Clinical characteristics and prognostic factors influencing respiratory syncytial virus pneumonia in children with atopy: a retrospective cohort study
Highlight box
Key findings
• Children with respiratory syncytial virus pneumonia (RSVP) and atopy (determined post-hospitalization) are more likely to develop severe pneumonia and may have a higher percentage of requiring oxygen inhalation and hormone therapy than children without atopy. Shortness of breath and pulmonary rales are risk factors of severe RSVP in children with atopy, which have good clinical predictive value for the occurrence of severe pneumonia in children with atopy.
What is known and what is new?
• Respiratory syncytial virus (RSV) is a major threat to the health of children. Children with atopy are susceptible to respiratory tract infections and symptoms become more severe after infection. Different atopy subtypes might be associated with the severity of pneumonia and the poor prognosis of RSV infections.
• Children with atopic RSVP also have a higher rate of needing oxygen and hormone therapy. Shortness of breath and pulmonary rales have good clinical predictive value for the occurrence of severe pneumonia in children with atopy. The risk of asthma is significantly increased in children with atopy after discharge.
What is the implication, and what should change now?
• The risk of asthma in children with atopy and RSVP still existed after hospital discharge. Therefore, early identification and intervention are needed and the respiratory rate and lung auscultation are recommended as expeditious bedside tools.
Introduction
Respiratory syncytial virus (RSV) is one of the main pathogens causing acute lower respiratory tract infections in children <5 years of age. Approximately 33.1 million cases of lower respiratory tract infections are caused by RSV globally each year (1). RSV pneumonia (RSVP) is associated with recurrent wheezing and potentially leads to a future diagnosis of asthma in some cases (2). Severe RSV bronchiolitis may even progress to obliterative bronchiolitis, which may become a major threat to the health of children (2).
Atopy refers to a susceptibility to allergic diseases, including atopic dermatitis, rhinitis, and asthma. A previous study indicated that children with atopy are susceptible to respiratory tract infections and symptoms become more severe after infection (3). Specifically, it has been reported that RSV-specific immunoglobulin E (IgE) and immunoglobulin G4 (IgG4) antibodies are risk markers for the development of atopic diseases in children (4). Previous studies have emphasized the association between RSV infection and asthma (4,5). Different subtypes of atopy might be associated with the severity of pneumonia and the poor prognosis of RSV infections (4,5), while the clinical diversity, risk of severe disease, and long-term prognosis of the atopic subgroups warrant further study.
In the current study, the clinical characteristics and prognostic factors influencing children with atopic RSVP were retrospectively analyzed and the predictive clinical indicators of severe RSVP were investigated. We present this article in accordance with the STROBE reporting checklist (available at https://tp.amegroups.com/article/view/10.21037/tp-2026-1-0045/rc).
Methods
Subjects of the study
This retrospective study was conducted in accordance with the Declaration of Helsinki and its subsequent amendments. The study was approved by the Ethics Committee of the Xi’an Children’s Hospital (No. 2025-013-02) and individual consent was obtained from the parents of the children. Children diagnosed with RSVP and admitted to the Second Respiratory Department of Xi’an Children’s Hospital between January 2023 and December 2024 were selected. Atopic status of children was determined post-hospitalization for RSVP, which was independent of the outcome assessment to ensure methodological transparency and avoid bias. According to the atopic status, these children were divided into atopic and non-atopic groups.
Diagnostic criteria
The diagnostic criteria for RSVP as well as the intra- and extrapulmonary complications and severity were as follows: the 2019 norms for the diagnosis and treatment of children’s community-acquired pneumonia (6); and the 2023 Chinese expert consensus on the clinical diagnosis and treatment of RSV infections in children based on the epidemiologic history, clinical and imaging manifestations, and RSV pathogenesis (7). Nasopharyngeal swab virus nucleic acid testing was performed on all children.
The diagnostic criteria for atopy were as follows (8,9): (I) a clear personal allergic history diagnosed by the physician, including atopic dermatitis, allergic rhinitis or food allergies, or a positive skin prick test; or (II) positive serum and food allergen screening results [specific IgE (sIgE) ≥0.35 kUA/L].
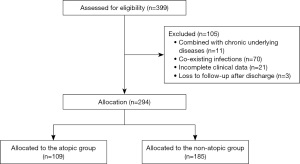
The exclusion criteria were as follows: (I) chronic underlying diseases, such as congenital pulmonary dysplasia, airway malformation, malnutrition, congenital inherited metabolic diseases, or immunodeficiency; (II) co-existing infections like bacterial, fungal, Mycoplasma or other viral mixed infections; and (III) incomplete clinical data, patients who were not treated based on standardized clinical pathway guidelines, or patients who missed follow-up appointments after discharge.
Data collection
Basic information of children, such as age, gender, length of hospital stay, and clinical manifestations, including fever (axillary body temperature >37 ℃), severe cough (cough at night or vomiting after cough), wheezing, shortness of breath, pulmonary rales, intra- and extra-pulmonary complications, and incidence of severe pneumonia, were collected. The laboratory indicators measured at the time of admission included routine blood tests, inflammatory markers, liver enzymes, lactate dehydrogenase, and creatine kinase isoenzyme. The main treatment measures for the children included oxygen inhalation, use of immunoglobulins, hormone therapy, bronchoscopy, and bronchoalveolar lavage. Persistent asthma and newly diagnosed asthma after discharge were also recorded.
Hormone therapy refers to systemic glucocorticoid therapy, including intravenous or oral administration methods. Oxygen inhalation indications (7) were defined as (I) at sea level while breathing room air; and (II) SaO2 ≤0.92 or arterial partial pressure of oxygen ≤80 mmHg (1 mmHg = 0.133 kPa). If central cyanosis was used as an oxygen inhalation indication, central cyanosis should be combined with signs, such as rapid breathing, inspiratory depression of the lower chest wall, restlessness, and moaning during breathing (7). Attention should also be paid to whether there was severe anemia, methemoglobinemia, and poor peripheral circulation. The oxygen inhalation method should be selected based on the degree of hypoxia, such as nasal catheters, masks, and hoods. Then, the oxygen supply was stopped until the blood oxygen saturation was stable and no support was required (7). Intrapulmonary complications referred to pleural effusion or empyema, pneumothorax, pyopneumothorax, pulmonary bullae, lung abscess, necrotizing pneumonia, bronchopleural fistula, acute respiratory distress syndrome, and acute respiratory failure.
Statistical analysis
SPSS 25.0 statistical software was used for statistical analysis of the data. The measurement data that conformed to a normal distribution are expressed as the mean ± standard deviation (SD) and measurement data not conforming to a normal distribution are expressed as median with interquartile range (IQR). An independent two-sample t-test was used for inter-group comparisons of data conforming to a normal distribution and homogeneity of variance. Otherwise, the Mann-Whitney U test was used. The counting data are expressed as frequencies and percentages and a chi-square test or Fisher’s exact test was used for inter-group comparisons. Logistic regression analysis was also performed for univariate and multivariate analyses of risk factors for severe pneumonia in children with atopic and RSVP. Statistical significance was considered at a P<0.05.
Results
General data and clinical characteristics
A total of 294 children with RSVP were included in the current study with 109 in the atopic group and 185 in the non-atopic group (Figure 1). Among the 109 atopic children, 68 (62.4%) were diagnosed with atopy based on a clear personal allergy history and 41 (37.6%) were diagnosed based on a positive skin prick test or positive serum and food allergen screening results (sIgE ≥0.35 kUA/L).
The general data of the children were shown in Table 1. No statistical difference between the atopic and non-atopic groups was detected with respect to age, gender ratio, hospitalization days, fever, febrile days, wheezing, and incidence of extrapulmonary complications (P>0.05). The children in the atopic group had a higher incidence of severe pneumonia, severe cough, shortness of breath, pulmonary rales, and intrapulmonary complications than the non-atopic group and the difference was statistically significant (P<0.05). A higher percentage of children required oxygen inhalation and hormone therapy in the atopic group than the non-atopic group (P<0.05).
Table 1
| Groups | Atopic (n=109) | Non-atopic (n=185) | χ2/Z | P |
|---|---|---|---|---|
| Age (months) | 21 (6.5, 43) | 14 (7, 30) | −1.663 | 0.10 |
| Gender (male/female) | 68/41 | 108/77 | 0.458 | 0.50 |
| Hospitalization days | 6 (5, 7) | 6 (5, 7) | −1.290 | 0.20 |
| Severe pneumonia | 30 (27.5) | 32 (17.3) | 4.310 | 0.04 |
| Fever | 59 (54.1) | 96 (51.9) | 0.069 | 0.79 |
| Febrile days | 2 (0, 4) | 1 (0, 3) | −0.940 | 0.35 |
| Severe cough | 43 (39.4) | 51 (27.6) | 4.452 | 0.04 |
| Wheezing | 80 (73.4) | 124 (67.0) | 1.309 | 0.25 |
| Shortness of breath | 35 (32.1) | 39 (21.1) | 4.430 | 0.04 |
| Pulmonary rales | 44 (40.3) | 50 (27) | 4.160 | 0.041 |
| Intrapulmonary complications | 8 (7.3) | 3 (1.6) | 4.74 | 0.03 |
| Extrapulmonary complications | 8 (7.3) | 6 (3.2) | 2.438 | 0.11 |
| Oxygen inhalation | 20 (18.3) | 16 (8.7) | 5.744 | 0.02 |
| Hormone therapy | 31 (28.4) | 34 (18.4) | 4.032 | 0.045 |
Data are presented as median (interquartile range) or n (%). RSVP, respiratory syncytial virus pneumonia.
Laboratory indicator differences
No differences were detected in the white blood cell, neutrophil, lymphocyte, eosinophil, and red blood cell counts and hemoglobin, procalcitonin, high-sensitivity C-reactive protein, aspartate aminotransferase, lactate dehydrogenase, and creatine kinase isoenzyme levels between the two groups (P>0.05). The platelet count [285 (IQR: 244, 370) vs. 353 (IQR: 256, 451.5) ×109/L] and alanine aminotransferase levels [20 (IQR: 15, 28) vs. 22 (IQR: 16, 31.5) U/L] differed significantly between the two groups (P<0.05, Table 2); however, the clinical relevance for these two indicators was limited because their values remained within the reference ranges.
Table 2
| Group | Atopic (n=109) | Non-atopic (n=185) | Z | P |
|---|---|---|---|---|
| White blood cell count (×109/L) | 7.55 (5.68, 10.21) | 7.87 (6.30, 10.26) | −1.305 | 0.19 |
| Neutrophil count (×109/L) | 2.5 (1.67, 4.29) | 2.6 (1.63, 3.94) | −0.23 | 0.98 |
| Lymphocyte count (×109/L) | 3.44 (2.22, 5.0) | 3.9 (2.54, 5.73) | −1.803 | 0.07 |
| Eosinophil count (×109/L) | 0.08 (0.02, 0.25) | 0.06 (0.02, 0.175) | −1.342 | 0.18 |
| Red blood cell count (×1012/L) | 4.42 (4.16, 4.69) | 4.43 (4.03, 4.73) | −1.209 | 0.23 |
| Hemoglobin (g/L) | 122 (113.5, 130.0) | 119 (109.5, 129.5) | −1.894 | 0.06 |
| Platelet count (×109/L) | 285 (244, 370) | 353 (256, 451.5) | −2.812 | 0.01 |
| Procalcitonin (ng/mL) | 0.06 (0, 0.16) | 0.06 (0, 0.11) | −1.026 | 0.31 |
| High-sensitive C-reactive protein (mg/L) | 1.82 (0.5, 7.44) | 1.82 (0.38, 7.16) | −0.755 | 0.45 |
| Alanine aminotransferase (U/L) | 22 (16, 31.5) | 20 (15, 28) | −2.082 | 0.04 |
| Aspartate aminotransferase (U/L) | 40 (33, 49) | 41 (33, 50.5) | −0.447 | 0.66 |
| Lactate dehydrogenase (U/L) | 265 (235.5, 324) | 282 (251.5, 323) | −1.218 | 0.22 |
| Creatine kinase isoenzyme (U/L) | 23 (19, 28.5) | 24 (20, 30) | −0.658 | 0.51 |
Data are presented as median (interquartile range). RSVP, respiratory syncytial virus pneumonia.
Risk factors for children with atopy and RSVP
Univariate logistic regression analysis was performed with the following independent variables: age; gender; febrile days; severe cough; shortness of breath; wheezing; pulmonary rales; white blood cell, neutrophil, lymphocyte, eosinophil, red blood cell, and platelet count; and hemoglobin, procalcitonin, high-sensitivity C-reactive protein, alanine aminotransferase, aspartate aminotransferase, lactate dehydrogenase, and creatine kinase isoenzyme levels. The incidence of severe pneumonia was the dependent variable. The factors with a statistical difference based on univariate logistic regression analysis were included in the multivariate logistic regression analysis. The multivariate logistic regression analysis indicated shortness of breath and pulmonary rales as independent risk factors in children with atopy and severe pneumonia (P<0.05; Table 3).
Table 3
| Variables | Univariate logistic regression analysis | Multivariate logistic regression analysis | |||||
|---|---|---|---|---|---|---|---|
| B | P | OR (95% CI) | B | P | OR (95% CI) | ||
| Age (months) | −0.014 | 0.21 | 0.986 (0.964–1.008) | ||||
| Gender (female) | 0.679 | 0.15 | 1.973 (0.783–4.973) | ||||
| Febrile days | 0.026 | 0.78 | 1.027 (0.854–1.235) | ||||
| Severe cough | 0.789 | 0.07 | 2.201 (0.936–5.174) | ||||
| Shortness of breath | 1.917 | <0.001* | 6.801 (2.701–17.122) | 1.397 | 0.02* | 4.042 (1.292–12.642) | |
| Wheezing | 1.485 | 0.02* | 4.415 (1.225–15.908) | 0.790 | 0.28 | 2.202 (0.526–9.217) | |
| Pulmonary rales | 1.737 | <0.001* | 5.681 (2.265–14.248) | 1.276 | 0.02* | 3.583 (1.268–10.118) | |
| White blood cell count (×109/L) | 0.221 | 0.003* | 1.247 (1.077–1.444) | 0.109 | 0.35 | 1.115 (0.888–1.400) | |
| Neutrophil count (×109/L) | 0.156 | 0.11 | 1.169 (0.965–1.416) | ||||
| Lymphocyte count (×109/L) | 0.216 | 0.03* | 1.241 (1.024–1.505) | 0.010 | 0.90 | 1.010 (0.873–1.168) | |
| Eosinophil count (×109/L) | 0.210 | 0.35 | 1.234 (0.791–1.923) | ||||
| Red blood cell count (×1012/L) | −0.624 | 0.23 | 0.536 (0.193–1.489) | ||||
| Hemoglobin (g/L) | −0.013 | 0.31 | 0.987 (0.962–1.012) | ||||
| Platelet count (×109/L) | 0.004 | 0.04* | 1.004 (1.000–1.008) | 0.002 | 0.51 | 0.998 (0.992–1.004) | |
| Procalcitonin (ng/mL) | 0.367 | 0.51 | 1.444 (0.488–4.276) | ||||
| High-sensitivity C-reactive protein (mg/L) | 0.026 | 0.10 | 1.026 (0.995–1.059) | ||||
| Alanine aminotransferase (U/L) | 0.024 | 0.02* | 1.025 (1.004–1.045) | 0.003 | 0.90 | 0.997 (0.955–1.041) | |
| Aspartate aminotransferase (U/L) | 0.031 | 0.02* | 1.032 (1.006–1.058) | 0.028 | 0.32 | 1.028 (0.974–1.085) | |
| Lactate dehydrogenase (U/L) | 0.222 | 0.058 | 1.249 (0.992–1.572) | ||||
| Creatine kinase isoenzyme (U/L) | 0.040 | 0.07 | 1.040 (0.998–1.085) | ||||
*, P<0.05. CI, confidence interval; OR, odds ratio; RSVP, respiratory syncytial virus pneumonia.
Persistent and newly diagnosed asthma among children with RSVP after hospital discharge
No statistical difference was detected in the number of children with persistent asthma discharged within 1 and 3 months between the two groups (P>0.05). However, the number of children with newly diagnosed asthma in the atopic group within 6 months after hospital discharge was greater than the non-atopic group (P<0.05; Table 4).
Table 4
| Group | Atopic (n=109) | Non-atopic (n=185) | χ2 | P |
|---|---|---|---|---|
| Persistent asthma at 1 month | 2 (1.8) | 1 (0.5) | 1.138 | 0.29 |
| Persistent asthma at 3 months | 4 (3.7) | 1 (0.5) | 2.364 | 0.12 |
| Newly diagnosed asthma at 6 months | 6 (5.5) | 1 (0.5) | 5.293 | 0.02 |
Data are presented as n (%). RSV, respiratory syncytial virus.
Discussion
The children in the atopic group had a higher rate of severe pneumonia, shortness of breath, pulmonary rales, and use of oxygen and hormone therapy than the non-atopic group. The findings herein are consistent with previous findings involving atopy and Mycoplasma pneumoniae pneumonia (3-5,10), which indicated that immune system disorders of children with atopy may be associated with intensified inflammatory reactions. Children with atopy tend to have higher levels of inflammatory mediators, which continually cause tissue inflammation and damage and lead to more severe airway inflammation and clinical manifestations (11). In addition, the airway hyperresponsiveness of children with atopy may intensify the symptoms, including shortness of breath and pulmonary rales. In addition, children with atopy had a higher incidence of severe cough, which agreed with the findings reported by Li et al. (12). All the results indicated a significant role for atopy in the development of pneumonia in children, highlighting the need for early identification and intervention. Therefore, the follow-up period should be extended for future studies to dynamically evaluate pulmonary dysfunction and the conversion rate of asthma (13,14).
Although children in the atopic group required more oxygen inhalation and hormone therapy, bronchoscopic or bronchoalveolar lavage requirements, hospitalization days, and short-term prognosis between the two groups were not significantly different. All the results indicated that even though the symptoms were more severe during the acute period, children with atopy still have a comparable short-term prognosis to children without atopy if standard treatment is performed. The previous report (15) also showed that hormone therapy caused delayed clearance of the viral load even though the symptoms were quickly relieved, which demonstrated the necessity of more precise immunoregulation strategies for atopic children.
Previous studies also showed adaptive remodeling of airway ultrastructure caused by the immune response, neural regulatory mechanisms, and persistence of RSV in children after infection might also lead to airway hyperresponsiveness and increased risk of long-term asthma (5,16-18). The follow-up results in the current study also showed that a higher percentage of children with atopy and RSVP have newly diagnosed asthma within 6 months after hospital discharge than the non-atopic group, emphasizing the need of establishing a long-term follow-up system for children with atopy and RSVP.
Although the statistical analysis of the platelet counts and the alanine aminotransferase level had a P<0.05, the clinical value of the platelet count [285 (IQR: 244, 370) vs. 353 (IQR: 256, 451.5) ×109/L] and alanine aminotransferase level [20 (IQR:15, 28) vs. 22 (IQR: 16, 31.5) U/L] had no clinical significance. Therefore, these laboratory results had no clinical value.
The risk factors for severe RSVP in children included age, immune status, basic health status, and clinical manifestations after viral infection. The current study also showed shortness of breath and pulmonary rales to be independent risk factors for the incidence of severe pneumonia in children with atopy and RSVP because of the interaction between the atopic status of children and RSV infection, which is consistent with previous studies (3-5,19,20). Antiviral immune dysfunction in children with atopy significantly increases susceptibility to viral infections and worsens the severity of disease progression (21). Children with atopy experience immune overactivation caused by the Th2 immunodominant response after RSV infection, which leads to massive accumulation of inflammatory cells and mediators in the lungs. Immune overactivation induces proliferation, differentiation, and airway remodeling of airway epithelial cells and further increases bronchi and alveoli exudation and impacts the function of ventilation and air exchange, causing shortness of breath (13,22,23). In addition, inflammatory exudation and airway spasm will generate abnormal sounds when the airflow is passing through narrow or irregular airways, leading to more widespread pulmonary inflammation and rales (13,22,23). This result provided further support for the respiratory rate and lung auscultation as expeditious bedside tools, especially for grassroots medical institutions with limited resources. In addition, allergic diseases may gradually develop into allergic asthma and allergic rhinitis in childhood with increasing age, which is defined as the “atopic march” (24,25). The older children in the atopic group of the current study may have a longer period of exhibiting atopic symptoms, which may affect the identification and recording of their allergy history.
However, severe pneumonia was not associated with hospitalization days or febrile days in this study. The length of hospital stay is determined by the recovery of organ function and complications, while the number of febrile days better reflects the acute inflammatory response. The length of hospital stay is more dependent on severe complications, such as respiratory failure and sepsis (26). Moreover, each child has a different constitution and the responses to treatment and immune regulation also vary (27). These clinical variables may be the reasons for the lack of an apparent association between severe pneumonia and the length of hospital stay, as well as the number of febrile days.
There were limitations in this study. First, there may have been bias for cohort selection because this was a single-center retrospective cohort design. A multi-center prospective study is needed for verification. Second, the current length of follow up was only 6 months. A 12- and possibly 24-month post-hospitalization follow-up is required to fully understand the association of developing persistent asthma. We will endeavor to work on the follow-up data in the future. Third, the exclusion of patients “who missed follow-up appointments” may introduce attrition bias, and may lead to an overestimation or underestimation of post-discharge asthma incidence. Fourth, although some differences, such as the platelet count and alanine aminotransferase level, between the two groups were statistically significant, the laboratory results had limited clinical relevance. Fifth, the personal allergic history confirmed by physicians through retrospective data extraction may be inconsistent and vulnerable to recall bias; and older children have had more time to manifest atopic symptoms due to atopic march, which may lead to some bias in the recognition of allergy history. Finally, the severity of atopy or multiple allergies was not stratified due to the limitation of sample size. Further studies, considering age matching or correction based on age in multivariate analysis, including severity of atopy classification, are still needed.
Conclusions
Children with RSVP and atopy (determined post-hospitalization) are more likely to develop severe pneumonia and may have a higher percentage of requiring oxygen inhalation and hormone therapy than children without atopy. Shortness of breath and pulmonary rales are risk factors for severe RSVP in children with atopy, which have good clinical predictive value for the occurrence of severe pneumonia in children with atopy. Furthermore, the risk of asthma in children with atopy and RSVP still existed after hospital discharge. Therefore, early identification and intervention are needed and the respiratory rate and lung auscultation are recommended as expeditious bedside tools.
Acknowledgments
None.
Footnote
Reporting Checklist: The authors have completed the STROBE reporting checklist. Available at https://tp.amegroups.com/article/view/10.21037/tp-2026-1-0045/rc
Data Sharing Statement: Available at https://tp.amegroups.com/article/view/10.21037/tp-2026-1-0045/dss
Peer Review File: Available at https://tp.amegroups.com/article/view/10.21037/tp-2026-1-0045/prf
Funding: This study was supported by
Conflicts of Interest: All authors have completed the ICMJE uniform disclosure form (available at https://tp.amegroups.com/article/view/10.21037/tp-2026-1-0045/coif). The authors have no conflicts of interest to declare.
Ethical Statement: The authors are accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved. This retrospective study was conducted in accordance with the Declaration of Helsinki and its subsequent amendments. The study was approved by the Ethics Committee of the Xi’an Children’s Hospital (No. 2025-013-02) and individual consent was obtained from the parents of the children.
Open Access Statement: This is an Open Access article distributed in accordance with the Creative Commons Attribution-NonCommercial-NoDerivs 4.0 International License (CC BY-NC-ND 4.0), which permits the non-commercial replication and distribution of the article with the strict proviso that no changes or edits are made and the original work is properly cited (including links to both the formal publication through the relevant DOI and the license). See: https://creativecommons.org/licenses/by-nc-nd/4.0/.
References
- Li Y, Wang X, Blau DM, et al. Global, regional, and national disease burden estimates of acute lower respiratory infections due to respiratory syncytial virus in children younger than 5 years in 2019: a systematic analysis. Lancet 2022;399:2047-64. [Crossref] [PubMed]
- Soni A, Kabra SK, Lodha R. Respiratory Syncytial Virus Infection: An Update. Indian J Pediatr 2023;90:1245-53. [Crossref] [PubMed]
- Narayanan D, Grayson MH. Comparing respiratory syncytial virus and rhinovirus in development of post-viral airway disease. J Asthma 2022;59:434-41. [Crossref] [PubMed]
- Tesari Crnković H, Bendelja K, Drkulec V, et al. Respiratory Syncytial Virus-Specific Antibodies and Atopic Diseases in Children: A 10-Year Follow-Up. Pathogens 2023;12:546. [Crossref] [PubMed]
- Rosas-Salazar C, Chirkova T, Gebretsadik T, et al. Respiratory syncytial virus infection during infancy and asthma during childhood in the USA (INSPIRE): a population-based, prospective birth cohort study. Lancet 2023;401:1669-80. [Crossref] [PubMed]
- National Health Commission of the People’s Republic of China SAoTCMNAoTCM. Guideline for diagnosis and treatment of community-acquired pneumonia in Children (2019 version). Chinese Journal of Clinical Infectious Diseases 2019;12:6-13.
- National Clinical Research Center for Child Health and Disorders. Infectious group of Chinese Pediatric Society CMAIDBoSMA. Chinese expert consensus on clinical diagnosis and treatment of respiratory syncytial virus infection in children (2023). Journal of Clinical Pediatrics 2024;42:1-14.
- Bian C, Li S, Huo S, et al. Association of atopy with disease severity in children with Mycoplasma pneumoniae pneumonia. Front Pediatr 2023;11:1281479. [Crossref] [PubMed]
- Lv JL, Wang WJ. Clinical characteristics of adenovirus pneumonia in children with atopic constitution. Chinese Pediatric Emergency Medicine 2024;31:761-6.
- Qin Y, Yang Y, Li J, et al. The impact of atopy on the clinical characteristics of mycoplasma pneumoniae pneumonia in pediatric patients. Sci Rep 2025;15:2503. [Crossref] [PubMed]
- Wang ZH, Wang YS, Liu Y, et al. Clinical characteristics and the levels of interleukin-17 in Mycoplasma pneumoniae pneumonia children with atopic sensitization. Chinese Journal of Applied Clinical Pediatrics 2017;32:668-71.
- Li M, Han XH, Liu LY, et al. Epidemiological characteristics, clinical characteristics, and prognostic factors of children with atopy hospitalised with adenovirus pneumonia. BMC Infect Dis 2021;21:1051. [Crossref] [PubMed]
- Akdis CA, Arkwright PD, Brüggen MC, et al. Type 2 immunity in the skin and lungs. Allergy 2020;75:1582-605. [Crossref] [PubMed]
- Zhang Y, Li J, Zhang L, et al. Vascular endothelial growth factors in airway allergic diseases: pathophysiological functions and therapeutic prospects. Expert Rev Clin Immunol 2025;21:577-86. [Crossref] [PubMed]
- Li J, Liao X, Zhou Y, et al. Association between glucocorticoids treatment and viral clearance delay in patients with COVID-19: a systematic review and meta-analysis. BMC Infect Dis 2021;21:1063. [Crossref] [PubMed]
- Qin L, Qiu KZ, Hu CP, et al. Bronchial Epithelial Cells Promote the Differentiation of Th2 Lymphocytes in Airway Microenvironment through Jagged/Notch-1 Signaling after RSV Infection. Int Arch Allergy Immunol 2019;179:43-52. [Crossref] [PubMed]
- Rossi GA, Colin AA. Respiratory syncytial virus-Host interaction in the pathogenesis of bronchiolitis and its impact on respiratory morbidity in later life. Pediatr Allergy Immunol 2017;28:320-31. [Crossref] [PubMed]
- Schuurhof A, Janssen R, de Groot H, et al. Local interleukin-10 production during respiratory syncytial virus bronchiolitis is associated with post-bronchiolitis wheeze. Respir Res 2011;12:121. [Crossref] [PubMed]
- China Medicine Education Association Committee on Pediatrics. The Subspecialty Group of Respiratory Diseases TSoP, Chinese Medical Association; Chinese Medical Doctor Association Committee on Respirology Pediatrics; et al. Chinese pediatric guideline for diagnosis, treatment, and prevention of respiratory syncytial virus infection (medical version, 2024). Chinese Journal of Applied Clinical Pediatrics 2024;39:723-32.
- Gong C, Wang X, Luo M, et al. Disease Surveillance 2020;35:613-7.
- Lv JL, Wang WJ. Research on mechanisms of action between respiratory viral infections and allergic diseases in children. Chinese Journal of Applied Clinical Pediatrics 2025;40:153-6.
- Gandhi NA, Pirozzi G, Graham NMH. Commonality of the IL-4/IL-13 pathway in atopic diseases. Expert Rev Clin Immunol 2017;13:425-37. [Crossref] [PubMed]
- Norlander AE, Peebles RS Jr. Innate Type 2 Responses to Respiratory Syncytial Virus Infection. Viruses 2020;12:521. [Crossref] [PubMed]
- Yang L, Fu J, Zhou Y. Research Progress in Atopic March. Front Immunol 2020;11:1907. [Crossref] [PubMed]
- Choi UE, Deng J, Parthasarathy V, et al. Risk factors and temporal associations of progression of the atopic march in children with early-onset atopic dermatitis. J Am Acad Dermatol 2025;92:732-40. [Crossref] [PubMed]
- Yang S, Lu S, Guo Y, et al. A comparative study of general and severe mycoplasma pneumoniae pneumonia in children. BMC Infect Dis 2024;24:449. [Crossref] [PubMed]
- Lin Y, Shi Q, Yang J, et al. Association of anthropometric z score with complications and length of hospital stay in children with severe pneumonia aged 3 months to 5 years. Nutr Clin Pract 2024;39:459-69. [Crossref] [PubMed]