Association between nadir albumin concentration and mortality in pediatric patients undergoing postcardiotomy extracorporeal membrane oxygenation
Highlight box
Key findings
• This retrospective study identified an association between lower nadir albumin concentration during pediatric postcardiotomy extracorporeal membrane oxygenation (ECMO) and an elevated risk of mortality at both 30 and 180 days, providing a potentially valuable biomarker for risk stratification in this population.
What is known and what is new?
• Lower albumin concentration is closely associated with adverse outcomes and increased mortality after cardiac surgery; however, its prognostic significance in pediatric patients receiving postcardiotomy ECMO remains insufficiently characterized.
• This study demonstrates, for the first time, an independent and linear inverse association between nadir albumin concentration during ECMO and both 30-day and 180-day mortality in pediatric postcardiotomy patients, identifying a clinically relevant threshold of approximately 31.4 g/L, which remains robust in subgroup and sensitivity analysis.
What is the implication, and what should change now?
• Serum albumin may serve as a readily available dynamic biomarker for risk stratification in pediatric postcardiotomy ECMO. Enhanced monitoring and individualized albumin-targeted management strategies may help optimize outcomes and reduce complications in this vulnerable population.
Introduction
Extracorporeal membrane oxygenation (ECMO) is an advanced life support technique that can be categorized into veno-venous ECMO (VV-ECMO) and veno-arterial ECMO (VA-ECMO), each serving distinct clinical indications (1). Statistical data indicate that from 2009 to 2022, the Extracorporeal Life Support Organization (ELSO) registered over 150,000 ECMO cases, with approximately 70% involving adult patients and 30% involving pediatric patients (2). Notably, nearly 42% of these cases were implemented for post-cardiac surgery support. Advances in the bio-compatibility of circuit oxygenators and improvements in management techniques have led to a significant increase in ECMO survival rates. Nonetheless, the management of patients on ECMO remains challenging due to complications such as bleeding and thrombosis, as well as the influence of underlying medical conditions, contributing to a persistently high ECMO-related mortality (3).
Albumin holds significant clinical importance in this context. As the major plasma protein synthesized by the liver, albumin is essential for maintaining colloid osmotic pressure, facilitating drug transport, exerting anti-inflammatory and antioxidant effects, and providing endothelial protection (4). Extensive research has indicated that reduced albumin concentrations are independently correlated with complications such as acute kidney injury (AKI) and infection, as well as increased mortality (5-8). This makes it an important and easily accessible dynamic biomarker for perioperative risk stratification.
However, ECMO may profoundly disrupt albumin homeostasis. During ECMO, substantial volumes of crystalloid are typically administered to ensure adequate venous drainage and maintain intravascular volume (9). Furthermore, the interaction between blood and the ECMO circuit can promote the release of inflammatory cytokines, such as interleukin-6 (IL-6) and tumor necrosis factor-alpha (TNF-α), which may contribute to the degradation of endothelial tight junction proteins and increased vascular permeability, thus leading to capillary leakage (10-14). These factors collectively lead to substantial and dynamic fluctuations in albumin concentration during ECMO support. In pediatric patients, the immature hepatic synthetic capacity and rapid metabolic rates may further exacerbate the decline in albumin concentration during ECMO. Given the dynamic nature of albumin changes, the nadir albumin concentration during ECMO may better reflect the period of greatest physiological vulnerability than baseline measurements. Previous studies have also suggested that nadir albumin is more strongly associated with adverse clinical outcomes than baseline values (15). Nevertheless, there is a notable absence of research investigating the relationship between nadir albumin concentration and adverse outcomes in pediatric patients undergoing ECMO.
Consequently, this study aims to elucidate the association between nadir albumin concentration and mortality in pediatric postcardiotomy ECMO population. This research seeks to provide a foundation for developing pediatric-specific albumin intervention strategies. We present this article in accordance with the STROBE reporting checklist (available at https://tp.amegroups.com/article/view/10.21037/tp-2026-1-0008/rc).
Methods
Trial design and population
This single-center retrospective observational study included pediatric patients (aged <18 years) who required VA-ECMO following cardiopulmonary bypass (CPB) without remaining surgical deformities in Fuwai Hospital between January 2010 and June 2020. Four children were excluded because the duration of ECMO support was less than 24 hours, resulting in a final cohort of 96 patients. The study was conducted in accordance with the Declaration of Helsinki and its subsequent amendments. The study was approved by the Ethics Committee of Fuwai Hospital (approval No. 2020-1346). Given the retrospective nature of the research, the requirement for informed consent was waived.
ECMO management
The composition, management strategies, and weaning criteria of ECMO at our center have been described in detail previously (16). Briefly, the ECMO system consists of a centrifugal pump, a membrane oxygenator, and polyvinyl chloride tubing. After cannulation, appropriate blood flow and arterial pressure are maintained, and ventilator settings are adjusted to ensure adequate gas exchange. Systemic anticoagulation is achieved with unfractionated heparin (UFH), titrated to maintain target activated clotting time (ACT) levels. Routine surveillance of the ECMO circuit is performed to detect thrombosis or hemolysis, and circuit replacement is undertaken when necessary. During ECMO, albumin supplementation followed a standardized institutional protocol: albumin supplementation was administered as 4% albumin solution, with the dose tailored according to fluid management needs, and a maximum daily dose of 1–2 g/kg. In addition, continuous renal replacement therapy (CRRT) was initiated during ECMO support when clinically indicated and at the discretion of the treating physicians. Specifically, CRRT was considered when patients developed stage ≥2 AKI, fluid overload greater than 20%, life-threatening or refractory electrolyte disturbances, toxic or metabolic derangements, or other clinical conditions requiring renal replacement therapy. CRRT was performed using a circuit connected in parallel with the ECMO circuit. Anticoagulation and circuit function were closely monitored throughout the treatment. In the event of circuit thrombosis, the circuit was promptly replaced to ensure treatment continuity and safety (17).
ECMO weaning is considered when hemodynamics and end-organ function show improvement. Flow is gradually reduced, and decannulation is attempted when flow decreases to <0.3 L/min or <20–30 mL/kg/min. If mean arterial pressure decreases by ≥30% during the trial, the weaning attempt is aborted.
Variables
Clinical data were extracted from electronic medical records and telephone follow-up. Collected variables included demographic characteristics (such as age, sex, weight), pre-ECMO laboratory parameters [such as white blood cell count, hemoglobin, platelet count, serum albumin concentration, serum creatinine, lactate, aspartate aminotransferase (AST) and alanine aminotransferase (ALT)], as well as detailed ECMO parameters (such as pump speed, flow rate). Transfusion details during ECMO were also recorded, including volumes of packed red blood cells (RBC), platelets, and fresh frozen plasma administered. Clinical outcomes included the requirement for CRRT, the occurrence of major bleeding events, hemolysis, and thrombosis, as well as all-cause mortality at both 30 and 180 days following ECMO initiation. Surgical complexity was categorized using the Society of Thoracic Surgeons and European Association for Cardiothoracic Surgery Congenital Heart Surgery (STAT) Mortality Categories.
Disease definition
In this study, all events were predefined according to established pediatric critical care standards. Mortality was defined as the death of a pediatric patient since the initiation of ECMO support due to any cause, including mortality at 30 and 180 days. During ECMO support, hemolysis was identified when free hemoglobin concentration surpassed 50 mg/dL. The definition of major bleeding included its presence at surgical or intubation sites, gastrointestinal, pulmonary, intracranial, or any other clinically significant bleeding. AKI was classified according to Kidney Disease: Improving Global Outcomes (KDIGO) criteria (18). Weaning from ECMO was considered successful if the patient survived for over 24 hours post-weaning. Liver injury was defined as a peak ALT and AST concentration during ECMO exceeding twice the upper limit of normal; thrombocytopenia was defined as a minimum platelet count during ECMO below 50×109/L. The STAT scores were categorized based on previously published literature, in which STAT 1 refers to the lowest mortality risk and STAT 5 refers to the highest mortality risk (19). The vasoactive inotrope score (VIS) was calculated using validated formulas employed in prior studies (20).
Statistical analysis
All statistical analyses were performed using R software (version 4.5.1). Descriptive statistics were first generated to summarize baseline characteristics. Continuous variables were presented as medians with interquartile ranges (IQRs) and compared between groups using the Kruskal-Wallis test. Categorical variables were reported as counts and percentages and compared using the chi-square test. To examine the association between albumin concentration and mortality, albumin was initially analyzed as a continuous variable using univariable Cox proportional hazards regression. Considering potential confounding, multivariable Cox proportional hazards models were subsequently constructed using a stepwise adjustment strategy. Specifically, model 1 was adjusted for baseline demographic and pre-ECMO clinical characteristics, including age, sex, pre-ECMO infection status, pre-ECMO VIS, and CPB duration. Based on model 1, model 2 was further adjusted for pre-ECMO laboratory parameters, including white blood cell count, hemoglobin, AST, creatinine, and baseline albumin concentration. Model 3 additionally incorporated parameters during ECMO, specifically pump speed and flow rate, C-reactive protein (CRP) levels prior to the occurrence of nadir albumin, cumulative fluid overload before nadir albumin, and albumin supplementation dose prior to nadir albumin. Proportional hazards assumptions were verified for all models and confirmed to be satisfied (Table S1). Multicollinearity was evaluated, with all variance inflation factors (VIFs) <5, indicating no significant collinearity among covariates (Tables S2,S3). Restricted cubic spline (RCS) curves were then plotted, and patients were re-grouped based on the inflection points of the RCS curves. Patients below the inflection point were classified as the low albumin group, and those above the inflection point were classified as the high albumin group. Subsequently, Kaplan-Meier curves were plotted for the two groups. Additionally, exploratory analyses were performed to assess the relationship between albumin concentration and ECMO-related complications using logistic regression, aiming to further characterize potential factors associated with lower nadir albumin concentration and mortality. For complications significantly associated with albumin concentration, additional multivariable Cox proportional hazard model was conducted to assess their associations with 30-day and 180-day mortality. Subgroup analyses were performed according to age, body weight, and STAT category to assess the consistency of the association between albumin and mortality across clinically relevant strata. Finally, two sensitivity analyses were conducted to assess the robustness of the findings. First, to minimize potential time-related bias and reverse causation, patients whose nadir albumin occurred beyond 48 hours after ECMO initiation were excluded. Second, to evaluate the potential influence of albumin supplementation, patients who did not receive albumin supplementation were excluded in an additional sensitivity analysis. The packages used throughout the process included “rms”, “ggplot2”, “survival”, “broom”, “tableone”, “flextable”, “officer”, “tidyverse”, “car” and “nortest”.
Results
Characteristics
A total of 96 pediatric patients who received VA-ECMO support between January 2010 and June 2020 were included in this study. The median age at ECMO initiation was 12.42 months (IQR: 6.42–40.06 months), with 64% being male. Among the cohort, 4 patients (4.2%) were neonates, while 43 patients (44.8%) were infants. The median body weight was 8.50 kg (IQR: 6.00–12.62 kg), with 60 patients (62.5%) weighing less than 10 kg. As for surgical complexity, 49 patients (51.0%) were classified as STAT category 4–5. The median duration of ECMO support was 123 hours (IQR: 91.50–167.00 hours), and the median intensive care unit (ICU) length of stay was 28 days (IQR: 12.75–48.00 days). Overall, 69.8% of patients were successfully weaned from ECMO, whereas the 30-day and 180-day all-cause mortality rates were 37.5% and 52.1%, respectively. Before ECMO initiation, the median albumin concentration was 42.30 g/L (IQR: 39.08–44.12 g/L). During ECMO support, the nadir albumin concentration typically occurred within the first 48 hours after ECMO initiation, with a median value of 32.45 g/L (IQR: 27.70–35.42 g/L). Across the entire ECMO course, the overall median nadir albumin concentration was 31.45 g/L (IQR: 27.28–34.50 g/L), and these nadir values generally preceded the occurrence of major adverse events (Table 1).
Table 1
| Variables | Survivors (n=46) | Non-survivors (n=50) | P |
|---|---|---|---|
| Age (months) | 14.57 [6.66, 42.78] | 10.12 [5.78, 37.27] | 0.44 |
| Gender | |||
| Male | 30 (65.2) | 31 (62.0) | 0.91 |
| Female | 16 (34.8) | 19 (38.0) | |
| Weight (kg) | 9.55 [6.20, 13.73] | 8.30 [6.00, 11.47] | 0.32 |
| Indication | 0.12 | ||
| LCOS | 7 (15.2) | 6 (12.0) | |
| Failure to wean from CPB | 27 (58.7) | 19 (38.0) | |
| ECPR | 8 (17.4) | 17 (34.0) | |
| Respiratory | 4 (8.7) | 8 (16.0) | |
| Left heart unloading | 34 (73.9) | 35 (70.0) | 0.84 |
| Laboratory parameters | |||
| WBC count (×109/L) | 8.70 [7.32, 11.32] | 8.91 [6.56, 14.64] | 0.61 |
| Hemoglobin (g/L) | 141.50 [121.00, 164.00] | 137.50 [121.25, 155.00] | 0.85 |
| Platelet count (×109/L) | 265.50 [187.50, 323.00] | 273.00 [205.75, 332.25] | 0.75 |
| ALT (U/L) | 17.00 [13.00, 27.00] | 20.50 [15.25, 30.00] | 0.33 |
| AST (U/L) | 40.00 [32.25, 48.75] | 38.00 [31.25, 49.75] | 0.92 |
| Albumin (g/L) | 42.35 [38.18, 44.05] | 41.80 [39.50, 44.05] | 0.90 |
| Creatinine (μmol/L) | 29.00 [23.84, 35.75] | 30.98 [21.80, 39.65] | 0.68 |
| Lactate (mmol/L) | 6.70 [4.82, 9.00] | 8.85 [5.62, 13.92] | 0.02 |
| Nadir albumin during ECMO (g/L) | 32.55 [28.90, 36.62] | 29.65 [24.75, 32.92] | 0.002 |
| Nadir albumin within 48 h (g/L) | 32.55 [30.82, 37.53] | 30.40 [25.55, 34.45] | 0.02 |
| Infection before ECMO | 4 (8.7) | 5 (10.0) | >0.99 |
| STAT | 0.16 | ||
| 1 | 1 (2.2) | 2 (4.0) | |
| 2 | 15 (32.6) | 13 (26.0) | |
| 3 | 9 (19.6) | 7 (14.0) | |
| 4 | 21 (45.7) | 22 (44.0) | |
| 5 | 0 (0.0) | 6 (12.0) | |
| VIS | 22.00 [16.00, 42.25] | 27.50 [18.00, 46.75] | 0.28 |
| CPB duration (min) | 272.50 [162.25, 370.25] | 246.00 [145.25, 326.50] | 0.55 |
| Clamp time (min) | 118.00 [86.00, 154.25] | 125.00 [78.25, 151.00] | 0.97 |
| Management | |||
| Median flow rate (mL/min) | 665.00 [527.50, 800.00] | 600.00 [505.00, 915.00] | 0.89 |
| Median pump speed (rpm) | 2,686.00 [2,535.75, 2,828.75] | 2,750.00 [2,570.00, 2,861.25] | 0.41 |
| Transfusion | |||
| RBC transfusion (mL/kg/d) | 23.59 [15.70, 33.98] | 28.54 [20.11, 38.31] | 0.08 |
| Platelet transfusion (mL/kg/d) | 7.37 [2.32, 11.55] | 8.07 [2.83, 19.09] | 0.27 |
| Plasma transfusion (mL/kg/d) | 7.97 [6.02, 16.08] | 11.01 [6.41, 18.10] | 0.29 |
| Albumin infusion (mL/kg/d) | 0.52 [0.28, 1.16] | 0.66 [0.38, 1.31] | 0.27 |
| Cumulative fluid overload (mL) | −35.15 [−177.50, 137.32] | −26.35 [−151.10, 136.55] | 0.60 |
| Cumulative fluid overload (%) | −2.94 [−24.03, 10.89] | −2.16 [−20.92, 21.30] | 0.36 |
| Major bleeding | 28 (60.9) | 39 (78.0) | 0.11 |
| Re-exploration | 28 (60.9) | 32 (64.0) | 0.92 |
| Intracranial hemorrhage | 0 (0.0) | 1 (2.0) | >0.99 |
| Gastrointestinal bleeding | 0 (0.0) | 8 (16.0) | 0.01 |
| Pulmonary bleeding | 1 (2.2) | 7 (14.0) | 0.09 |
| Hemolysis | 14 (30.4) | 33 (66.0) | 0.001 |
| Early hemolysis | 5 (10.9) | 14 (28.0) | 0.07 |
| Late hemolysis | 9 (19.6) | 19 (38.0) | 0.08 |
| Thrombocytopenia | 4 (8.7) | 24 (48.0) | <0.001 |
| ECMO duration (h) | 100.62 [90.25, 133.25] | 145.50 [102.50, 210.25] | 0.001 |
| CRRT | 3 (6.5) | 8 (16.0) | 0.26 |
| Successfully weaned from ECMO | 46 (100.0) | 21 (42.0) | <0.001 |
| Hospital stays (d) | 51.50 [41.25, 83.25] | 25.50 [14.50, 45.00] | <0.001 |
| ICU stays (d) | 33.50 [24.50, 55.50] | 15.50 [7.25, 37.25] | <0.001 |
| Ventilation time (h) | 562.00 [299.00, 934.50] | 331.50 [147.00, 818.00] | 0.01 |
| 30-day mortality | 0 (0.0) | 36 (72.0) | <0.001 |
| 180-day mortality | 0 (0.0) | 50 (100.0) | <0.001 |
Data are presented as median [interquartile range] or n (%). ALT, alanine aminotransferase; AST, aspartate aminotransferase; CPB, cardiopulmonary bypass; CRRT, continuous renal replacement therapy; ECMO, extracorporeal membrane oxygenation; ECPR, extracorporeal cardiopulmonary resuscitation; ICU, intensive care unit; LCOS, low cardiac output syndrome; RBC, red blood cell; STAT, Society of Thoracic Surgeons and European Association for Cardiothoracic Surgery Congenital Heart Surgery Mortality Categories; VIS, vasoactive-inotropic score; WBC, white blood cell.
Comparative analyses between groups indicated that, compared with survivors, non-survivors had significantly higher lactate levels, greater ECMO flow rates, greater RBC transfusion volumes, and increased UFH dosing. Non-survivors also had significantly lower nadir albumin concentration during ECMO, prolonged ECMO duration, extended hospital length of stay, and higher incidences of gastrointestinal bleeding and hemolysis (P<0.05). The remaining demographic and clinical characteristics were well balanced between groups (P>0.05).
Association between albumin and mortality
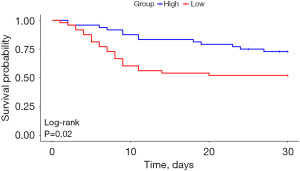
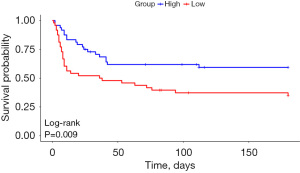
In Cox proportional hazards analyses, nadir albumin concentration during ECMO was found to be inversely associated with 30-day mortality, indicating that lower nadir albumin concentration corresponded to an increased risk of 30-day mortality. In the fully adjusted model, a reduction of 1 g/L in nadir albumin concentration was associated with a 7.6% increase in the risk of 30-day mortality [hazard ratio (HR): 0.924, 95% confidence interval (CI): 0.866–0.987]. A similar relationship was identified for the 180-day mortality risk (HR: 0.929, 95% CI: 0.880–0.981) (Table 2). The RCS curve demonstrated a linear relationship between nadir albumin concentration and mortality, with an inflection point identified at 31.42 g/L (Figures 1,2). This threshold was subsequently used to categorize the cohort into a low albumin group (≤31.42 g/L) and a high albumin group (>31.42 g/L) for survival analysis. Kaplan-Meier survival analysis revealed that the cumulative survival rates at 30 days (log-rank P=0.02) (Figure 3) and 180 days (log-rank P=0.009) (Figure 4) were significantly lower in the low albumin group compared to the high albumin group.
Table 2
| Model | 30-day mortality | 180-day mortality |
|---|---|---|
| Univariate | 0.925 (0.878–0.974) | 0.927 (0.887–0.969) |
| Multivariate | ||
| Model 1 | 0.919 (0.870–0.971) | 0.924 (0.882–0.967) |
| Model 2 | 0.908 (0.852–0.966) | 0.917 (0.871–0.965) |
| Model 3 | 0.924 (0.866–0.987) | 0.929 (0.880–0.981) |
Data are presented as hazard ratio (95% confidence interval).


Association between albumin and ECMO-related complications
To further explore potential pathways by which albumin may influence mortality, we conducted an assessment of the relationship between nadir albumin concentration and ECMO-related complications using multivariable logistic regression. The findings revealed that lower albumin concentration did not demonstrate a statistically significant association with the incidence of CRRT [odds ratio (OR): 0.851, 95% CI: 0.672–1.032], hemolysis (OR: 0.933, 95% CI: 0.852–1.016), major bleeding (OR: 0.917, 95% CI: 0.826–1.008), thrombosis (OR: 0.953, 95% CI: 0.871–1.039), or liver injury (OR: 0.997, 95% CI: 0.906–1.097) (Table 3).
Table 3
| Outcome | OR (95% CI) |
|---|---|
| Hemolysis | 0.933 (0.852–1.016) |
| Major bleeding | 0.917 (0.826–1.008) |
| CRRT | 0.851 (0.672–1.032) |
| Thrombosis | 0.953 (0.871–1.039) |
| Liver injury | 0.997 (0.906–1.097) |
CI, confidence interval; CRRT, continuous renal replacement therapy; ECMO, extracorporeal membrane oxygenation; OR, odds ratio.
Subgroup analysis
To evaluate whether the association between nadir albumin concentration and mortality differed across clinically relevant subgroups, we performed prespecified subgroup analyses stratified by age, body weight, and STAT category. It turned out that no significant interactions were observed between albumin concentration and any of the subgroup variables (all P for interaction >0.05), indicating that the association between lower nadir albumin concentration and increased mortality risk was generally consistent across these subgroups (Table 4).
Table 4
| Variables | Groups | 30-day mortality | 180-day mortality | |||
|---|---|---|---|---|---|---|
| HR (95% CI) | P for interaction | HR (95% CI) | P for interaction | |||
| Age | ≤12 months | 0.842 (0.753–0.942) | 0.22 | 0.889 (0.817–0.968) | 0.68 | |
| >12 months | 0.925 (0.818–1.047) | 0.921 (0.873–1.005) | ||||
| Weight | ≤10 kg | 0.847 (0.752–0.954) | 0.40 | 0.931 (0.845–1.026) | 0.96 | |
| >10 kg | 1.078 (0.880–1.321) | 0.977 (0.867–1.101) | ||||
| STAT | 1–3 | 0.920 (0.813–1.041) | 0.20 | 0.942 (0.853–1.040) | 0.17 | |
| 4–5 | 0.797 (0.613–1.035) | 0.856 (0.749–0.979) | ||||
CI, confidence interval; HR, hazard ratio; STAT, Society of Thoracic Surgeons and European Association for Cardiothoracic Surgery Congenital Heart Surgery Mortality Categories.
Sensitivity analysis
To assess the robustness of our findings, sensitivity analyses were performed. Restricting the cohort to patients whose nadir albumin occurred within 48 hours of ECMO initiation, the association between nadir albumin concentration and increased mortality remained consistent. Similarly, excluding patients who did not receive albumin supplementation did not materially alter the results, supporting the robustness of our findings (Table 5).
Table 5
| Group | Number | 30-day mortality (HR, 95% CI) | 180-day mortality (HR, 95% CI) |
|---|---|---|---|
| Group 1 | 83 | 0.925 (0.856–0.999) | 0.937 (0.882–0.995) |
| Group 2 | 91 | 0.926 (0.867–0.988) | 0.934 (0.884–0.986) |
Group 1: excluding participants whose nadir albumin occurred beyond 48 hours after ECMO initiation. Group 2: excluding participants who did not receive administration of albumin. CI, confidence interval; ECMO, extracorporeal membrane oxygenation; HR, hazard ratio.
Discussion
This study retrospectively analyzed 96 pediatric patients who underwent VA-ECMO support following CPB without remaining surgical deformities. It was found that lower nadir albumin concentration during ECMO was associated with higher 30-day and 180-day mortality, which remains robust in subgroup and sensitivity analysis. These findings were further demonstrated by Kaplan-Meier survival curves, which showed a clear separation between groups stratified by albumin concentration.
In this study, lower albumin concentration during ECMO was highly prevalent, a finding that is highly consistent with prior reports. Existing evidence has revealed that baseline albumin concentration measured before ECMO initiation is an independent predictor of 30-day mortality, and even with albumin supplementation, lower albumin concentration during ECMO is still associated with higher mortality and ECMO-related complications (21-24). In pediatric critical care, researchers also found that lower albumin concentration, whether present prior to ICU admission or within the first 24 hours of admission, has been identified as robust predictors of adverse clinical outcomes, with a tendency for albumin concentration to progressively decline (25-27). Beyond the ECMO and ICU settings, lower albumin concentration has been consistently associated with poorer outcomes across multiple pediatric populations, including children undergoing cardiac surgery, as well as neonates and premature infants (28-30). Collectively, these data reinforce the concept that serum albumin is not merely a nutritional marker but a dynamic indicator of illness severity, with broad prognostic relevance in critically ill children.
The findings presented suggest that serum albumin concentration, as a simple and accessible biomarker, may play a clinically meaningful role in risk stratification for pediatric patients receiving postcardiotomy ECMO support. However, several issues require exploration. Firstly, the underlying cause of lower albumin concentration must be addressed; in our study, the median pre-ECMO albumin concentration was 42.30 g/L, which decreased markedly to 31.45 g/L during ECMO, indicating that the reduction in albumin concentration was largely an ECMO-associated phenomenon rather than a purely pre-existing condition. Impaired hepatic synthetic function might be the most immediate consideration, as the liver is the primary site of albumin production and hepatic dysfunction has been linked to reduced albumin concentration (31). However, our multivariable logistic regression analysis didn’t identify a significant association between liver injury and lower albumin concentration, suggesting that impaired hepatic synthesis alone was insufficient to account for the marked decline observed during ECMO (32). Systemic inflammatory activation represents another plausible mechanism. ECMO is known to be accompanied by a pronounced inflammatory response, which may lead to endothelial dysfunction in capillaries, increased endothelial permeability, and in severe cases, capillary leak syndrome, thereby causing protein loss (33). Although CRP levels were comparable between the low- and high-albumin groups in our cohort (Table S4), additional inflammatory biomarkers were not routinely measured, limiting definitive conclusions regarding the contribution of inflammation. Fluid overload is also likely relevant. Large volumes of crystalloid are commonly administered during ECMO to maintain intravascular volume, potentially leading to hemodilution. Indeed, fluid overload was significantly greater in the low-albumin group, suggesting that dilutional effects may have contributed to reduced albumin concentration. Although these factors may all contribute, owing to the retrospective design, we were unable to fully disentangle the relative contributions of these mechanisms, and future studies should explore strategies to mitigate these factors and assess their relationship with albumin dynamics.
Secondly, it remains unclear whether albumin exerts a direct effect on mortality, contributes indirectly through increased complications, or primarily serves as a prognostic marker. To address this question, we evaluated the association between albumin concentration and major ECMO complications. In our cohort, lower albumin concentration was not significantly associated with CRRT requirement, major bleeding, hemolysis, thrombosis, or liver injury. It may be due to insufficient statistical power, but we think these findings can support the interpretation that lower nadir albumin concentration is more appropriately regarded as a prognostic biomarker rather than a direct mediator of adverse events. The observed association between lower albumin concentration and increased mortality risk may relate to the physiological functions of albumin in maintaining vascular homeostasis, modulating inflammatory responses, and exerting antioxidant effects (34). ECMO is often accompanied by excessive inflammatory responses and oxidative stress, which in some cases may precipitate systemic inflammatory response syndrome (11,35,36). In the setting of reduced albumin concentrations, the buffering and endothelial-protective functions of albumin may be impaired, potentially contributing to increased vascular permeability, glycocalyx degradation, and microcirculatory dysfunction (37). These pathophysiological interactions may help explain the observed relationship between lower albumin concentration and adverse clinical outcomes, although causality cannot be inferred from the present analysis.
Considering the clinical associations discussed above, an important question is whether therapeutic albumin supplementation could alleviate ECMO-related declines in albumin concentration and improve outcomes. Nonetheless, the existing evidence is quite controversial. Several studies have indicated that early administration of albumin for volume resuscitation is associated with improved survival in critically ill patients, including those receiving ECMO and extracorporeal cardiopulmonary resuscitation (ECPR) (38-40). In contrast, other investigations indicate that albumin supplementation could be associated with increased risk of bleeding and AKI or other complications (41-43). Additional studies have reported no clear benefit or impairment of albumin supplementation on outcomes following cardiac surgery or other critical illnesses (44,45). Emerging evidence even further complicates the situation. Lin et al. reported that early albumin supplementation may be associated with higher risk of mortality compared with late use, while hyperoncotic albumin solutions appear to be associated with greater mortality risk compared with iso-oncotic albumin (46,47). Collectively, these findings imply that the efficacy and safety of albumin may vary according to patient group and the specific albumin formulation administered. In our study, the association between lower nadir albumin concentration and increased mortality remained statistically significant even after adjusting for albumin supplementation dose. This finding reinforces the interpretation of nadir albumin primarily as a prognostic biomarker rather than direct evidence supporting albumin supplementation as a therapeutic intervention. Whether albumin replacement is beneficial in pediatric ECMO patients—and, if so, the optimal timing, dosing strategies, and patient selection criteria—requires rigorous prospective evaluation.
However, there are certain limitations in this study. Firstly, as a single-center retrospective analysis, it is inherently susceptible to selection bias and information bias, which may limit the generalizability of the findings. Additionally, given the observational design of our study, residual confounding such as treatment-response dynamics and reverse causality cannot be excluded, and these pathophysiologic links remain speculative. Second, the relatively small sample size may reduce the statistical power and increase the uncertainty of effect estimates. This concern is particularly relevant to the neonatal population, in whom serum albumin may play a distinct and clinically important role. Previous studies have suggested that lower albumin concentration is a potentially modifiable risk factor for adverse events in neonates (48). However, the number of neonates included in our cohort was small, precluding meaningful subgroup analyses within this specific population. Third, serum albumin serves as a dynamic biomarker that is affected by the severity of acute illness, fluid balance, nutritional reserves, and the burden of inflammation. Our analysis concentrated solely on the nadir albumin concentration, which may not comprehensively reflect longitudinal trends or the patient’s overall nutritional and inflammatory status. Finally, considering the prolonged study period, the possibility of an era effect cannot be ruled out, and secular changes in clinical practice over time may have introduced residual confounding.
Conclusions
In summary, this retrospective study identified an association between lower nadir albumin concentration during pediatric ECMO and an elevated risk of mortality at both 30 and 180 days, and may serve as a readily available marker for risk stratification, warranting validation in prospective studies. It is recommended that routine monitoring of albumin concentration be incorporated into daily assessments.
Acknowledgments
None.
Footnote
Reporting Checklist: The authors have completed the STROBE reporting checklist. Available at https://tp.amegroups.com/article/view/10.21037/tp-2026-1-0008/rc
Data Sharing Statement: Available at https://tp.amegroups.com/article/view/10.21037/tp-2026-1-0008/dss
Peer Review File: Available at https://tp.amegroups.com/article/view/10.21037/tp-2026-1-0008/prf
Funding: This work was supported by
Conflicts of Interest: All authors have completed the ICMJE uniform disclosure form (available at https://tp.amegroups.com/article/view/10.21037/tp-2026-1-0008/coif). The authors have no conflicts of interest to declare.
Ethical Statement: The authors are accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved. The study was conducted in accordance with the Declaration of Helsinki and its subsequent amendments. The study was approved by the Ethics Committee of Fuwai Hospital (approval No. 2020-1346). Given the retrospective nature of the research, the requirement for informed consent was waived.
Open Access Statement: This is an Open Access article distributed in accordance with the Creative Commons Attribution-NonCommercial-NoDerivs 4.0 International License (CC BY-NC-ND 4.0), which permits the non-commercial replication and distribution of the article with the strict proviso that no changes or edits are made and the original work is properly cited (including links to both the formal publication through the relevant DOI and the license). See: https://creativecommons.org/licenses/by-nc-nd/4.0/.
References
- Ali J, Vuylsteke A. Extracorporeal membrane oxygenation: indications, technique and contemporary outcomes. Heart 2019;105:1437-43. [Crossref] [PubMed]
- Tonna JE, Boonstra PS, MacLaren G, et al. Extracorporeal Life Support Organization Registry International Report 2022: 100,000 Survivors. ASAIO J 2024;70:131-43. [Crossref] [PubMed]
- Millar JE, Fanning JP, McDonald CI, et al. The inflammatory response to extracorporeal membrane oxygenation (ECMO): a review of the pathophysiology. Crit Care 2016;20:387. [Crossref] [PubMed]
- Belinskaia DA, Voronina PA, Shmurak VI, et al. Serum Albumin in Health and Disease: Esterase, Antioxidant, Transporting and Signaling Properties. Int J Mol Sci 2021;22:10318. [Crossref] [PubMed]
- Li N, Qiao H, Guo JF, et al. Preoperative hypoalbuminemia was associated with acute kidney injury in high-risk patients following non-cardiac surgery: a retrospective cohort study. BMC Anesthesiol 2019;19:171. [Crossref] [PubMed]
- Hansrivijit P, Yarlagadda K, Cheungpasitporn W, et al. Hypoalbuminemia is associated with increased risk of acute kidney injury in hospitalized patients: A meta-analysis. J Crit Care 2021;61:96-102. [Crossref] [PubMed]
- Yoshida B, Nguyen A, Formanek B, et al. Hypoalbuminemia and Elevated CRP are Risk Factors for Deep Infections and Urinary Tract Infections After Lumbar Spine Surgery in a Large Retrospective Patient Population. Global Spine J 2023;13:33-44. [Crossref] [PubMed]
- Zou Y, Kuang M, Xiong S, et al. Association of albumin trajectories and cumulative exposure with in-hospital mortality in acute pancreatitis: a retrospective cohort study. Int J Surg 2026;112:132-45. [Crossref] [PubMed]
- Staudacher DL, Gold W, Biever PM, et al. Early fluid resuscitation and volume therapy in venoarterial extracorporeal membrane oxygenation. J Crit Care 2017;37:130-5. [Crossref] [PubMed]
- Caprarola SD, Ng DK, Carroll MK, et al. Pediatric ECMO: unfavorable outcomes are associated with inflammation and endothelial activation. Pediatr Res 2022;92:549-56. [Crossref] [PubMed]
- Liu T, Amos SH, Ganga AL, et al. Evaluation of cytokine response to extracorporeal membrane oxygenation. Artif Organs 2022;46:146-54. [Crossref] [PubMed]
- Atkinson JP, Waldmann TA, Stein SF, et al. Systemic capillary leak syndrome and monoclonal IgG gammopathy; studies in a sixth patient and a review of the literature. Medicine (Baltimore) 1977;56:225-39. [Crossref] [PubMed]
- Hellenthal KEM, Brabenec L, Wagner NM. Regulation and Dysregulation of Endothelial Permeability during Systemic Inflammation. Cells 2022;11:1935. [Crossref] [PubMed]
- Siddall E, Khatri M, Radhakrishnan J. Capillary leak syndrome: etiologies, pathophysiology, and management. Kidney Int 2017;92:37-46. [Crossref] [PubMed]
- Peterson EJ, Ng TMH, Patel KA, et al. Association of admission vs. nadir serum albumin concentration with short-term treatment outcomes in patients with acute heart failure. J Int Med Res 2018;46:3665-74.
- Jin Y, Feng Z, Zhao J, et al. Outcomes and factors associated with early mortality in pediatric postcardiotomy veno-arterial extracorporeal membrane oxygenation. Artif Organs 2021;45:6-14. [Crossref] [PubMed]
- Experts consensus on the application of continuous blood purification in critically ill children. Zhonghua Er Ke Za Zhi 2021;59:352-60.
- Chawla LS, Bellomo R, Bihorac A, et al. Acute kidney disease and renal recovery: consensus report of the Acute Disease Quality Initiative (ADQI) 16 Workgroup. Nat Rev Nephrol 2017;13:241-57. [Crossref] [PubMed]
- O'Brien SM, Clarke DR, Jacobs JP, et al. An empirically based tool for analyzing mortality associated with congenital heart surgery. J Thorac Cardiovasc Surg 2009;138:1139-53. [Crossref] [PubMed]
- Gaies MG, Jeffries HE, Niebler RA, et al. Vasoactive-inotropic score is associated with outcome after infant cardiac surgery: an analysis from the Pediatric Cardiac Critical Care Consortium and Virtual PICU System Registries. Pediatr Crit Care Med 2014;15:529-37. [Crossref] [PubMed]
- Miyoshi T, Nishimura T, Higashi H, et al. The effect of serum albumin levels before IMPELLA insertion on mortality risk in patients with cardiogenic shock. Heart Vessels 2025;40:905-12. [Crossref] [PubMed]
- Jeon JB, Lee CH, Lim Y, et al. Hypoalbuminemia and Albumin Replacement during Extracorporeal Membrane Oxygenation in Patients with Cardiogenic Shock. J Chest Surg 2023;56:244-51. [Crossref] [PubMed]
- Huang M, Ong BH, Hoo AEE, et al. Prognostic Factors for Survival After Extracorporeal Membrane Oxygenation for Cardiogenic Shock. ASAIO J 2020;66:141-5. [Crossref] [PubMed]
- Raja KM, Plazak M, Rabin J, et al. Hypoalbuminemia is a predictor of mortality in patients with cardiogenic shock requiring veno-arterial extracorporeal membrane oxygenation. Perfusion 2025;40:1193-201. [Crossref] [PubMed]
- Ari HF, Turanli EE, Yavuz S, et al. Association between serum albumin levels at admission and clinical outcomes in pediatric intensive care units: a multi-center study. BMC Pediatr 2024;24:844. [Crossref] [PubMed]
- Bekhit OE, Yousef RM, Abdelrasol HA, et al. Serum Albumin Level as a Predictor of Outcome in Patients Admitted to Pediatric Intensive Care Units. Pediatr Emerg Care 2021;37:e855-60. [Crossref] [PubMed]
- Gowa MA, Tauseef U, Ahmed SH. A relation between serum albumin level and prognosis of critically ill children admitted to the paediatric Intensive Care Unit. J Pak Med Assoc 2023;73:1034-42. [Crossref] [PubMed]
- Watchko JF, Spitzer AR, Clark RH. Prevalence of Hypoalbuminemia and Elevated Bilirubin/Albumin Ratios in a Large Cohort of Infants in the Neonatal Intensive Care Unit. J Pediatr 2017;188:280-286.e4. [Crossref] [PubMed]
- Henry BM, Borasino S, Ortmann L, et al. Perioperative serum albumin and its influence on clinical outcomes in neonates and infants undergoing cardiac surgery with cardiopulmonary bypass: a multi-centre retrospective study. Cardiol Young 2019;29:761-7. [Crossref] [PubMed]
- Torer B, Hanta D, Yapakci E, et al. Association of Serum Albumin Level and Mortality in Premature Infants. J Clin Lab Anal 2016;30:867-72. [Crossref] [PubMed]
- Kumar-M P, Mishra S, Jha DK, et al. Coronavirus disease (COVID-19) and the liver: a comprehensive systematic review and meta-analysis. Hepatol Int 2020;14:711-22. [Crossref] [PubMed]
- Spinella R, Sawhney R, Jalan R. Albumin in chronic liver disease: structure, functions and therapeutic implications. Hepatol Int 2016;10:124-32. [Crossref] [PubMed]
- Kapoor PM, Narula J, Chowdhury UK, et al. Serum albumin perturbations in cyanotics after cardiac surgery: Patterns and predictions. Ann Card Anaesth 2016;19:300-5. [Crossref] [PubMed]
- Manolis AA, Manolis TA, Melita H, et al. Low serum albumin: A neglected predictor in patients with cardiovascular disease. Eur J Intern Med 2022;102:24-39. [Crossref] [PubMed]
- Passmore MR, Ki KK, Chan CHH, et al. The effect of hyperoxia on inflammation and platelet responses in an ex vivo extracorporeal membrane oxygenation circuit. Artif Organs 2020;44:1276-85. [Crossref] [PubMed]
- Edinger F, Zajonz T, Holtz L, et al. New Insights into Hepatic and Intestinal Microcirculation and Pulmonary Inflammation in a Model of Septic Shock and Venovenous Extracorporeal Membrane Oxygenation in the Rat. Int J Mol Sci 2024;25:6621. [Crossref] [PubMed]
- Tang F, Zhao XL, Xu LY, et al. Endothelial dysfunction: Pathophysiology and therapeutic targets for sepsis-induced multiple organ dysfunction syndrome. Biomed Pharmacother 2024;178:117180. [Crossref] [PubMed]
- Thang C, Marella P, Kumar A, et al. Early Albumin Exposure After Cardiac Surgery. J Cardiothorac Vasc Anesth 2022;36:1310-7. [Crossref] [PubMed]
- Wengenmayer T, Hirth ML, Jäckel M, et al. Early Albumin Administration in Veno-Arterial Extracorporeal Membrane Oxygenation. Artif Organs 2025;49:872-9. [Crossref] [PubMed]
- Wengenmayer T, Schroth F, Biever PM, et al. Albumin fluid resuscitation in patients on venoarterial extracorporeal membrane oxygenation (VA-ECMO) therapy is associated with improved survival. Intensive Care Med 2018;44:2312-4. [Crossref] [PubMed]
- Talvasto A, Ilmakunnas M, Raivio P, et al. Albumin Infusion and Blood Loss After Cardiac Surgery. Ann Thorac Surg 2023;116:392-9. [Crossref] [PubMed]
- Shehabi Y, Balachandran M, Al-Bassam W, et al. Postoperative 20% Albumin Infusion and Acute Kidney Injury in High-Risk Cardiac Surgery Patients: The ALBICS AKI Randomized Clinical Trial. JAMA Surg 2025;160:835-44. [Crossref] [PubMed]
- Li XY, Chen WS, Qu ZK, et al. Early use of albumin may increase the risk of sepsis-associated acute kidney injury in sepsis patients: a target trial emulation. Mil Med Res 2025;12:51. [Crossref] [PubMed]
- Pesonen E, Vlasov H, Suojaranta R, et al. Effect of 4% Albumin Solution vs Ringer Acetate on Major Adverse Events in Patients Undergoing Cardiac Surgery With Cardiopulmonary Bypass: A Randomized Clinical Trial. JAMA 2022;328:251-8. [Crossref] [PubMed]
- Skubas NJ, Callum J, Bathla A, et al. Intravenous albumin in cardiac and vascular surgery: a systematic review and meta-analysis. Br J Anaesth 2024;132:237-50. [Crossref] [PubMed]
- Lin F, Xu D, Tang J, et al. Early human albumin infusion was associated with 28-day mortality in patients with acute respiratory failure and hypoalbuminaemia in the intensive care unit: a retrospective study of the MIMIC-IV database. Respir Med 2025;247:108273. [Crossref] [PubMed]
- Patanwala AE, Flannery AH, Mehta HB, et al. Comparative Effectiveness of Albumin vs No Albumin on Renal Replacement Therapy and Mortality in Patients With Septic Shock and Renal Impairment. Chest 2025;167:1090-8. [Crossref] [PubMed]
- Nada A, Askenazi D, Kupferman JC, et al. Low albumin levels are independently associated with neonatal acute kidney injury: a report from AWAKEN Study Group. Pediatr Nephrol 2022;37:1675-86. [Crossref] [PubMed]