary laparoscopic hepaticojejunostomy in children with choledochal cysts. Surg Endosc 2016;30:3910-5. [Crossref] [PubMed]
- Umefune G, Kogure H, Hamada T, et al. Procalcitonin is a useful biomarker to predict severe acute cholangitis: a single-center prospective study. J Gastroenterol 2017;52:734-45. [Crossref] [PubMed]
- Todani T, Watanabe Y, Toki A, et al. Classification of congenital biliary cystic disease: special reference to type Ic and IVA cysts with primary ductal stricture. J Hepatobiliary Pancreat Surg 2003;10:340-4. [Crossref] [PubMed]
- Zhu L, Xiong J, Lv Z, et al. Type C Pancreaticobiliary Maljunction Is Associated With Perforated Choledochal Cyst in Children. Front Pediatr 2020;8:168. [Crossref] [PubMed]
- Fujishiro J, Masumoto K, Urita Y, et al. Pancreatic complications in pediatric choledochal cysts. J Pediatr Surg 2013;48:1897-902. [Crossref] [PubMed]