A simplified prediction model for in-hospital mortality in children with congenital heart disease after surgery
Highlight box
Key findings
• This study developed a parsimonious risk scoring system to predict in-hospital mortality for children with congenital heart disease (CHD) following cardiac surgery. Derived from the Pediatric Intensive Care (PIC) database, the model identifies seven independent predictors: sex, age, hemoglobin, mean corpuscular volume, albumin, calcium, and pH. This simplified tool demonstrates robust performance for early postoperative risk stratification in the intensive care setting.
What is known and what is new?
• Postoperative mortality for complex CHD remains significant (5–15%). Existing risk models are often complex or reliant on intraoperative data, limiting their utility for rapid bedside assessment in the Pediatric Intensive Care Unit (PICU).
• This work addresses this gap by introducing a novel prediction model specifically designed for the postoperative PICU phase. Its innovation lies in utilizing only seven routine, immediately available variables to create a practical bedside score. This facilitates proactive, data-driven risk stratification immediately after surgery, filling a critical need in current clinical practice.
What is the implication, and what should change now?
• The clinical implementation of this simplified risk score enables a proactive shift in postoperative care, allowing for early identification of high-risk children to guide intensified monitoring and timely interventions. We recommend prospective validation and integration of this tool into standard PICU workflows for children after CHD surgery.
Background
Congenital heart disease (CHD) refers to a group of diseases characterized by structural or functional defects of the heart, resulting from abnormal development of the heart and great vessels during the fetal period (1-3). As the most common birth defect in newborns worldwide, its clinical management and prognosis improvement have always been important challenges in the field of pediatric cardiovascular medicine (4,5). According to the latest data from the National University Heart Centre, Singapore (NUHCS), the incidence of CHD ranges from 6 to 13 per 1,000 live births, affecting approximately 1 in every 120–166 newborns and it has shown a significant upward trend in recent years, imposing a heavy public health burden (6,7). CHD covers a wide range from simple defects [such as ventricular septal defect (VSD) and atrial septal defect (ASD)] to complex malformations (such as patients with single ventricle physiology). Its pathological heterogeneity directly determines the selection of treatment strategies and differences in prognosis (8).
Besides cardiac catheterization, surgery is one of the important treatment methods for CHD. By repairing or replacing diseased cardiac tissues, it can help restore normal cardiac anatomy and physiology, provide a stable source of systemic or pulmonary blood flow, and improve cardiac function (9). With the innovation of surgical techniques and advances in postoperative management, the expectations after CHD surgery have shifted from simple anatomical correction to improvement of quality of life (10). However, the postoperative mortality rate of some children with complex CHD (especially those with single-ventricle physiology or combined with important vascular malformations) remains as high as 5–15%. Premature birth, small for gestational age, low birth weight, preoperative malnutrition, recurrent pulmonary infections, heart failure, and genetic syndromes such as DiGeorge syndrome all diminish surgical tolerance (11,12). Active identification of risk factors for postoperative death during the postoperative period can help evaluate the patient’s general condition in advance, strengthen postoperative monitoring and management, reduce complications, and even lower the mortality rate (13,14). Therefore, establishing an accurate preoperative risk assessment system, especially identifying children at high risk of postoperative death, is of urgent practical significance for optimizing surgical decision-making and rational allocation of medical resources.
Currently, internationally commonly used risk prediction models for postoperative mortality and complication rates in children with CHD mainly include the Risk Adjustment in Congenital Heart Surgery-1 (RACHS-1) (15), the Society of Thoracic Surgeons-European Association for Cardiothoracic Surgery (STS-EACTS) Congenital Heart Surgery Mortality Score (16), and the Aristotle Basic Complexity Score (ABC) (17). These models have played a certain role in clinical practice but also have certain limitations (18). For example, the RACHS-1 score mainly conducts risk stratification based on the type of surgery, which may not accurately reflect the real risk for some complex cases; in addition, these scores require specialized surgical knowledge and are not familiar to non-surgical intensive care unit (ICU) medical staff. This study uses the large-scale Chinese Pediatric Intensive Care (PIC) database to construct a scoring system for predicting postoperative death risk in CHD children based on postoperative clinical indicators, aiming to provide a potential new approach for the prognosis evaluation and management of CHD children. We present this article in accordance with the TRIPOD reporting checklist (available at https://tp.amegroups.com/article/view/10.21037/tp-2026-1-0051/rc).
Methods
Study subjects
This study adopted a retrospective cohort design, with data sourced from the publicly available PIC database. Established by the Children’s Hospital of Zhejiang University School of Medicine, this database contains clinical data of children admitted to multiple pediatric ICU of the hospital from 2010 to 2018, including structured information such as demographic characteristics, diagnostic data, vital signs, laboratory tests, therapeutic interventions, and outcome indicators. All identifiers have been de-identified. The establishment and public use of this database were approved by the Medical Ethics Committee of the Children’s Hospital of Zhejiang University School of Medicine, and individual informed consent was waived for this study. The study was conducted in accordance with the Declaration of Helsinki and its subsequent amendments.
Inclusion criteria: (I) children who underwent cardiac repair surgery for CHD with corresponding surgical name records; (II) children with baseline information on gender, age, and ethnicity; (III) children who underwent laboratory blood cell tests, blood biochemistry, and blood gas analysis after being admitted to the ICU during the postoperative period. Exclusion criteria: (I) cases with implausible or logically inconsistent temporal data (e.g., PICU admission time earlier than birth time or other clearly erroneous time orders); (II) missing key indicators in laboratory blood cell tests, blood biochemistry, or blood gas analysis, such as major cytological classifications, serum proteins, and acid-base balance indicators.
Collection of clinical data
The PIC surgical database was systematically screened for surgical name records to identify CHD children meeting the above criteria. Information including surgical name, operation date, hospital number, and unique patient identifier was extracted. Based on the hospital number and unique patient identifier, data on baseline characteristics, length of hospital stay, laboratory blood cell indices, blood biochemistry, blood gas analysis indicators, and in-hospital outcomes were extracted from the PIC database’s basic characteristic data table, admission information table, hospitalization data table, and laboratory test data table, respectively.
All but one patient was admitted to the PICU within 24 hours postoperatively. The initial laboratory values obtained following PICU admission were used for subsequent analysis. Laboratory indicators were classified as follows: H (higher than normal), L (lower than normal), and N (within normal range) (classification criteria on website of PIC: http://pic.nbscn.org). The primary outcome indicator of this study was in-hospital mortality, defined as all-cause death at any time from the operation day to discharge.
Statistical analysis
Categorical variables were expressed as frequencies (n) and percentages (%), and comparisons between groups were performed using the Pearson Chi-squared test or Fisher’s exact test. Continuous variables were tested for normality using the Shapiro-Wilk test; those with a normal distribution were described as mean ± standard deviation (mean ± SD), and those with a non-normal distribution were described as median and interquartile range (IQR).
Taking in-hospital mortality as the dependent variable, laboratory indicators with P<0.05 in univariate analysis and preset clinical covariates (age, gender) were included in multivariate logistic regression based on Fisher scoring. To address potential collinearity and redundancy among predictors, we employed a multi-step variable selection strategy. First, for variables capturing the same clinical dimension, we selected representative predictors based on clinical relevance and statistical efficiency to reduce dimensionality. We then performed forward stepwise logistic regression for variable selection, with entry significance levels set at α=0.05 (Tables S1,S2). Following model estimation, we assessed collinearity using tolerance and the variance inflation factor (VIF) (Table S3).
A simplified factor scoring system was developed based on the regression coefficients of each independent variable in the final Logistic regression model. Receiver operating characteristic (ROC) curves were plotted, and the area under the curve (AUC) were calculated. Differences in AUC values were compared using the DeLong non-parametric test, and the optimal cut-off value was determined based on the maximum Youden index. To quantify optimism and assess model stability, we performed bootstrap internal validation with 500 resamples. A P<0.05 was considered statistically significant. All statistical analyses were two-tailed and performed using SAS version 9.4 (SAS Institute Inc.).
Results
Basic characteristics and postoperative outcomes of children with CHD
A total of 1,982 children with CHD who underwent cardiac surgery and were admitted to the ICU were finally included (Figure 1). The median age was 13.5 months (IQR: 5.0–35.8 months), among which children over 3 months old accounted for 84.9%. VSD repair (861 cases, 43.4%), ASD repair (625 cases, 31.5%), and patent ductus arteriosus (PDA) closure (330 cases, 16.7%) were the top three most common types of surgery in sequence. The median length of ICU stay was 2.0 days (IQR: 1.0–4.3 days). There were 25 cases of in-hospital death in the entire cohort, with an in-hospital mortality of 1.26% [95% confidence interval (CI): 0.77–1.75%]. The median postoperative survival time of deceased children was 7.0 days (IQR: 0.7–17.7 days), with the shortest survival time being 1 day and the longest 48.3 days.

Risk factors for in-hospital death after CHD surgery
The univariate analysis showed that male and younger children had a significantly increased risk of in-hospital death (Table 1). Among peripheral blood indicators, patients with abnormal red blood cell (RBC), hematocrit, mean corpuscular volume (MCV), hemoglobin, white blood cell (WBC), lymphocyte percentage, eosinophil percentage, and neutrophil count had a higher in-hospital mortality (Table 1). Among biochemical indicators, patients with abnormal albumin, aspartate transaminase, urea, calcium, cholesterol, creatinine, γ-glutamyl transferase, indirect bilirubin, magnesium, phosphorus, and uric acid had a higher in-hospital mortality (Table S4). Among blood gas analysis indicators, patients with abnormal partial pressure of carbon dioxide, partial pressure of oxygen, oxygen saturation, lactic acid, pH, and base excess had a higher in-hospital mortality (Table S5).
Table 1
| Variables | Total | Death in hospital | P | |
|---|---|---|---|---|
| No (n=1,957) | Yes (n=25) | |||
| Gender | ||||
| Female | 1,043 (52.62) | 1,037 (99.42) | 6 (0.58) | 0.004 |
| Male | 939 (47.38) | 920 (97.98) | 19 (2.02) | |
| Age | ||||
| <28 day | 106 (5.35) | 96 (90.57) | 10 (9.43) | <0.001 |
| 28 day–3 months | 194 (9.79) | 189 (97.42) | 5 (2.58) | |
| >3 months | 1,682 (84.86) | 1,672 (99.41) | 10 (0.59) | |
| WBC | ||||
| H | 326 (16.45) | 319 (97.85) | 7 (2.15) | <0.001 |
| L | 108 (5.45) | 101 (93.52) | 7 (6.48) | |
| N | 1,548 (78.10) | 1,537 (99.29) | 11 (0.71) | |
| Percentage of lymphocyte | ||||
| H | 1,623 (81.89) | 1,611 (99.26) | 12 (0.74) | <0.001 |
| L | 73 (3.68) | 67 (91.78) | 6 (8.22) | |
| N | 286 (14.43) | 279 (97.55) | 7 (2.45) | |
| Percentage of eosinophils | ||||
| H | 135 (6.81) | 134 (99.26) | 1 (0.74) | <0.001 |
| L | 346 (17.46) | 334 (96.53) | 12 (3.47) | |
| N | 1,501 (75.73) | 1,489 (99.20) | 12 (0.80) | |
| Neutrophils | ||||
| H | 100 (5.05) | 91 (91.00) | 9 (9.00) | <0.001 |
| L | 162 (8.17) | 161 (99.38) | 1 (0.62) | |
| N | 1,720 (86.78) | 1,705 (99.13) | 15 (0.87) | |
| Percentage of basophils | ||||
| H | 139 (7.01) | 139 (100.0) | 0 (0.00) | 0.25 |
| N | 1,843 (92.99) | 1,818 (98.64) | 25 (1.36) | |
| Hematocrit | ||||
| H | 133 (6.71) | 122 (91.73) | 11 (8.27) | <0.001 |
| L | 174 (8.78) | 170 (97.70) | 4 (2.30) | |
| N | 1,675 (84.51) | 1,665 (99.40) | 10 (0.60) | |
| Hemoglobin | ||||
| H | 41 (2.07) | 36 (87.80) | 5 (12.20) | <0.001 |
| L | 523 (26.39) | 508 (97.13) | 15 (2.87) | |
| N | 1,418 (71.54) | 1,413 (99.65) | 5 (0.35) | |
| Mean corpuscular volume | ||||
| H | 248 (12.51) | 234 (94.35) | 14 (5.65) | <0.001 |
| L | 171 (8.63) | 167 (97.66) | 4 (2.34) | |
| N | 1,563 (78.86) | 1,556 (99.55) | 7 (0.45) | |
| Blood platelet | ||||
| H | 460 (23.21) | 457 (99.35) | 3 (0.65) | 0.27 |
| L | 31 (1.56) | 30 (96.77) | 1 (3.23) | |
| N | 1,491 (75.23) | 1,470 (98.59) | 21 (1.41) | |
| RBC | ||||
| H | 94 (4.74) | 88 (93.62) | 6 (6.38) | <0.001 |
| L | 250 (12.61) | 236 (94.40) | 14 (5.60) | |
| N | 1,638 (82.64) | 1,633 (99.69) | 5 (0.31) | |
Data are presented as n (%). H, higher than normal; L, lower than normal; N, normal; RBC, red blood cell; WBC, white blood cell.
Predictive model for postoperative mortality in children with CHD
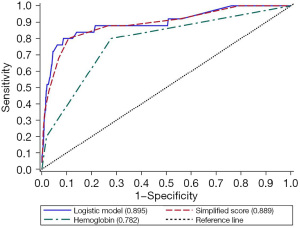
Through stepwise screening, a total of seven independent variables, namely gender, age, hemoglobin, MCV, albumin, calcium, and pH, were retained in the logistic regression model (Table 2). The AUC of the Logistic regression model was 0.895 (95% CI: 0.818–0.973). After transforming the logistic model into a simplified scoring system (Table 3), the AUC of the scoring system’s ROC curve was 0.889 (95% CI: 0.813–0.966), showing no statistical difference compared with the logistic model (P=0.19). However, it was higher than the highest single factor, hemoglobin (0.782, 95% CI: 0.694–0.869; P<0.001) (Figure 2). Bootstrap internal validation yielded an optimism-corrected AUC of 0.888, indicating negligible overfitting. Detailed calibration metrics (Brier score, sensitivity, and specificity) were provided in Table S6 and Figure S1. When the cut-off value of the simplified scoring system was set at 5 points, the sensitivity and specificity of prediction were 80.0% and 89.3%, respectively. Using 5 and 10 points as cut-off values, children with CHD were divided into low-risk (<5 points), moderate-risk (5–10 points), and high-risk (≥10 points) groups. The mortality rates of these groups were 0.21% (3/1,439), 2.55% (13/510), and 27.27% (9/33), respectively (P<0.001). Compared with the low-risk group, the odds ratios (ORs) of in-hospital death for the moderate- and high-risk groups were 12.52 (95% CI: 3.55–44.12) and 179.50 (95% CI: 45.73–704.63), respectively.
Table 2
| Variables | Multivariable logistic regression model | OR (95% CI) | ||
|---|---|---|---|---|
| Parameter | SE | P | ||
| Gender | ||||
| F vs. M | −0.534 | 0.266 | 0.045 | 0.344 (0.121–0.977) |
| Age | ||||
| <1 m vs. ≥3 m | 0.558 | 0.466 | 0.17 | 5.125 (0.775–33.912) |
| 1–3 m vs. ≥3 m | 0.518 | 0.406 | 4.927 (0.882–27.538) | |
| Hemoglobin | ||||
| H vs. N | 2.029 | 0.437 | 0.001 | 35.986 (8.108–159.716) |
| L vs. N | −0.475 | 0.370 | 2.944 (0.831–10.43) | |
| MCV | ||||
| H vs. N | −0.086 | 0.539 | >0.99 | 2.524 (0.565–11.272) |
| L vs. N | 1.099 | 0.549 | 8.258 (1.773–38.46) | |
| Albumin | ||||
| H vs. N | 1.841 | 0.915 | 0.09 | 17.502 (1.315–232.89) |
| L vs. N | −0.819 | 0.670 | 1.224 (0.275–5.458) | |
| Calcium | ||||
| H vs. N | −0.176 | 0.737 | 0.008 | 1.867 (0.219–15.898) |
| L vs. N | 0.976 | 0.504 | 5.905 (1.919–18.164) | |
| pH | ||||
| H vs. N | 0.007 | 0.749 | 0.04 | 2.181 (0.207–23.03) |
| L vs. N | 0.766 | 0.449 | 4.658 (1.424–15.235) | |
CI, confidence interval; F, female; H, higher than normal; L, lower than normal; m, month; M, male; MCV, mean corpuscular volume; N, normal; OR, odds ratio; SE, standard error.
Table 3
| Variables | Score† |
|---|---|
| Gender | |
| Male | 1 |
| Female | 0 |
| Age | |
| <3 months | 1 |
| ≥3 months | 0 |
| Hemoglobin | |
| Higher than normal | 4 |
| Lower than normal | 2 |
| Normal | 0 |
| MCV | |
| Higher than normal | 2 |
| Lower than normal | 3 |
| Normal | 0 |
| Albumin | |
| Higher than normal | 4 |
| Lower than normal | 1 |
| Normal | 0 |
| Calcium | |
| Higher than normal | 1 |
| Lower than normal | 3 |
| Normal | 0 |
| pH | |
| Higher than normal | 1 |
| Lower than normal | 2 |
| Normal | 0 |
†, a higher score corresponds to an increased risk of mortality. MCV, mean corpuscular volume;
Discussion
In this study, children with CHD who underwent cardiac surgery from the PIC database were included and developed a risk prediction model for postoperative in-hospital mortality based on postoperative clinical data. Seven predictors (gender, age, hemoglobin, MCV, albumin, calcium, and pH) were identified, and the multivariate model constructed with these seven factors showed strong predictive ability. Furthermore, this complex multivariate regression model was successfully transformed into a simple numerical risk scoring system, which also demonstrated equally strong predictive performance. Unlike existing risk scoring models for CHD surgery, the simple numerical risk scoring system developed in this study is designed for enhanced applicability in real-world clinical settings. Through straightforward table lookup and calculation, clinicians can rapidly assess postoperative mortality risk in children with CHD. This tool is particularly valuable for healthcare providers without specialized pediatric cardiac training, offering them a quantitative approach to monitor high-risk patients. Our simplified scoring model is intended to complement, rather than replace, existing risk stratification systems.
This study found that male gender and younger age are strong predictors of mortality, which is highly consistent with the physiological characteristics in cardiac surgery. Younger children (<3 months old) have lower body weight, immature myocardial development, and weak immune response, making them prone to low cardiac output syndrome after cardiopulmonary bypass (19,20). A single-center cohort study identified low body weight as a risk factor for postoperative mortality in children with CHD (21). The increased risk in males may be attributed to differences in sex hormone-related or sex chromosome-associated immune gene expression (22), a phenomenon also reported in coronavirus disease 2019 (COVID-19) infections (23). In addition, high hemoglobin levels (rather than anemia, as traditionally recognized) predict poor outcomes, which may reflect compensatory erythrocytosis secondary to cyanotic CHD, leading to high blood viscosity and microcirculatory disorders, thereby increasing the risk of postoperative mortality (24). Meanwhile, low MCV suggests iron metabolism disorders, which may exacerbate tissue oxygen supply impairment. Notably, a prospective observational study has identified the post-walk oxygen saturation (SpO2) level following a preoperative 6-minute walk test (6MWT) as an independent prognostic indicator for children undergoing CHD surgery (25). Surgical technical performance (including cardiopulmonary bypass time, aortic cross-clamp time, the use of deep hypothermic circulatory arrest, and surgical complexity) has a significant impact on postoperative survival (26). Of course, the quality of surgical technical performance can, to a certain extent, be reflected in postoperative laboratory indicators.
As predictors, low albumin (<30 g/L) and low blood calcium (<2.1 mmol/L) highlight the critical impact of nutritional and metabolic homeostasis on surgical prognosis in children with CHD. As a core protein for maintaining colloid osmotic pressure and drug binding, reduced albumin levels can aggravate capillary leakage and organ edema. Meanwhile, hypoalbuminemia is a recognized indicator for predicting CHD surgical risks, commonly observed in patients with cyanotic CHD (27). Cardiac automaticity and contractile function are mediated by calcium ions; hypocalcemia can directly impair myocardial contractility and vascular tone, particularly during CHD surgery with cardiopulmonary bypass, where calcium ions are heavily consumed (28). This model further confirms that acidosis (pH <7.35) is a strong predictor of postoperative mortality, possibly due to anaerobic metabolism-induced lactic acid accumulation and hepatorenal dysfunction (29).
The simple risk scoring system constructed in this study (requiring only 7 commonly available clinical indicators) significantly improves clinical practicality while maintaining the discriminative power of the original model. Its stratification efficiency (mortality rate >27% in the high-risk group) is superior to existing tools, and a score ≥5 points can achieve 80% sensitivity, ensuring no missed diagnosis of high-risk children. In addition, for children in the moderate-risk group (5–10 points, mortality rate 2.55%), this model can prompt PICU medical staff to strengthen postoperative monitoring of CHD, enabling early intervention based on early identification to improve the prognosis of children. This scoring system is applicable to rapid postoperative risk assessment in hospitals at all levels, providing a basis for referral decisions and individualized cardiopulmonary bypass strategies.
Due to these limitations, caution should be exercised when interpreting and applying the prediction model established in this study, as the model was developed in an exploratory rather than confirmatory manner. Firstly, the PIC database is a single-center retrospective design; despite strict data cleaning in this study, selection bias may still exist. Secondly, the sample size of outcome events in the cohort leads to potential instability of the model, despite good performance in internal validation. Thirdly, categorizing continuous laboratory indicators, although intended to enhance clinical applicability, inevitably results in information loss and may introduce residual confounding. Future studies should conduct external validation through multi-center prospective studies and explore the integration of real-time dynamic scoring systems with electronic medical records to optimize clinical decision support tools and improve medical quality and patient care.
Conclusions
In conclusion, based on 7 commonly available postoperative clinical indicators, this study successfully established a simple scoring system with high ability to predict and distinguish postoperative mortality risks in children with CHD. This provides a new tool for the prognosis evaluation and management of children patients.
Acknowledgments
None.
Footnote
Reporting Checklist: The authors have completed the TRIPOD reporting checklist. Available at https://tp.amegroups.com/article/view/10.21037/tp-2026-1-0051/rc
Peer Review File: Available at https://tp.amegroups.com/article/view/10.21037/tp-2026-1-0051/prf
Funding: None.
Conflicts of Interest: All authors have completed the ICMJE uniform disclosure form (available at https://tp.amegroups.com/article/view/10.21037/tp-2026-1-0051/coif). The authors have no conflicts of interest to declare.
Ethical Statement: The authors are accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved. The study was conducted in accordance with the Declaration of Helsinki and its subsequent amendments.
Open Access Statement: This is an Open Access article distributed in accordance with the Creative Commons Attribution-NonCommercial-NoDerivs 4.0 International License (CC BY-NC-ND 4.0), which permits the non-commercial replication and distribution of the article with the strict proviso that no changes or edits are made and the original work is properly cited (including links to both the formal publication through the relevant DOI and the license). See: https://creativecommons.org/licenses/by-nc-nd/4.0/.
References
- Amdani S, Conway J, George K, et al. Evaluation and Management of Chronic Heart Failure in Children and Adolescents With Congenital Heart Disease: A Scientific Statement From the American Heart Association. Circulation 2024;150:e33-50. [Crossref] [PubMed]
- Paul T, Krause U, Sanatani S, et al. Advancing the science of management of arrhythmic disease in children and adult congenital heart disease patients within the last 25 years. Europace 2023;25:euad155. [Crossref] [PubMed]
- Barker P. Council on Pediatric and Congenital Heart Disease (PCHD) Year-In-Review. J Am Soc Echocardiogr 2021;34:A13-4. [Crossref] [PubMed]
- Singh Y, Lakshminrusimha S. Perinatal Cardiovascular Physiology and Recognition of Critical Congenital Heart Defects. Clin Perinatol 2021;48:573-94. [Crossref] [PubMed]
- Chowdhury D, Elliott PA, Asaki SY, et al. Addressing Disparities in Pediatric Congenital Heart Disease: A Call for Equitable Health Care. J Am Heart Assoc 2024;13:e032415. [Crossref] [PubMed]
- Hummel K, Whittaker S, Sillett N, et al. Development of an international standard set of clinical and patient-reported outcomes for children and adults with congenital heart disease: a report from the International Consortium for Health Outcomes Measurement Congenital Heart Disease Working Group. Eur Heart J Qual Care Clin Outcomes 2021;7:354-65. [Crossref] [PubMed]
- Yan MX, Zhao Y, Zhao DD, et al. The Association of Folic Acid, Iron Nutrition during Pregnancy and Congenital Heart Disease in Northwestern China: A Matched Case-Control Study. Nutrients 2022;14:4541. [Crossref] [PubMed]
- Haga T. Characteristics of Patients With Congenital Heart Disease Requiring ICU Admission From Japanese Emergency Departments. Pediatr Crit Care Med 2020;21:e1106-12. [Crossref] [PubMed]
- Lim JCE, Elliott MJ, Wallwork J, et al. Cardiac surgery and congenital heart disease: reflections on a modern revolution. Heart 2022;108:787-93. [Crossref] [PubMed]
- Mandalenakis Z, Giang KW, Eriksson P, et al. Survival in Children With Congenital Heart Disease: Have We Reached a Peak at 97%? J Am Heart Assoc 2020;9:e017704. [Crossref] [PubMed]
- Massey SL, Weinerman B, Naim MY. Perioperative Neuromonitoring in Children with Congenital Heart Disease. Neurocrit Care 2024;40:116-29. [Crossref] [PubMed]
- Edelson JB, Rossano JW, Griffis H, et al. Resource Use and Outcomes of Pediatric Congenital Heart Disease Admissions: 2003 to 2016. J Am Heart Assoc 2021;10:e018286. [Crossref] [PubMed]
- Nasr VG, Markham LW, Clay M, et al. Perioperative Considerations for Pediatric Patients With Congenital Heart Disease Presenting for Noncardiac Procedures: A Scientific Statement From the American Heart Association. Circ Cardiovasc Qual Outcomes 2023;16:e000113. [Crossref] [PubMed]
- Peyvandi S, Xu D, Barkovich AJ, et al. Declining Incidence of Postoperative Neonatal Brain Injury in Congenital Heart Disease. J Am Coll Cardiol 2023;81:253-66. [Crossref] [PubMed]
- Allen P, Zafar F, Mi J, et al. Risk Stratification for Congenital Heart Surgery for ICD-10 Administrative Data (RACHS-2). J Am Coll Cardiol 2022;79:465-78. [Crossref] [PubMed]
- Jacobs JP, Kumar SR, St Louis JD, et al. Variation in Case-Mix Across Hospitals: Analysis of The Society of Thoracic Surgeons Congenital Heart Surgery Database. Ann Thorac Surg 2023;115:485-92. [Crossref] [PubMed]
- Vida VL, Zanotto L, Torlai Triglia L, et al. Surgery for Adult Patients with Congenital Heart Disease: Results from the European Database. J Clin Med 2020;9:2493. [Crossref] [PubMed]
- Limratana P, Maisat W, Tsai A, et al. Perioperative Factors and Radiographic Severity Scores for Predicting the Duration of Mechanical Ventilation After Arterial Switch Surgery. J Cardiothorac Vasc Anesth 2024;38:992-1005. [Crossref] [PubMed]
- Woo JL, Laternser C, Anderson BR, et al. Association Between Prenatal Diagnosis and Age at Surgery for Noncritical and Critical Congenital Heart Defects. Circ Cardiovasc Qual Outcomes 2023;16:e009638. [Crossref] [PubMed]
- Yoon YM, Bae SP, Kim YJ, et al. New modified version of the Risk Adjustment for Congenital Heart Surgery category and mortality in premature infants with critical congenital heart disease. Clin Exp Pediatr 2020;63:395-401. [Crossref] [PubMed]
- Kim M, Okunowo O, Ades AM, et al. Single-Center Comparison of Outcomes Following Cardiac Surgery in Low Birth Weight and Standard Birth Weight Neonates. J Pediatr 2021;238:161-167.e1. [Crossref] [PubMed]
- Bhamidipati CM, Garcia IC, Kim B, et al. Racial Disparity: The Adult Congenital Heart Disease Surgery Perspective. Pediatr Cardiol 2024;45:1275-83. [Crossref] [PubMed]
- Pivonello R, Auriemma RS, Pivonello C, et al. Sex Disparities in COVID-19 Severity and Outcome: Are Men Weaker or Women Stronger? Neuroendocrinology 2021;111:1066-85. [Crossref] [PubMed]
- Chlench S, Freudenthal NJ. Associations Between Clinical Factors and Postoperative Thrombosis in Pediatric Cardiac Surgery Patients: A Single-Center Retrospective Study. Crit Care Explor 2024;6:e1170. [Crossref] [PubMed]
- Inoue AS, Lopes AAB, Tanaka ACS, et al. Impact of Preoperative Functional Capacity on Postoperative Outcomes in Congenital Heart Surgery: An Observational and Prospective Study. Arq Bras Cardiol 2022;118:411-9. [Crossref] [PubMed]
- Toale C, Kavanagh DO, Devine M, et al. High-stakes Technical Performance Assessments Across the Continuum of Surgical Training. Ann Surg 2025;282:56-62. [Crossref] [PubMed]
- Aşır A, Aldudak B, Matur Okur N. The Impact of Postoperative Albumin Levels on Furosemide Efficacy in Infants with Congenital Heart Disease. Life (Basel) 2024;14:1679. [Crossref] [PubMed]
- Srichuachom W, Krintratun S, Chenthanakij B, et al. Prevalence and outcomes of hypocalcemia on ED arrival in traumatic patients before blood transfusions: a systematic review and meta-analysis. Scand J Trauma Resusc Emerg Med 2025;33:43. [Crossref] [PubMed]
- Weisert M, Menteer J, Durazo-Arvizu R, et al. Early prediction of failure to progress in single ventricle palliation: A step toward personalizing care for severe congenital heart disease. J Heart Lung Transplant 2022;41:1268-76. [Crossref] [PubMed]