Two distinct growth patterns of preterm infants from birth to 18 months of corrected gestational age: a retrospective cohort study
Highlight box
Key findings
• Two distinct growth patterns (40% lower and 60% higher) were identified in preterm infants up to 18 months corrected age.
• Small for gestational age [relative risk (RR) =2.23, 95% confidence interval (CI): 1.40–3.57], maternal hypertensive disorders of pregnancy (HDOP) (RR =1.84, 95% CI: 1.01–3.36), and twin pregnancy (RR =1.74, 95% CI: 1.00–3.03) are independent risk factors for lower growth pattern.
What is known and what is new?
• Preterm infants are at risk of inadequate growth; maternal and infant factors may influence growth trajectories.
• This study applied group-based multi-trajectory modeling to simultaneously model weight, length, and head circumference, identifying two growth patterns. It further identified lower birthweight, HDOP, and twin pregnancy, as independent risk factors for poorer growth trajectories.
What is the implication, and what should change now?
• A substantial proportion of preterm infants remains at risk of lower growth pattern after discharge. Intensive post-discharge follow-up and personalized nutritional interventions are needed for preterm infants with low birthweight, those exposed to maternal HDOP, or those from twin pregnancies.
Introduction
Preterm birth [<37 weeks of gestational age (GA)] remains a significant global health challenge. An estimated 13.4 million newborns (9.9% of all births) were born preterm globally in 2020, with China ranking fourth and accounting for over 750,000 cases (1). A nationwide observational study revealed an annual increase of 1.3% in the preterm rate, from 5.9% in 2012 to 6.4% in 2018 in China (2).
Advances in perinatal and neonatal care have improved the survival of preterm infants (3). However, these survivors still suffer from numerous adverse outcomes, including cardiovascular and metabolic disorders (4,5), neurodevelopmental delays (6), and respiratory complications (7). Among these, impaired catch-up growth is a pressing concern (8,9), which is usually evaluated by weight and length (10). Most studies on preterm growth trajectories stratified the study population by predefined subgroups [e.g., GA (11) or sex (10)], analyzing either relative values [e.g., World Health Organization (WHO) Z-scores for weight and length] or absolute measurements (e.g., weight in kilograms and length in centimeters). Only one study used the group-based trajectory modeling (GBTM) method to explore weight, length, and body mass index (BMI) trajectories without predefined subgroups. However, that study focused on term babies (GA: 37–41 weeks), identifying three distinct physical growth trajectories (i.e., low-stable, moderate-stable, and high-increasing) (12). Furthermore, group-based multi-trajectory modeling (GBMTM) identifies potential populations of individuals that simultaneously follow similar trajectories across two or more distinct but related indicators (13). Some studies have successfully applied this method to trajectories in blood pressure and heart rate (14). Similarly, weight, length, and head circumference are three closely related core indicators that are commonly monitored in children’s growth and development assessments. However, to date, no such studies have been conducted in preterm populations. We propose that multi-trajectory groups of these growth indicators may provide more useful information for pediatric health management and growth intervention strategies.
Currently, insufficient data on head circumference limits researchers’ ability to assess growth restriction due to potential neurodevelopmental problems. Moreover, data on maternal disease history are unavailable in many studies (15,16), restricting investigations into maternal risk factors for preterm growth impairment. Therefore, we conducted an exploratory study to investigate the growth trajectories of preterm infants by using integrated measurements of weight, length, and head circumference, and to assess which risk factors affect their developmental status. We present this article in accordance with the STROBE reporting checklist (available at https://tp.amegroups.com/article/view/10.21037/tp-2026-1-0011/rc).
Methods
Study design
This was a retrospective cohort study incorporating repeated measurements of infants’ growth parameters from birth up to 18 months of corrected gestational age (CGA). This study was conducted at Shanghai General Hospital, Affiliated to Shanghai Jiao Tong University School of Medicine, from January 1, 2021, to June 30, 2025. This study was conducted in accordance with the Declaration of Helsinki and its subsequent amendments. The study was approved by the ethics committee of the Shanghai General Hospital affiliated to Shanghai Jiao Tong University School of Medicine (No. 2025-495). Informed consent was waived by the ethics committee because of the retrospective nature of this study.
Study population
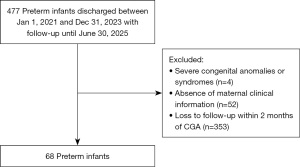
The initial cohort comprised all preterm infants (GA <37 weeks, n=477) discharged from Department of Neonatology between January 1, 2021, and December 31, 2023, with follow-up data collected until June 30, 2025. Exclusion criteria included infants with severe congenital anomalies or syndromes (n=4), those without maternal information (n=52), and those loss to follow-up within 2 months of CGA (n=353). The final analysis included 68 infants who had attended at least two outpatient follow-up visits (Figure 1).
Exposures
GA was determined by obstetric ultrasound examination. Preterm infants were categorized according to WHO subcategories: late preterm (34 to <37 weeks), moderately preterm (32 to <34 weeks), and very preterm (28 to <32 weeks). Baseline maternal and infant characteristics were extracted from electronic hospitalization records. Maternal variables included age, history of gestational diseases (diabetes mellitus, thyroid disease, hypertensive disorders), twin pregnancy, and conception by in-vitro fertilization (IVF). Infant variables included sex, birthweight, and GA.
Outcomes
After discharge, parents were scheduled for outpatient clinic visits every month. At each visit, physical examinations (i.e., weight, length, and head circumference) were performed by trained clinical staff. All growth parameters were converted to the Z-score [weight-for-age (WAZ), length-for-age (LAZ), head circumference-for age (HCAZ), and weight-for-length (WLZ)] using the λ-µ-σ method (LMS) based on the WHO Child Growth Standard (17). We used the infant’s exact CGA to derive the Z-scores. Infants were followed from discharge until 18 months of CGA. Given potential variability in visit time, data were collected for time points from 1 to 18 months of CGA, with a tolerance of ±2 weeks per time point. If multiple measurements were available for a single time point, the mean value was computed. The primary outcome was the trajectory of WAZ, LAZ, HCAZ, and WLZ during follow-up.
Statistical analysis
Identification of preterm growth patterns
Since changes in one dimension may influence changes in others, GBMTM was employed to identify distinct heterogeneous trajectories for weight, length, and head circumference simultaneously. This method uses maximum likelihood to identify latent subgroups of individuals with similar trajectory. The optimal number of trajectory groups and the degree of polynomials in each trajectory group were selected on the basis of a combination of rigorous criteria (18), including the Bayesian Information Criterion (closer to 0), average posterior probability (greater than 0.7), odds of correct classification (greater than 5), and the lowest sample size of trajectories (greater than 5%). All trajectory models consisting of 2–4 trajectory groups were fitted until the best model was obtained.
Baseline characteristics of different preterm growth patterns
Baseline characteristics were compared across the identified trajectory groups. Continuous variables with a normal distribution are presented as mean [standard deviation (SD)] and were compared using Independent Samples t-tests. For continuous variables with a non-normal distribution, data are presented as median [interquartile range (IQR)] and group differences were assessed using the Mann-Whitney U test. Categorical variables are presented as numbers (proportions) and were compared using Pearson’s Chi-squared test or Fisher’s exact test, as appropriate.
The association between baseline characteristics and preterm growth trajectories
Candidate variables with a P value <0.1 on univariate analysis were included into multivariable regression models. Poisson regression models with robust variance estimators (19) were used to calculate the adjusted relative risk (RR) and 95% confidence interval (CI) of baseline characteristics with infant growth trajectories. Simulation studies have demonstrated that this approach remains reliable with small sample sizes (19).
Sensitivity analyses
Two sensitivity analyses were performed: (I) additionally adjusting for maternal pre-pregnancy BMI, and (II) excluding participants with less than 6 months of follow-up time.
All analyses were performed using SAS 9.4 (SAS Institute Inc., Cary, NC, USA), with GBMTM conducted using the PROC TRAJ plug-in. All statistical tests were 2-sided with a 5% significance level.
Results
We found no significant differences in sex, GA (week), or birthweight categories, suggesting that the attrition was possibly random (Table 1). The median follow-up time was 11.50 (IQR, 8.75–15.00) months of CGA. The two-trajectory model demonstrated the best. As shown in Table 2, the average posterior probability exceeded 0.7 for both groups (Group1: 98.52%; Group 2: 99.31%). The odds of correct classification were high for both groups (95.56 and 100.23, respectively). All groups comprised a substantial proportion of the cohort, with Group 1 representing 41.18% and Group 2 representing 58.82% of infants. Group 1 exhibited lower growth, with a mean WAZ, LAZ, HCAZ, and WLZ of −0.5, −1.0, −1.0, and 0.0, respectively. In contrast, Group 2 was characterized by a WAZ, LAZ, HCAZ, and WLZ of nearly +1.0 (Figure 2).
Table 1
| Variables | Included population (n=68) | Excluded population (n=409) | Z/χ2 | P |
|---|---|---|---|---|
| Boy | 48 (70.59) | 236 (57.70) | 3.50 | 0.06 |
| Gestational age (week) | 34.43 (34.00, 35.21) | 34.29 (33.00, 35.43) | 1.54 | 0.12 |
| Birthweight categories | – | 0.19 | ||
| SGA | 12 (17.65) | 53 (12.96) | ||
| AGA | 55 (80.88) | 331 (80.93) | ||
| LGA | 1 (1.47) | 25 (6.11) |
Data are presented as n (%) or median (interquartile range). AGA, appropriate for gestational age; LGA, large for gestational age; SGA, small for gestational age.
Table 2
| Groups | AvePP (%) | OCC | Pj (%) | πj (%) |
|---|---|---|---|---|
| Lower growth pattern | 98.515 | 95.562 | 41.176 | 40.969 |
| Higher growth pattern | 99.312 | 100.227 | 58.824 | 59.031 |
Pj, posterior probability of group membership; πj, probability of group membership; AvePP, average posterior probability; OCC, odds of correct classification.

Table 3 showed the basic characteristics of participants by group. No significant differences were observed in the following variables: maternal age at expected date of delivery, history of gestational diabetes mellitus, history of gestational thyroid diseases, infant sex, and infant GA at birth. Compared with mothers in the higher growth pattern group, those in the lower growth pattern group had a higher proportion of hypertensive disorders of pregnancy (HDOP) (39.29% vs. 20.00%) and twin pregnancy (46.43% vs. 25.00%), though some of these differences were only marginally statistically significant. Infants in the lower growth pattern group also had significantly lower birthweight Z-score (P<0.001).
Table 3
| Variables | Total (n=68) | Lower growth pattern (n=28) | Higher growth pattern (n=40) | Z/c2 | P |
|---|---|---|---|---|---|
| Maternal factors | |||||
| Age (years) | 32.00 (28.50, 34.00) | 30.50 (27.00, 34.00) | 32.00 (29.00, 34.00) | 0.61 | 0.54 |
| History of gestational diseases | |||||
| Diabetes mellitus | 21 (30.88) | 9 (32.14) | 12 (30.00) | 0.04 | 0.85 |
| Thyroid disease | 22 (32.35) | 9 (32.14) | 13 (32.50) | 0.001 | 0.98 |
| HDOP | 19 (27.94) | 11 (39.29) | 8 (20.00) | 3.04 | 0.08 |
| Twin pregnancy | 23 (33.82) | 13 (46.43) | 10 (25.00) | 3.38 | 0.07 |
| IVF | 12 (17.65) | 7 (25.00) | 5 (12.50) | 1.77 | 0.18 |
| Infant factors | |||||
| Boy | 48 (70.59) | 18 (64.29) | 30 (75.00) | 0.91 | 0.34 |
| BWZ | −0.61 (−1.10, −0.16) | −0.98 (−1.58, −0.58) | −0.33 (−0.78, 0.17) | 3.85 | <0.001 |
| Birthweight categories | – | 0.002 | |||
| SGA | 12 (17.65) | 10 (35.71) | 2 (5.00) | ||
| AGA | 55 (80.88) | 18 (64.29) | 37 (92.50) | ||
| LGA | 1 (1.47) | 0 | 1 (2.50) | ||
| Preterm categories | – | 0.61 | |||
| Late preterm (34–<37 weeks) | 52 (76.47) | 23 (82.14) | 29 (72.50) | ||
| Moderately preterm (32 to <34 weeks) | 14 (20.59) | 4 (14.29) | 10 (25.00) | ||
| Very preterm (28–<32 weeks) | 2 (2.94) | 1 (3.57) | 1 (2.50) |
Data are presented as n (%) or median (interquartile range). AGA, appropriate for gestational age; HDOP, hypertensive disorders of pregnancy; IVF, in-vitro fertilization; LGA, large for gestational age; SGA, small for gestational age.
Table 4 presented the results of multivariable Poisson regression model. For SGA infants, the risk of lower growth was increased by 123% (RR =2.23, 95% CI: 1.40–3.57). Also, infants born to mothers with HDOP had a 1.84-fold higher risk of lower growth compared with those born to mothers without HDOP (RR =1.84, 95% CI: 1.01–3.36). Infants from twin pregnancies had a 1.74-fold higher risk than those from singleton pregnancies (RR =1.74, 95% CI: 1.00–3.03). After excluding infants with less than 6 months of follow-up, we observed similar results, though the effect sizes were slightly attenuated.
Table 4
| Variables | Model 1 | Model 2 | Model 3 | |||||
|---|---|---|---|---|---|---|---|---|
| RR (95% CI) | P | RR (95% CI) | P | RR (95% CI) | P | |||
| SGA, yes vs. no | 2.16 (1.35–3.44) | 0.001 | 2.23 (1.40–3.57) | <0.001 | 2.11 (1.27–3.50) | 0.004 | ||
| HDOP, yes vs. no | 1.52 (0.87–2.66) | 0.14 | 1.84 (1.01–3.36) | 0.05 | 1.54 (0.86–2.75) | 0.14 | ||
| Twin pregnancy, yes vs. no | 1.78 (1.04–3.05) | 0.04 | 1.74 (1.00–3.03) | 0.05 | 1.60 (0.91–2.82) | 0.10 | ||
Model 1 was the main analysis. Model 2 was additionally adjusted for maternal overweight/obesity before pregnancy. Model 3 excluded 7 infants with less than 6 months of follow-up. CI, confidence interval; HDOP, hypertensive disease of pregnancy; RR, relative ratio; SGA, small for gestational age.
Discussion
This study identified two distinct growth patterns among preterm infants during the first 1.5 years of life using GBMTM. Approximately 40% of the cohort exhibited a pattern of lower growth, while the remaining 60% demonstrated higher growth, with their Z-scores approaching +1.0. Multivariable analyses identified low birthweight, HDOP, and twin pregnancy as significant risk factors for lower growth pattern.
Current research findings on the preterm infant population are inconsistent. A study conducted in Indonesia involving 306 preterm infants found that WAZ decreased during the first three months, followed by a slow increase until 2 years of age (11). Their study population was recruited from the Neonatal Intensive Care Unit, suggesting the possible presence of underlying diseases, which may explain the difference from our findings. In contrast, a large-scale community-based study in China (n=10,624) reported steady increases in both height and weight among infants, with overall higher measurements in males than in females (20). In their subgroup analyses, WAZ and LAZ ranges for infants of large-for-gestational-age, appropriate-for-gestational-age, and small-for-gestational-age were +1.0 to +2.0, 0.0 to +1.0, and −1.0 to -0.5, respectively. This trend is similar to the patterns observed in our curves. Notably, none of these studies included head circumference measurements. Given that head circumference is closely linked to neurodevelopment (21), the inclusion of this parameter provides a proxy measure relevant to the assessment of developmental status.
In terms of the association of maternal HDOP with infant inadequate growth, Kiy et al. reported that HDOP was not significantly associated with inadequate growth at 2 years of age (WAZ and LAZ less than 3rd percentile on the WHO curve) after adjusting for potential confounders among preterm low birthweight infants (22). Another large-scale study including all neonates (n=31,734) found that gestational hypertension was negatively correlated with WAZ and LAZ from birth to 36 months using generalized estimated equation analysis (23). However, these associations did not show statistical significance after further adjustment for birthweight. Nevertheless, our results were inconsistent with these studies. The discrepancies across studies may be due to different definition of variables (e.g., inadequate growth) and different GA of study population. In a cohort of 395 infants born before 32 weeks of GA, Jain et al. reported that HDOP was negatively associated with subsequent neurodevelopment, with birthweight acting as a significant mediator (24). Our study did not confirm the mediating role of birthweight, only suggesting a direct effect between HDOP and head circumference; this discrepancy might be related to our smaller sample size and warrants further investigation.
Twins tend to exhibit slower growth than singletons, and the underlying mechanisms may include intrinsically pathological processes and a form of physiological adaptation (25). In our study of preterm infants, we observed a similar association between twin pregnancy and relatively lower growth trajectory. Furthermore, twin pregnancies with growth discordance contribute to excess risk of prematurity, as well as neonatal morbidity (26,27). In this study, we also found that the larger twin in pairs with growth discordance was more likely to be classified into the higher growth group (data not shown). However, due to the small sample size, this finding remains to be verified in larger-scale studies.
There are several strengths of this study. First, it incorporated four growth indicators (WAZ, LAZ, head circumference, and WLZ), providing a more comprehensive assessment of growth status. Second, we had information on many possible confounders, including both mother (e.g., HDOP and other metabolic diseases) and offspring (e.g., birth weight and twin status) variables. Several limitations should be acknowledged. First, the sample size was relatively small, which may reduce the statistical power of the study. Accordingly, the results are presented as preliminary and hypothesis-generating. Second, the follow-up duration was insufficient to capture long-term growth trajectories such as preschool age and school age. Third, critical information was lacking, including infant comorbidities, infant feeding, the severity of maternal HDOP and details of maternal medication use during pregnancy. Finally, due to the missing rate of preterm infants, selection bias cannot be completely ruled out.
Conclusions
Our findings highlight that a substantial proportion (~40%) of preterm infants are at risk of lower growth pattern after discharge. Those with low birthweight, whose mothers had HDOP, and who were born from twin pregnancy need more intensive follow-up and personalized nutritional interventions. Future research could focus on multicenter studies with longer follow-up to validate these patterns and investigate their associations with all the health-related outcomes. Furthermore, exploring the underlying mechanisms, including prenatal and postnatal factors, is crucial for developing precision prevention strategies.
Acknowledgments
None.
Footnote
Reporting Checklist: The authors have completed the STROBE reporting checklist. Available at https://tp.amegroups.com/article/view/10.21037/tp-2026-1-0011/rc
Data Sharing Statement: Available at https://tp.amegroups.com/article/view/10.21037/tp-2026-1-0011/dss
Peer Review File: Available at https://tp.amegroups.com/article/view/10.21037/tp-2026-1-0011/prf
Funding: This work was supported by
Conflicts of Interest: All authors have completed the ICMJE uniform disclosure form (available at https://tp.amegroups.com/article/view/10.21037/tp-2026-1-0011/coif). The authors have no conflicts of interest to declare.
Ethical Statement: The authors are accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved. This study was conducted in accordance with the Declaration of Helsinki and its subsequent amendments. The study was approved by the ethics committee of the Shanghai General Hospital affiliated to Shanghai Jiao Tong University School of Medicine (No. 2025-495). Informed consent was waived by the ethics committee because of the retrospective nature of this study.
Open Access Statement: This is an Open Access article distributed in accordance with the Creative Commons Attribution-NonCommercial-NoDerivs 4.0 International License (CC BY-NC-ND 4.0), which permits the non-commercial replication and distribution of the article with the strict proviso that no changes or edits are made and the original work is properly cited (including links to both the formal publication through the relevant DOI and the license). See: https://creativecommons.org/licenses/by-nc-nd/4.0/.
References
- Ohuma EO, Moller AB, Bradley E, et al. National, regional, and global estimates of preterm birth in 2020, with trends from 2010: a systematic analysis. Lancet 2023;402:1261-71. [Crossref] [PubMed]
- Deng K, Liang J, Mu Y, et al. Preterm births in China between 2012 and 2018: an observational study of more than 9 million women. Lancet Glob Health 2021;9:e1226-41. [Crossref] [PubMed]
- Zhu Z, Yuan L, Wang J, et al. Mortality and Morbidity of Infants Born Extremely Preterm at Tertiary Medical Centers in China From 2010 to 2019. JAMA Netw Open 2021;4:e219382. [Crossref] [PubMed]
- Makker K, Kuiper JR, Brady T, et al. Prematurity, Neonatal Complications, and the Development of Childhood Hypertension. JAMA Netw Open 2025;8:e2527431. [Crossref] [PubMed]
- Hofman PL, Regan F, Jackson WE, et al. Premature birth and later insulin resistance. N Engl J Med 2004;351:2179-86. [Crossref] [PubMed]
- Thalhammer M, Seidlitz J, Neubauer A, et al. Heterogeneous, temporally consistent, and plastic brain development after preterm birth. Nat Commun 2025;16:8269. [Crossref] [PubMed]
- van Hasselt TJ, Dorner RA, Katheria A, et al. Neonatal Morbidities and Hospitalization in the First 2 Years of Life Among Infants Born Very Preterm. JAMA Netw Open 2025;8:e2530123. [Crossref] [PubMed]
- Morniroli D, Tiraferri V, Maiocco G, et al. Beyond survival: the lasting effects of premature birth. Front Pediatr 2023;11:1213243. [Crossref] [PubMed]
- Bradley E, Blencowe H, Moller AB, et al. Born too soon: global epidemiology of preterm birth and drivers for change. Reprod Health 2025;22:105. [Crossref] [PubMed]
- Kang L, Wang H, He C, et al. Postnatal growth in preterm infants during the first year of life: A population-based cohort study in China. PLoS One 2019;14:e0213762. [Crossref] [PubMed]
- Rohsiswatmo R, Hikmahrachim HG, Nadobudskaya DU, et al. Growth trajectory at 24 months of preterm infants after discharge: a longitudinal study in Indonesia. Br J Nutr 2024;132:1300-7. [Crossref] [PubMed]
- Chen K, Song L, Liu B, et al. Low length/weight growth trajectories of early-term infants during the first year: evidence from a longitudinal study in China. BMJ Open 2022;12:e051436. [Crossref] [PubMed]
- Nagin DS, Odgers CL. Group-based trajectory modeling in clinical research. Annu Rev Clin Psychol 2010;6:109-38. [Crossref] [PubMed]
- Shen X, Li J, Yan H, et al. Combined blood pressure and heart rate trajectories are associated with prognosis in critically ill patients with acute aortic dissection: A group-based multi-trajectory analysis. Heliyon 2024;10:e29934. [Crossref] [PubMed]
- Matsumoto N, Kubo T, Nakamura K, et al. Trajectory of body mass index and height changes from childhood to adolescence: a nationwide birth cohort in Japan. Sci Rep 2021;11:23004. [Crossref] [PubMed]
- Matsunaga Y, Inoue H, Miyauchi Y, et al. Neurodevelopmental Changes and Postnatal Growth in the First 3 Years of Extremely Preterm Infants. Neonatology 2025;122:181-90. [PubMed]
- Cole TJ. The LMS method for constructing normalized growth standards. Eur J Clin Nutr 1990;44:45-60. [PubMed]
- Nagin DS. Group-based trajectory modeling: an overview. Ann Nutr Metab 2014;65:205-10. [Crossref] [PubMed]
- Zou G. A modified poisson regression approach to prospective studies with binary data. Am J Epidemiol 2004;159:702-6. [Crossref] [PubMed]
- Han J, Jiang Y, Huang J, et al. Postnatal growth of preterm infants during the first two years of life: catch-up growth accompanied by risk of overweight. Ital J Pediatr 2021;47:66. [Crossref] [PubMed]
- Mayrink MLS, Villela LD, Méio MDBB, et al. The trajectory of head circumference and neurodevelopment in very preterm newborns during the first two years of life: a cohort study. J Pediatr (Rio J) 2024;100:483-90. [Crossref] [PubMed]
- Kiy AM, Rugolo LM, Luca AK, et al. Growth of preterm low birth weight infants until 24 months corrected age: effect of maternal hypertension. J Pediatr (Rio J) 2015;91:256-62. [Crossref] [PubMed]
- Jiang W, Mo M, Si S, et al. Association of hypertensive disorders of pregnancy with infant growth in the first 36 months of life. Eur J Pediatr 2022;181:133-41. [Crossref] [PubMed]
- Jain S, Fu TT, Barnes-Davis ME, et al. Maternal Hypertension and Adverse Neurodevelopment in a Cohort of Preterm Infants. JAMA Netw Open 2025;8:e257788. [Crossref] [PubMed]
- Hiersch L, Barrett J, Fox NS, et al. Should twin-specific growth charts be used to assess fetal growth in twin pregnancies? Am J Obstet Gynecol 2022;227:10-28. [Crossref] [PubMed]
- Amaru RC, Bush MC, Berkowitz RL, et al. Is discordant growth in twins an independent risk factor for adverse neonatal outcome? Obstet Gynecol 2004;103:71-6. [Crossref] [PubMed]
- Prasad S, Ayhan I, Mohammed D, et al. Longitudinal twin growth discordance patterns and adverse perinatal outcomes. Am J Obstet Gynecol 2025;233:73.e1-73.e14. [Crossref] [PubMed]