Unlocking the puzzle of primary monosymptomatic nocturnal enuresis: a comprehensive study on the management of Thai children through alarm therapy and medication
Highlight box
Key findings
• Alarm therapy alone or in combination therapy provides better long-term results than desmopressin alone.
• Age and sex have minimal predictive value.
• Cultural context and family engagement significantly influence adherence and treatment success.
What is known and what is new?
• Primary mono-symptomatic nocturnal enuresis (PMNE) affects about 15% of 5-year-old children and declines with age. It has multifactorial causes. Alarm therapy is first-line due to low recurrence, while desmopressin is used for nocturnal polyuria or rapid control. Combination therapy may be used in difficult cases.
• This study highlights the importance of culturally adapted management and caregiver involvement, demonstrating their significant impact on treatment adherence and outcomes in Thai children.
What is the implication, and what should change now?
• In Thailand, enuresis is often viewed as a behavioral issue, causing delayed consultation and punitive approaches.
• Families prefer non-medical methods. However, shared sleeping environments limit the use of alarm therapy.
• Clinical guidelines should include the cultural differences that explain the biology of PMNE and promote empathetic parenting.
• Tailored interventions that include community education, tele-coaching, and modified alarm systems for shared settings should be developed.
• Guidelines should include family education about the causes of PMNE, ways to give support, and the impact of effective parenting.
Introduction
Primary monosymptomatic nocturnal enuresis (PMNE) is identified as an involuntary nocturnal urinary episode, occurring with a frequency of at least once per week over a consecutive 3-month period, predominantly in children aged 5 years and older (1). Epidemiological assessments indicate that 15% of this age group are impacted by PMNE, with a greater incidence in male children as compared to females. Notably, the prevalence of this condition demonstrates a decremental trend with increasing age across various Asian locales, including Taiwan (2), Malaysia (3), and Saudi Arabia (4), culminating from 15% at age 5 years to 1–2% post-15 years (5,6). In Thailand, a study conducted (7) a decade and a half ago showcased a 3.9% prevalence rate among school-aged youth, aligning with the Diagnostic and Statistical Manual of Mental Disorders, 5th edition (DSM-V) categorization (8). In stark comparison (9), the year 2023 recorded a prevalence of 9.7%, 11.6%, and 8.2% for primary monosymptomatic, non-monosymptomatic, and secondary nocturnal enuresis, respectively, among Thai children aged between 5 and 15 years, mirroring the international prevalence metrics (1-3). The etiological framework of PMNE suggests a substantial genetic (10) contribution, where the likelihood of occurrence in progeny is 44% with one affected parent and ascends to 77% with both. Analysis in twin cohorts underscores a heightened concurrence in monozygotic twins relative to dizygotic twins, hinting at a significant genetic influence (11), yet a singular genetic locus (12) remains unidentified.
Contemporary guidelines (13-15) propose assertive management for children older than 7 years who exhibit persistent PMNE symptoms post-behavioral modification, aimed at averting potential detrimental impacts (16) on psychological well-being, educational performance (17), and inter-familial relationships (18). The imperative of such interventions is underscored by the potential escalation of anxiety levels in mothers (19) of enduring PMNE children. Addressing PMNE mandates a comprehensive treatment paradigm, incorporating psychological (20) and physiological considerations (21), given its propensity to disrupt sleep (22,23). Debates persist regarding the initiation timing and choice of the optimal treatment modality (24-26).
The implementation of an enuretic alarm, innovated by P. Faundler in the early 20th century (27), remains a cornerstone in PMNE management, endorsed for its efficacy (28), cost-effectiveness, and safety, accompanied by a notable reduction in recurrence rates. Contemporary research (29) is dedicated to pinpointing specific pediatric subgroups that would most benefit from this intervention, suggesting that children with standard or reduced bladder capacities and typical nocturnal urine production exhibit improved outcomes with alarm therapy. Nevertheless, the relationship between bladder capacity and response to treatment remains variably reported across different studies (30). Recent scholarly work (29) suggests that the integration of alarm therapy with pharmacological treatment or the exclusive use of pharmacotherapy may enhance therapeutic outcomes. A study from Denmark (28) highlighted that patients responding favorably to alarm therapy typically show a reduction in nocturnal urine output over time, whereas those responding to medication demonstrate increased nocturnal urination, with no significant difference in bladder functionality discerned between the responsive and non-responsive cohorts.
The formulation of inclusive, culturally sensitive treatment guidelines for PMNE is a subject of ongoing scholarly debate, emphasizing the need to respect diverse cultural, societal, and familial values globally. This research aims to assess the efficacy of various bedwetting interventions—alarm therapy, medication, and their combination—within the Thai pediatric population, aiming to identify key factors that influence treatment success and recurrence. The ultimate goal is to develop effective, culturally adapted management strategies for PMNE in Thai children. We present this article in accordance with the STROBE reporting checklist (available at https://tp.amegroups.com/article/view/10.21037/tp-2025-aw-716/rc).
Methods
Study population and diagnostic protocol
This retrospective study spans 5 years and involves data from 300 children, aged 5 to 18 years, who were treated for nocturnal enuresis at Queen Sirikit National Institute of Child Health from 2020 to 2024. Of these children, 160 were diagnosed with various forms of nocturnal enuresis, while 140 were diagnosed with PMNE and underwent treatment at the child development center. These 140 children provided information on standard treatment approaches to both the children themselves and their families. All participants voluntarily chose to use an enuresis alarm as the initial treatment. After 2 weeks of using the alarm, 52 children showed improvement, whereas 88 continued to experience bedwetting. Among those who did not respond to the alarm treatment alone, 48 received a combined treatment of the alarm and desmopressin, and 40 were treated with desmopressin alone (Figure 1).

The cohort, encompassing both genders, exhibited a mean age of 11 years, with an age dispersion ranging from 11.2 to 11.8 years. The incidence of nocturnal episodes was quantified between 5.13 and 6.23 episodes per week. Rigorous physical and urological assessments were conducted for each participant. Exclusion criteria encompassed manifestations of lower urinary tract symptoms (LUTS), exceeding eight urination instances per day, or the presence of significant physical, neurological, urological, or developmental irregularities. Subjects presenting with constipation or snoring underwent urinalysis. Urinary frequencies were meticulously documented during diurnal and nocturnal periods across two successive weekends, spanning a fortnight. Following the aggregation of these data, nocturnal monitoring was sustained until symptomatic amelioration was observed. Calibration and validation of all measurement apparatuses were executed by the Child Development Center at Queen Sirikit National Institute of Child Health. The intervention utilized one model of enuresis alarms: the Stay Dry® Bedwetting Alarm for Kids. The enuresis alarm model is equipped with an auditory signaling device and offers the option of vibration alerts, allowing for customization based on individual needs.
Enuresis alarm intervention protocol
Uniformity in treatment was maintained by allocating the same bedwetting alarm model to all participants under the vigilant supervision of a developmental pediatrician. The treatment regimen was complemented by online consultations and monthly in-person evaluations. Response to treatment was stratified based on the International Children’s Continence Society’s (ICCS’s) (1) 2015 benchmarks: non-responsiveness was ascribed to a diminution in enuresis frequency of less than 50% by the second week, partial response corresponded to a 50–99% reduction, and complete response was defined by an absolute cessation of bedwetting incidents by week 2.
Among 140 children, 52 showed improvement after 2 weeks of using alarms. This group continued the same treatment with ongoing follow-up until sustained improvement. For non-responders, adjunctive strategies were deliberated, encompassing exclusive pharmacotherapy or a hybrid of medication and alarm intervention. Desmopressin (31) was administered at an inaugural dosage of 0.2 mg or a 120-mcg sublingual lyophilizate, 1 hour pre-sleep, with a stipulation for abstinence from fluid intake 1 hour pre- and up to 8 hours post-administration to avert hyponatremia. Follow-up assessments were scheduled biweekly to evaluate therapeutic progress, with potential dosage escalation to 0.4 mg or 240 mcg if warranted. A gradual reduction in desmopressin dosage was advocated following symptomatic improvement, decrementing by 50% biweekly until cessation.
Statistical analysis
Statistical analyses were performed using SPSS version 26.0 (IBM Corp., Armonk, NY, USA). Categorical variables were summarized as frequencies and percentages and compared using the Chi-squared test or Fisher’s exact test, as appropriate. Continuous variables were expressed as mean ± standard deviation and analyzed using one-way analysis of variance (ANOVA). Post-hoc comparisons were conducted using Tukey’s method. Changes in enuresis frequency over time within each treatment group were evaluated using paired t-tests. A two-sided P value <0.05 was considered statistically significant.
Ethical consideration
This research study was approved by the Office for Ethics in Human Research, Queen Sirikit National Institute of Child Health, Thailand in December 15, 2022 (ref. REC.119/2565), and was conducted in accordance to the Declaration of Helsinki and it subsequent amendments, The Belmont Report, CIOMS Guideline International Conference on Harmonization in Good Clinical Practice (ICH-GCP). Verbal consent was obtained from the participants’ parents/guardians.
Results
Table 1 provides an overview of demographic data, bladder characteristics, and nocturnal urine production for the three treatment groups: alarm clock, drug, and a combination of both. Notably, there are no significant differences in characteristics among the groups, except for the nocturnal urine volume (NUV) (Figure 2). There are no significant differences in the characteristics of the participants.
Table 1
| Characteristics | Alarm clock (n=52) | Desmopressin (n=30) | Alarm clock + desmopressin (n=58) |
|---|---|---|---|
| Male | 27 (51.92) | 20 (66.67) | 35 (60.34) |
| Age (years) | 11.6±0.11 | 11.8±0.12 | 11.2±0.23 |
| Body weight (kg) | 37.80±0.01 | 38.43±0.02 | 35.62±0.01 |
| Enuresis episodes (/week) | 5.13±0.32 | 6.73±0.24 | 6.43±0.22 |
| EBC (mL) | 310.25±0.25 | 336.83±0.35 | 343.7±0.42 |
| NUV (mL) | 470.96±0.32 | 665.17±0.29 | 646.47±0.53 |
| MVV (mL) | 0.84±0.02 | 0.82±0.04 | 0.82±0.04 |
Data are presented as n (%) or mean ± SD. EBC, expected bladder capacity; MVV, maximal voided volume; NUV, nocturnal urine volume; SD, standard deviation.

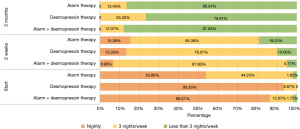
Figure 3 compares the effectiveness of various treatments for bedwetting over time periods starting at the initial treatment, then at 2 weeks, 3 months, and 6 months. The treatments compared are: alarm clock, drug, and alarm clock + drug.
Each bar is divided into three sections representing the frequency of bedwetting episodes: none, more than one episode, and one episode. Key observations from the chart:
- At the start, the ‘alarm clock’ treatment had the highest percentage of ‘none’ frequency at 93.33%, followed by ‘alarm clock + drug’ at 86.21%, and ‘drug’ at 53.85%.
- At 2 weeks, the ‘drug’ treatment showed significant improvement, with 87.93% of children having no bedwetting episodes. ‘Alarm clock’ and ‘alarm clock + drug’ also showed improvements.
- At 3 months, all treatment groups continued to improve, with the ‘alarm clock + drug’ combination showing the highest percentage of no episodes.
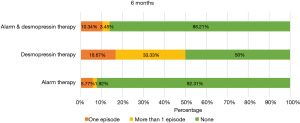
- After a 6-month follow-up, recurrence rates were found in the drug-using group (50%), followed by the combined treatment group (13.79%), with the lowest recurrence rate found in the group using a urination alarm (7.69%), where 6-month recurrence rates are reported (Figure 4).
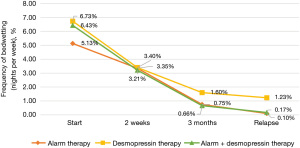
Figure 5 shows that no significant differences were observed in the frequency of wet nights among the three groups (generalized linear model and Tukey’s method), although frequencies in all three groups decreased significantly with therapeutic duration (paired t-test).
The graph elucidates the weekly incidence rates of bedwetting, indicating that the cohort treated with a urinary alarm device manifested the minimal frequency at 5.13%, with the medication and combination treatment groups following at 6.73% and 6.43%, respectively. Upon the 2-week follow-up, a pronounced decline in bedwetting incidents was noted predominantly in the medication group, followed by the combination treatment and alarm device groups, sequentially. Subsequent to the 3-month follow-up, the urinary alarm device and combination treatment cohorts exhibited similar nocturnal bedwetting frequencies, recorded at 0.66 and 0.75, respectively. In contrast, the medication group demonstrated a substantial average decrease of 1.6 incidents per week. At the 6-month juncture, the recurrence rate for the urinary alarm device group was a mere 0.1%, marginally lower than the 0.17% observed in the combination treatment group. The medication group, on the other hand, exhibited a relatively elevated average weekly bedwetting rate of 1.23, positioning it as the least advantageous treatment modality within the comparative framework.
To conduct a thorough statistical analysis, use SPSS to perform tests such as Chi-squared for categorical variables (e.g., improvement rates, sex distribution) and ANOVA for continuous variables [e.g., age, body weight, enuresis episodes, expected bladder capacity (EBC), NUV] to determine if differences are statistically significant. Patient improvement rate: the alarm clock method shows the highest improvement rate (92.31%), followed by the alarm clock and drug combination (86.21%), and the drug-only group shows the least improvement (50%). This suggests that the alarm clock method, alone or combined with medication, might be more effective for this patient group. Gender distribution: there’s a higher percentage of males in the drug-only group (66.67%) compared to the alarm clock (51.92%) and the combination group (60.34%). Age, body weight, and episodes per week: the groups are relatively similar in age and body weight. However, the mean enuresis episodes per week are slightly lower in the alarm clock group. Mean NUV (mL) is lowest in the alarm clock group, suggesting that this group has less NUV, which might be a factor influencing the success of the alarm clock treatment. In comparing the successful cases at 6 months in each group, no significant differences were found in sex, age, body weight, enuretic frequency before treatment, or functional bladder capacity, except nocturnal urinary volume, where predictive factors among improved cases at 6 months are compared (Table 2).
Table 2
| Characteristic | Alarm clock | Drug | Combination | P |
|---|---|---|---|---|
| Total | 48 (92.31) | 15 (50.00) | 50 (86.21) | 0.21 |
| Male | 25/48 (51.92) | 10/15 (66.67) | 37/50 (60.34) | 0.32 |
| Age (years) | 12.6±0.11 | 11.6±0.12 | 11.4±0.23 | 0.41 |
| Body weight (kg) | 38.80±0.01 | 37.42±0.02 | 37.42±0.01 | 0.35 |
| Enuresis episodes (/week) | 6.23±0.32 | 6.53±0.24 | 6.43±0.22 | 0.24 |
| EBC (mL) | 310.25±0.25 | 336.83±0.35 | 343.7±0.42 | 0.09 |
| NUV (mL) | 470.96±0.32 | 645.17±0.29 | 635.47±0.53 | 0.01* |
Data are presented as n (%), n/total (%), or mean ± SD. *, significant (P<0.05). EBC, expected bladder capacity; NUV, nocturnal urinary volume; SD, standard deviation.
Discussion
Nocturnal enuresis, predominantly attributed to physiological (32) factors such as sleep arousal disturbances, nocturnal polyuria, and bladder overactivity, necessitates a multifactorial understanding. It is imperative for caregivers to acknowledge (33) that bedwetting transcends mere psychological origins (34); however, it can be intensified by certain behavioral or mental health conditions, including attention deficits and anxiety (35). Despite initial interventions, children in this cohort, averaging 9 years old, consistently exhibited enuresis, highlighting the critical need for proactive family involvement in management strategies. Parents must be equipped with comprehensive knowledge regarding the management of enuresis, understanding the associated risks and benefits, as well as the practicalities and potential complications of various treatment options. The employment of a voiding chart is a key strategy for elucidating the root causes of enuresis, facilitating tailored treatments (36), and ensuring early-stage collaborative engagement between children and their families. This research highlights the significance of consistent consultation and monitoring of the voiding chart during the initial period to facilitate accurate adjustments. A common oversight is that the recording of the first urine volume in the morning was not taken, leading to potential inaccuracies regarding the overall nightly urine volume. This oversight underscores the importance of preparing the child and family for cooperation, prompting monthly doctor appointments. These regular visits serve to monitor treatment outcomes, provide counseling, and collectively make informed decisions based on guidelines tailored to the specific circumstances of the child and family.
This study revealed the attitudes and perspectives of Thai parents and children towards bedwetting, portraying it as a troublesome yet not severe issue that impacts their mental well-being. Children with normal bladder capacity and nighttime urine volume, in accordance with medical practice guidelines from the ICCS (1) and recommendations from European countries and Taiwan (2), are advised to consider desmopressin or a bedwetting alarm as the initial treatment options, both demonstrating comparable efficacy. If a basic approach like behavior modification (25,26) proves ineffective, all participants in this study opted to begin with a 2-week trial of the bedwetting alarm, assessing outcomes thereafter. Successful responders, characterized by a urine volume reduction of less than 50%, continued with the alarm device. For those who did not show improvement, the other alternative was either medication alone or a combination of medication and an alarm device. Treatment progress was monitored at 2, 4, and 6 months, tracking the number of children and the frequency of bedwetting. Initial results indicated that 93.33%, 86.21%, and 53.85% of children were in the drug and combined treatment groups, and the alarm therapy group, respectively. After 2 weeks of treatment, there was an improved response in all groups. The medication treatment group revealed the fastest response, achieving an 80% decrease, followed by the combination treatment group at 79.31%, and the alarm device group showing the lowest decrease at 38.47%. Supporting researches (37-39) indicate that the frequency of bedwetting gradually diminishes in the initial phase when using alarm devices compared to medication alone. This is attributed to the combined classical conditioning (38), behavior therapy, and negative reinforcement therapy involved in using alarm devices, which require time and consistency. Upon a 6-month treatment follow-up, the lowest recurrence rate of 7.69% was identified in the alarm device group, consistent with previous research (37,38). The combined treatment group showed a recurrence rate of 13.79%, while the drug group exhibited the highest recurrence rate (40) at 50%. The response time towards the use of an alarm device was approximately 3 months, which differed from some studies (40) that reported a longer duration of 5 months. Some researchers (41-43) report difficulties associated with alarm device usage, including skepticism from some families who shared bedrooms and issues such as skin irritation and pressure sores, particularly with older models, which causes unsuccessful treatment with a 30% dropout rate in using alarm devices (44). This study is attributed to a close coaching system and the use of standard alarm devices. In teenagers, empowerment processes within the group reinforce the value of efforts to wake up to use the bathroom, emphasizing small successes over solely focusing on the number of dry nights. Adopting a growth mindset encourages children to reach their goals.
The results of participants with normal and small bladder capacity and experiencing nocturnal polyuria are different in this study. At present, a combination approach is more effective than treatment with an alarm device alone (44,45). In this study, characteristics of bladder capacity did not differ among the three treatment groups, irrespective of normal or low urine capacity and high NUV. The sole indication for drug use to enhance treatment effectiveness is a large amount of urine during the night, as this group of children may not respond adequately to the use of an alarm device alone. After 2- and 4-month follow-ups, an enhancement in treatment efficacy was observed in both the drug-only and combined treatment groups. However, the recurrence rate was higher in the drug-used-only group (46,47). A review article revealed that the combination of desmopressin did not demonstrate superiority (48) over the use of a urinary alarm in patients with normal nighttime urination. However, it proved beneficial in cases involving a substantial amount of nocturnal urine. No serious side effects were identified in this study from the medication, consistent with a systematic review (49) reporting mild side effects from desmopressin, which did not lead to drug discontinuation. Educating patients and families on the potential risk (50) of hyponatremia, limiting water intake, and advising associated symptoms such as headache, nausea, and vomiting are recommended, including recording a voiding diary continuously in order to plan the treatment accordingly.
In a comprehensive study (27,45) involving 423 subjects undergoing a combined therapeutic approach, it was observed that approximately 86% of the participants experienced significant improvements in symptoms. This contrasts with about 68% improvement in those subjected solely to an alarm device. An analysis of baseline demographic data and bladder capacities revealed no significant disparities among the groups responding to treatment, except in the nocturnal urine output. The synergy of pharmacological treatment with alarm device usage was deemed more efficacious in minimizing recurrence rates than the singular application of medication, as supported by a multitude of academic (1,2,35,51) references.
Pharmacotherapy alone is recommended in scenarios necessitating immediate outcomes, such as overnight stays, or in cases where children are unable to engage in behavioral modifications. The advised duration for medication post-improvement typically ranges between 2 weeks and 3 months. However, participants in this study extended their medication use to 4 to 6 weeks before initiating a phased discontinuation. This tapered approach to desmopressin administration was found to be more effective in reducing the likelihood of relapse than an abrupt cessation (50-53), possibly due to concerns over side effects and compliance with pre-sleep hydration restrictions to prevent hypernatremia. Despite the elevated costs associated with medication and bed wetting’s non-life-threatening nature, a preference for behavioral modification was noted among some parents in this research, deeming it a safer and more lasting solution, thus electing to terminate medication use 3 months earlier than internationally recommended (51). Desmopressin therapy has been studied for therapeutic effect prediction. Läckgren et al. (52) found no differences between responders and non-responders in urine output, osmolarity, or vasopressin secretion. In contrast, children with larger bladder capacities respond more to desmopressin. Furthermore, a retrospective (54) examination of the 399 children in the Swedish Enuresis Trial showed that those who improved or were dry with desmopressin medication were older 8 years, had fewer wet nights at baseline, and had just one wet episode throughout the night.
However, in the current investigation, no predictive factors or indicators of the treatment outcomes for the three modalities were observed, while sex, age, body weight, frequency of enuresis, bladder capacity, and preceding except night urine volume. In instances where behavioral modification was insufficient, participants engaged in a 2-week alarm trial, with subsequent outcome evaluations guiding further treatment decisions. Longitudinal monitoring demonstrated notable improvements across all groups, with the alarm group showing the lowest 6-month recurrence rate, advocating for the effectiveness of persistent and consistent alarm use. Notably, the study discussed challenges associated with alarm usage, including familial skepticism and device-related discomfort. It advocates for comprehensive coaching and standardization of alarm use to minimize discontinuation rates. Moreover, the study underscores the varying efficacy of treatment modalities based on individual nocturnal urine output, recommending medication for children with substantial nocturnal urine production who may not respond optimally to alarms alone.
Conclusions
Primary nonnocturnal enuresis treatment using an alarm device is highly effective with a low recurrence rate and is particularly suitable for children with normal nighttime urine volume. Predictive factors of the treatment affect nocturnal urinary volume. For children with nocturnal polyuria, medication may be recommended together with a bedwetting alarm. Notably, the recurrence of bedwetting was found to be more prevalent in the medication treatment group compared to the combined treatment with the bedwetting alarm group.
Acknowledgments
Our thanks to all families who participated in the study. Caregivers’ commitment to the all-day intervention and cooperation of their children were extremely important assets. We are humbled by the trust they placed in us and their gift of helping to gain information that may help other families.
Footnote
Reporting Checklist: The authors have completed the STROBE reporting checklist. Available at https://tp.amegroups.com/article/view/10.21037/tp-2025-aw-716/rc
Data Sharing Statement: Available at https://tp.amegroups.com/article/view/10.21037/tp-2025-aw-716/dss
Peer Review File: Available at https://tp.amegroups.com/article/view/10.21037/tp-2025-aw-716/prf
Funding: None.
Conflicts of Interest: All authors have completed the ICMJE uniform disclosure form (available at https://tp.amegroups.com/article/view/10.21037/tp-2025-aw-716/coif). The authors have no conflicts of interest to declare.
Ethical Statement: The authors are accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved. This research study was approved by the Office for Ethics in Human Research, Queen Sirikit National Institute of Child Health, Thailand in December 15, 2022 (ref. REC.119/2565), and was conducted in accordance to the Declaration of Helsinki and its subsequent amendments, The Belmont Report, CIOMS Guideline International Conference on Harmonization in Good Clinical Practice (ICH-GCP). Verbal consent was obtained from the participants’ parents/guardians.
Open Access Statement: This is an Open Access article distributed in accordance with the Creative Commons Attribution-NonCommercial-NoDerivs 4.0 International License (CC BY-NC-ND 4.0), which permits the non-commercial replication and distribution of the article with the strict proviso that no changes or edits are made and the original work is properly cited (including links to both the formal publication through the relevant DOI and the license). See: https://creativecommons.org/licenses/by-nc-nd/4.0/.
References
- Nevéus T, Fonseca E, Franco I, et al. Management and treatment of nocturnal enuresis-an updated standardization document from the International Children's Continence Society. J Pediatr Urol 2020;16:10-9. [Crossref] [PubMed]
- Chang P, Chen WJ, Tsai WY, et al. An epidemiological study of nocturnal enuresis in Taiwanese children. BJU Int 2001;87:678-81. [Crossref] [PubMed]
- Kanaheswari Y. Epidemiology of childhood nocturnal enuresis in Malaysia. J Paediatr Child Health 2003;39:118-23. [Crossref] [PubMed]
- Yousef KA, Basaleem HO. bin Yahiya MT. Epidemiology of nocturnal enuresis in basic schoolchildren in Aden Governorate, Yemen. Saudi J Kidney Dis Transpl 2011;22:167-73. [PubMed]
- Bakker E, van Sprundel M, van der Auwera JC, et al. Voiding habits and wetting in a population of 4,332 Belgian schoolchildren aged between 10 and 14 years. Scand J Urol Nephrol 2002;36:354-62. [Crossref] [PubMed]
- Forsythe WI, Redmond A. Enuresis and spontaneous cure rate. Study of 1129 enuretis. Arch Dis Child 1974;49:259-63. [Crossref] [PubMed]
- Hansakunachai T, Ruangdaraganon N, Udomsubpayakul U, et al. Epidemiology of enuresis among school-age children in Thailand. J Dev Behav Pediatr 2005;26:356-60. [Crossref] [PubMed]
- American Psychiatric Association. Diagnostic and statistical manual of mental disorders: DSM-5. 5th ed. Arlington: American Psychiatric Association; 2013.
- Sirimongkolchaiyakul O, Sutheparank C, Amornchaicharoensuk Y. The Prevalence of Nocturnal Enuresis in Bangkok, Thailand: A Descriptive and Questionnaire Survey of 5 to 15 Year-Old School Students. Glob Pediatr Health 2023;10:2333794X231189675.
- Nørgaard JP, Djurhuus JC, Watanabe H, et al. Experience and current status of research into the pathophysiology of nocturnal enuresis. Br J Urol 1997;79:825-35. [Crossref] [PubMed]
- Jørgensen CS, Horsdal HT, Rajagopal VM, et al. Identification of genetic loci associated with nocturnal enuresis: a genome-wide association study. Lancet Child Adolesc Health 2021;5:201-9. [Crossref] [PubMed]
- von Gontard A, Heron J, Joinson C. Family history of nocturnal enuresis and urinary incontinence: results from a large epidemiological study. J Urol 2011;185:2303-6. [Crossref] [PubMed]
- EAU Guidelines. Paediatric Urology. Available online: https://uroweb.org/guidelines/paediatric-urology
- National Clinical Guideline Centre. Nocturnal Enuresis: The Management of Bedwetting in Children and Young People. London: Royal College of Physicians; 2010.
- Bayne AP, Skoog SJ. Nocturnal enuresis: an approach to assessment and treatment. Pediatr Rev 2014;35:327-34; quiz 335. [Crossref] [PubMed]
- Harris J, Lipson A, Dos Santos J. Evaluation and management of enuresis in the general paediatric setting. Paediatr Child Health 2023;28:362-76. [Crossref] [PubMed]
- Fagundes SN, Lebl AS, Azevedo Soster L, et al. Monosymptomatic nocturnal enuresis in pediatric patients: multidisciplinary assessment and effects of therapeutic intervention. Pediatr Nephrol 2017;32:843-51. [Crossref] [PubMed]
- Weaver A. Implementing the new NICE guideline for enuresis. Br J Sch Nurs 2010;5:479-83. [Crossref]
- Quiroz-Guerrero J, Ortega-Pardo A, Maldonado-Valadez RE, et al. Maternal Anxiety Associated with Nocturnal Childhood Enuresis. Children (Basel) 2022;9:1232. [Crossref] [PubMed]
- Sarici H, Telli O, Ozgur BC, et al. Prevalence of nocturnal enuresis and its influence on quality of life in school-aged children. J Pediatr Urol 2016;12:159.e1-6. [Crossref] [PubMed]
- Kanaheswari Y, Poulsaeman V, Chandran V. Self-esteem in 6- to 16-year-olds with monosymptomatic nocturnal enuresis. J Paediatr Child Health 2012;48:E178-82. [Crossref] [PubMed]
- Nevéus T. Diagnosis and management of nocturnal enuresis. Curr Opin Pediatr 2009;21:199-202. [Crossref] [PubMed]
- Veeravigrom M, Desudchit T. Prevalence of Sleep Disorders in Thai Children. Indian J Pediatr 2016;83:1237-41. [Crossref] [PubMed]
- Deshpande AV, Caldwell PH, Sureshkumar P. Drugs for nocturnal enuresis in children (other than desmopressin and tricyclics). Cochrane Database Syst Rev 2012;12:CD002238. [Crossref] [PubMed]
- Nevéus T, Läckgren G, Tuvemo T, et al. Desmopressin resistant enuresis: pathogenetic and therapeutic considerations. J Urol 1999;162:2136-40. [Crossref] [PubMed]
- DiBianco JM, Morley C, Al-Omar O. Nocturnal enuresis: A topic review and institution experience. Avicenna J Med 2014;4:77-86. [Crossref] [PubMed]
- Turner RK. Conditioning treatment of nocturnal enuresis: present status. In: Kolvin I, MacKeith RC, Meadow SR. editors. Bladder control and enuresis. London: William Heinemann Medical Books; 1973:195-210.
- Kamperis K, Hagstroem S, Rittig S, et al. Combination of the enuresis alarm and desmopressin: second line treatment for nocturnal enuresis. J Urol 2008;179:1128-31. [Crossref] [PubMed]
- Robson L. How to cure bedwetting. Victoria: Friesen Press; 2016.
- Radvanska E, Kovács L, Rittig S. The role of bladder capacity in antidiuretic and anticholinergic treatment for nocturnal enuresis. J Urol 2006;176:764-8; discussion 768-9. [Crossref] [PubMed]
- Gasthuys E, Dossche L, Michelet R, et al. Pediatric Pharmacology of Desmopressin in Children with Enuresis: A Comprehensive Review. Paediatr Drugs 2020;22:369-83. [Crossref] [PubMed]
- Nevéus T. Pathogenesis of enuresis: Towards a new understanding. Int J Urol 2017;24:174-82. [Crossref] [PubMed]
- Maternik M. Understanding of and misconceptions around monosymptomatic nocturnal enuresis: findings from patient and physician surveys. J Pediatr Urol 2019;15:37.e1-8. [Crossref] [PubMed]
- Butler RJ, Holland P. The three systems: a conceptual way of understanding nocturnal enuresis. Scand J Urol Nephrol 2000;34:270-7. [Crossref] [PubMed]
- Joinson C, Heron J, Butler R, et al. A United Kingdom population-based study of intellectual capacities in children with and without soiling, daytime wetting, and bed-wetting. Pediatrics 2007;120:e308-16. [Crossref] [PubMed]
- Perrin N, Sayer L, While A. The efficacy of alarm therapy versus desmopressin therapy in the treatment of primary mono-symptomatic nocturnal enuresis: a systematic review. Prim Health Care Res Dev 2015;16:21-31. [Crossref] [PubMed]
- Naitoh Y, Kawauchi A, Yamao Y, et al. Combination therapy with alarm and drugs for monosymptomatic nocturnal enuresis not superior to alarm monotherapy. Urology 2005;66:632-5. [Crossref] [PubMed]
- Claudius G, Chhatwal J, Sohi I. Efficacy of alarm intervention in primary monosymptomatic nocturnal enuresis in children. Int J Contemp Pediatr 2016;3:621-4. [Crossref]
- Glazener CM, Evans JH, Peto RE. Alarm interventions for nocturnal enuresis in children. Cochrane Database Syst Rev 2005;CD002911. [Crossref] [PubMed]
- Bradbury MG, Meadow SR. Combined treatment with enuresis alarm and desmopressin for nocturnal enuresis. Acta Paediatr 1995;84:1014-8. [Crossref] [PubMed]
- Meadow SR. Buzzer ulcers. In: Kolvin I, MacKeith RC, Meadow SR. editors. Bladder control and enuresis. London: William Heinemann Medical Books; 1973:220-2.
- Hanks JW, Venters WJ. Nickel allergy from a bed-wetting alarm confused with herpes genitalis and child abuse. Pediatrics 1992;90:458-60. [Crossref] [PubMed]
- Schmitt BD. Nocturnal enuresis. Pediatr Rev 1997;18:183-90; quiz 91. [Crossref] [PubMed]
- Caldwell PH, Codarini M, Stewart F, et al. Alarm interventions for nocturnal enuresis in children. Cochrane Database Syst Rev 2020;5:CD002911. [PubMed]
- Neveus T, Eggert P, Evans J, et al. Evaluation of and treatment for monosymptomatic enuresis: a standardization document from the International Children's Continence Society. J Urol 2010;183:441-7. [Crossref] [PubMed]
- Kim JM, Park JW, Lee CS. Evaluation of nocturnal bladder capacity and nocturnal urine volume in nocturnal enuresis. J Pediatr Urol 2014;10:559-63. [Crossref] [PubMed]
- Hunsballe JM. Increased delta component in computerized sleep electroencephalographic analysis suggests abnormally deep sleep in primary monosymptomatic nocturnal enuresis. Scand J Urol Nephrol 2000;34:294-302. [Crossref] [PubMed]
- Van Herzeele C, Evans J, Eggert P, et al. Predictive parameters of response to desmopressin in primary nocturnal enuresis. J Pediatr Urol 2015;11:200.e1-8. [Crossref] [PubMed]
- Glazener CM, Evans JH. Desmopressin for nocturnal enuresis in children. Cochrane Database Syst Rev 2002;CD002112. [PubMed]
- Marschall-Kehrel D, Harms TWEnuresis Algorithm of Marschall Survey Group. Structured desmopressin withdrawal improves response and treatment outcome for monosymptomatic enuretic children. J Urol 2009;182:2022-6. [Crossref] [PubMed]
- Chua ME, Silangcruz JM, Chang SJ, et al. Desmopressin Withdrawal Strategy for Pediatric Enuresis: A Meta-analysis. Pediatrics 2016;138:e20160495. [Crossref] [PubMed]
- Läckgren G, Nevéus T, Stenberg A. Diurnal plasma vasopressin and urinary output in adolescents with monosymptomatic nocturnal enuresis. Acta Paediatr 1997;86:385-90. [Crossref] [PubMed]
- Rushton HG, Belman AB, Zaontz MR, et al. The influence of small functional bladder capacity and other predictors on the response to desmopressin in the management of monosymptomatic nocturnal enuresis. J Urol 1996;156:651-5. [Crossref] [PubMed]
- Hjälmås K, Hanson E, Hellström AL, et al. Long-term treatment with desmopressin in children with primary monosymptomatic nocturnal enuresis: an open multicentre study. Swedish Enuresis Trial (SWEET) Group. Br J Urol 1998;82:704-9. [Crossref] [PubMed]