Structural and temporal dynamics of pediatric influenza prevention and treatment: a comprehensive bibliometric analysis of historical evolution and emerging trends
Highlight box
Key findings
• This comprehensive bibliometric analysis identified three distinct developmental phases in pediatric influenza research. Persistent foundational themes include live attenuated vaccines and neuraminidase, while current cutting-edge frontiers emphasize respiratory syncytial virus (RSV) co-infections, novel antivirals (e.g., baloxavir), and strategies to optimize immunization.
What is known and what is new?
• Children bear a disproportionately high global burden of seasonal and pandemic influenza, facing significant risks of severe respiratory complications.
• By mapping 30 years of literature, this study reveals a critical paradigm shift in the field—moving from early foundational prevention to tackling complex modern challenges, including multi-pathogen co-circulation, changing immunization behaviors (vaccine hesitancy), and mechanisms of antiviral resistance.
What is the implication, and what should change now?
• The intersection of influenza with coronavirus disease 2019 and RSV, coupled with rising vaccine hesitancy and antiviral resistance, profoundly complicates pediatric clinical care.
• Clinical and public health practices must proactively adapt by integrating rapid multiplex diagnostics for co-infections, optimizing the use of novel endonuclease inhibitors, accelerating the development of broad-spectrum vaccines, and deploying targeted communication strategies to mitigate vaccine hesitancy.
Introduction
Influenza is an acute respiratory infection caused by influenza viruses (IV), triggering seasonal epidemics worldwide with significant morbidity and mortality rates (1). Approximately one billion people contract seasonal influenza annually, with 3–5 million severe cases (2). Children are more susceptible due to their immature immune systems and face higher risks of complications (3). Global data indicate that influenza accounts for 10% of respiratory hospitalizations among children under 18 years old (4), with those under 5 years old and children with underlying medical conditions facing particularly high risks of severe illness and death (5-7). In 2018, approximately 109.5 million influenza cases occurred among children under 5 globally. Of these, 10 million progressed to acute lower respiratory infections (ALRI), resulting in 870,000 hospitalizations and 35,000 deaths (8).
Vaccination remains the most effective preventive measure currently available, significantly reducing influenza incidence, hospitalization, and mortality risks (9). The American Academy of Pediatrics (AAP) recommends annual vaccination for all children aged ≥6 months (5). However, the virus’s high mutation rate necessitates continuous vaccine updates, posing challenges for both development and supply (10,11). Furthermore, insufficient vaccine coverage in low- and middle-income countries due to economic and resource constraints means that children continue to bear a heavy influenza burden (12,13). Antiviral drugs such as neuraminidase (NA) inhibitors (oseltamivir) and ribonucleic acid (RNA) polymerase inhibitors (baloxavir marboxil) are essential components of treatment (14), but drug resistance driven by rapid viral mutation threatens their efficacy (15).
In recent years, extensive research has focused on the prevention and control of influenza in children. Bibliometric analysis can objectively reveal trends and hotspots in this field. This study is designed as a comprehensive bibliometric analysis to contextualize the changing research interests in this field. Rather than serving as a rigid analytical framework, bibliometric tools (CiteSpace, HistCite Pro 2.1, and Alluvial Builder) are utilized as guideposts to systematically organize and visualize the historical evolution and structural dynamics of the literature. This methodological positioning allows for interpretative flexibility, enabling us to deeply synthesize research hotspots, acknowledge potential biases in literature selection, and engage in an extrapolative discussion regarding emerging trends. Ultimately, this review aims to summarize historical characteristics, identify research hotspots and emerging directions, and provide evidence for prevention and control practices as well as policy formulation. We present this article in accordance with the BIBLIO reporting checklist (available at https://tp.amegroups.com/article/view/10.21037/tp-2026-1-0086/rc).
Methods
Data collection
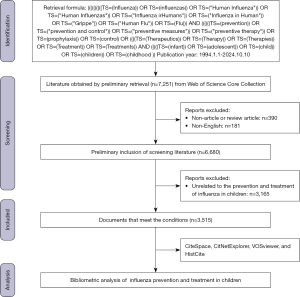
This study combines quantitative bibliometric visualization with qualitative literature synthesis. To establish a structured sample of the literature, we selected the Web of Science Core Collection (WoSCC) as the primary database. Although databases such as Scopus and PubMed offer broader coverage, WoSCC was exclusively chosen because it provides the standardized and comprehensive citation metadata (including full cited references) strictly required by advanced bibliometric software (e.g., CiteSpace and HistCite). Merging multiple databases often results in data format incompatibilities and loss of critical citation linkages, which can severely compromise the integrity of co-citation network analyses. Accordingly, we performed a systematic search in WoSCC using the following search criteria for the period 1994–2024: ((((((((((TS=(Influenza)) OR TS=(influenzas)) OR TS=(“Human Influenza”)) OR TS=(“Human Influenzas”)) OR TS=(“Influenza irHumans”)) OR TS=(“Influenza in Human”)) OR TS=(“Grippe”)) OR TS=(“Human Flu”)) OR TS=(Flu))) AND ((((((TS=(prevention)) OR TS=(“prevention and control”)) OR TS=(“preventive measures”)) OR TS=(“preventive therapy”)) OR TS=(prophylaxis)) OR TS=(control) OR ((((TS=(Therapeutics)) OR TS=(Therapy)) OR TS=(Therapies)) OR TS=(Treatment)) OR TS=(Treatments)) AND (((((TS=(infant)) OR TS=(adolescent)) OR TS=(child)) OR TS=(children)) OR TS=(childhood)). The retrieved literature records were then downloaded and saved as plain text files as samples of the data analyzed in this paper in the format of “Full Record and Cited References”. Subsequently, non-English literature, non-article, and non-review article literature were excluded. Then, by reading the titles and abstracts, literature unrelated to the prevention and treatment of influenza in children was further excluded. Finally, a total of 3,515 articles were collected and named DATA. Meanwhile, we collected raw data such as country/region, organization, journal, author, article type of publication, etc. The literature screening and analysis process is shown in Figure 1.
Bibliometric analysis tools
This study employs bibliometric tools as guideposts to identify key thematic shifts, core literature, and academic networks, thereby structurally scaffolding our in-depth qualitative synthesis. Rather than being confined to mere numbers, this approach grants us the interpretative flexibility to deeply synthesize research findings. Ultimately, the quantitative data serve as an objective starting point, enabling a richer and more extrapolative discussion of emerging trends and future policies.
CiteSpace
Co-occurrence networks
Scientific collaboration manifests as multiple authors, institutions, or countries jointly contributing to the same publication, holding significant importance for understanding field development dynamics. Collaboration is typically analyzed across three dimensions: authors, institutions, and countries. By importing relevant literature data into CiteSpace, co-occurrence networks can be constructed to systematically visualize collaborative and conceptual relationships. The software uses color to distinguish nodes and edges: edge colors represent the year the co-occurrence relationship was first established, while nodes are depicted with multi-colored “annual rings”, where ring thickness reflects co-occurrence frequency in a specific year. Red rings indicate citation bursts, while purple rings denote nodes with high intermediary centrality. The latter serve as critical hubs connecting different parts of the network, holding significant structural importance.
Burst detection
Jon Kleinberg noted that topics within literature streams often emerge intensely over short periods before fading away. This temporal pattern can be identified through text mining as an “activity outbreak” (16). CiteSpace supports emergence detection across disciplines, keywords, and references, where pronounced citation surges indicate that related topics have garnered widespread attention within the scientific community (17,18).
Cluster analysis
Data from the pediatric influenza prevention and control field were imported into the software (version 6.2.R4), with the time range set to 2002–2021 and a one-year interval between slices. Select “Title”, “Abstract”, “Author Keywords (DE)”, and “Keywords+” as term sources. Set node types according to analysis objectives, leaving other parameters at default values. Automatically generate network maps of countries, institutions, or author collaborations, then manually adjust and optimize visualization effects. The keyword clustering graph follows a similar construction method, with node type set to “Keywords” and time divided into five intervals: 1994–2000, 2001–2006, 2007–2012, 2013–2018, and 2019–2024. For co-citation maps, select “References” nodes and enable “Timeline View” in the “Layout” tab to generate a timeline visualization. Utilize the “View” function in the “Burstness” tab to detect and generate burstness maps for keywords, categories, or references.
HisCite
HistCite Pro 2.1 analyzes citation relationships and identifies highly cited literature within a field, visualizing the temporal evolution and knowledge accumulation of significant publications. The software employs two citation metrics: local citation score (LCS, internal citation frequency within the dataset) and global citation score (GCS, total citation count in the WoSCC). This study imported 3,515 pediatric influenza prevention and treatment publications into the tool, setting “Limit” to 30 with default parameters. The “Make Chart” function generated a research map to rapidly identify key publications.
Alluvial generator
Alluvial Generator visualizes the temporal evolution of research topics, collaborations, and citation trends through streaming graphs, enabling tracking of shifting research foci and disciplinary intersections (19). The specific workflow involves exporting time-segmented keyword co-occurrence networks from CiteSpace and importing them into the tool’s online platform (http://www.mapequation.org/apps/AlluvialGenerator.html). Keywords serve as nodes, clustered into modules by time period. Nodes split or merge into new modules over time, with the latest modules formed from the intersection of previous nodes.
Statistical analysis
Descriptive statistical analyses of the publication metadata, including annual publication volume, institutional contributions, and journal distribution, were systematically performed using Microsoft Excel 2019. Visual representations of categorical data (e.g., the donut plot) were generated using R software (version 4.2.2) with the ggplot2 package (version 3.4.4). For the bibliometric network analyses, quantitative metrics such as the LCS and GCS were calculated using HistCite Pro 2.1. Furthermore, CiteSpace was utilized to compute betweenness centrality to identify critical network hubs, and Kleinberg’s burst detection algorithm was applied to statistically quantify citation burst strength, identifying sudden surges in research interest over specific periods. As this study is based on bibliometric literature data rather than human subject or clinical trial data, traditional inferential statistics (e.g., P values) were not applicable; all quantitative findings are presented as descriptive statistics and network metrics.
Results
Historical features of the literature on influenza prevention and treatment
Distribution of publications
A total of 3,515 documents associated with the prevention and treatment of influenza in children were retrieved, comprising 3,112 research articles and 403 review articles. These publications involved 17,906 authors and 4,774 organizations, and were disseminated across 731 journals in 93 scientific categories (Table 1).
Table 1
| Categories | Amount |
|---|---|
| Publication | 3,515 |
| Articles | 3,112 |
| Review | 403 |
| Authors | 17,906 |
| Institutions | 4,774 |
| Journals | 731 |
| Subject categories | 93 |
The annual research results are shown in Figure 2A. From 1994 to 1999, the publication volume remained relatively low. However, from 2000 to 2011, there was a sharp increase in the number of publications, followed by a slight decline after 2012.

Vaccine ranked first in terms of the number of publications [290], followed by Pediatric Infectious Disease Journal [155] and PLoS One [133]. Figure 2B presents the top 20 most prolific journals, serving as a reference for researchers when making decisions regarding manuscript submission.
The vein of research on pediatric influenza prevention and treatment
The citation network diagram in Figure 3 reveals extensive connections among research in this field over the past two decades, comprising 1,787 nodes and 8,304 links. The overall structure evolves in a tree-like pattern: early-stage (1994–2010) gray nodes exhibit high density and extensive connections, forming the field’s foundation; mid-stage (2011–2017) blue nodes gradually spread to form main branches; while the recent period (2018–2024) shows nodes clustering into groups, reflecting a trend toward concentrated and specialized research themes. Key influential studies include Dawood FS (2009), Jain S (2009), and Poehling KA (2006), with co-citation frequencies of 125, 111, and 94, respectively. The convergence and differentiation of research clusters will be further elucidated in subsequent timeline diagrams. We additionally employed HisCite Pro 2.1 to plot citation history trajectories, with significant publications summarized in Table 2. Among these, the top three most influential include: mortality associated with influenza and respiratory syncytial virus (RSV) in the United States, the efficacy of live attenuated, cold-adapted, trivalent, intranasal influenza virus vaccine in children, and influenza-associated hospitalizations in the United States.

Table 2
| No. | Article information | Journal | LCS | GCS |
|---|---|---|---|---|
| 264 | Mortality associated with influenza and respiratory syncytial virus in the United States (20) | JAMA-J Am Med Assoc | 196 | 2,873 |
| 68 | The efficacy of live attenuated, cold-adapted, trivalent, intranasal influenzavirus vaccine in children (21) | New Engl J Med | 195 | 707 |
| 349 | Influenza-associated hospitalizations in the United States (22) | JAMA-J Am Med Assoc | 192 | 1,729 |
| 177 | Oral oseltamivir treatment of influenza in children (23) | Pediatr Infect Dis J | 180 | 505 |
| 415 | Influenza-associated deaths among children in the United States, 2003-2004 (24) | New Engl J Med | 143 | 487 |
| 183 | The Japanese experience with vaccinating schoolchildren against influenza (25) | New Engl J Med | 140 | 624 |
| 802 | Hospitalized Patients with 2009 H1N1 Influenza in the United States, April-June 2009 (26) | New Engl J Med | 134 | 1,313 |
| 222 | Burden of interpandemic influenza in children younger than 5 years: A 25-year prospective study (27) | J Infect Dis | 132 | 374 |
| 138 | Efficacy and safety of oseltamivir in treatment of acute influenza: a randomised controlled trial (28) | Lancet | 126 | 584 |
| 1303 | Efficacy and effectiveness of influenza vaccines: a systematic review and meta-analysis (29) | Lancet Infect Dis | 116 | 1,353 |
| 113 | Efficacy of vaccination with live attenuated, cold-adapted, trivalent, intranasal influenza virus vaccine against a variant (A/Sydney) not contained in the vaccine (30) | J Pediatr-US | 108 | 340 |
| 343 | Resistant influenza A viruses in children treated with oseltarnivir: descriptive study (31) | Lancet | 84 | 608 |
| 151 | Effectiveness of influenza vaccination of day care children in reducing influenza-related morbidity among household contacts (32) | JAMA-J Am Med Assoc | 78 | 231 |
| 293 | Effectiveness of inactivated influenza vaccine in preventing acute otitis media in young children - A randomized controlled trial (33) | JAMA-J Am Med Assoc | 77 | 278 |
| 4 | A randomized controlled trial of cold-adapted and inactivated vaccines for the prevention of influenza a disease (34) | J Infect Dis | 77 | 199 |
| 875 | Effect of Influenza Vaccination of Children on Infection Rates in Hutterite Communities A Randomized Trial (35) | JAMA-J Am Med Assoc | 75 | 279 |
| 137 | Zanamivir for treatment of symptomatic influenza A and B infection in children five to twelve years of age: a randomized controlled trial (36) | Pediatr Infect Dis J | 73 | 162 |
| 472 | Comparison of the efficacy and safety of live attenuated cold-adapted influenza vaccine, trivalent, with trivalent inactivated influenza virus vaccine in children and adolescents with asthma (37) | Pediatr Infect Dis J | 72 | 208 |
| 286 | Impact of oseltamivir treatment on influenza-related lower respiratory tract complications and hospitalizations (38) | Arch Intern Med | 71 | 317 |
| 178 | Effectiveness of oseltamivir in preventing influenza in household contacts - A randomized controlled trial (39) | JAMA-J Am Med Assoc | 71 | 387 |
| 735 | Seasonal Influenza in Adults and Children-Diagnosis, Treatment, Chemoprophylaxis, and Institutional Outbreak Management: Clinical Practice Guidelines of the Infectious Diseases Society of America (40) | Clin Infect Dis | 68 | 238 |
| 496 | Effectiveness of school-based influenza vaccination (41) | New Engl J Med | 68 | 490 |
| 22 | Influenza-A vaccine decreases the incidence of otitis-media in 6- to 30-month-old children in day-care (42) | Arch Pediat Adol Med | 67 | 209 |
| 196 | Efficacy of inactivated and cold-adapted vaccines against influenza A infection, 1985 to 1990: the pediatric experience (43) | Pediatr Infect Dis J | 67 | 176 |
| 992 | Early Oseltamivir Treatment of Influenza in Children 1-3 Years of Age: A Randomized Controlled Trial (44) | Clin Infect Dis | 64 | 132 |
| 495 | Safety, efficacy, and effectiveness of cold-adapted influenza vaccine-trivalent against community-acquired, culture-confirmed influenza in young children attending day care (45) | Pediatrics | 60 | 124 |
| 799 | Factors Associated With Death or Hospitalization Due to Pandemic 2009 Influenza A (H1N1) Infection in California (46) | JAMA-J Am Med Assoc | 59 | 258 |
| 160 | Inhaled zanamivir for the prevention of influenza in families (47) | New Engl J Med | 59 | 794 |
| 395 | Effectiveness of the 2003-2004 influenza vaccine among children 6 months to 8 years of age, with 1 vs 2 doses (48) | Pediatrics | 58 | 135 |
| 295 | Socioeconomic impact of influenza on healthy children and their families (49) | Pediatr Infect Dis J | 58 | 139 |
No., number of documents in the database imported into Hiscite pro 2.1. GCS, global citation score; LCS, local citation score.
Scientific cooperation
As shown in Figure 4, the National Collaboration Network (Figure 4A) comprises 137 nodes and 1,706 connections, with the United States occupying a central position. Nodes representing England, Italy, Japan, and China are larger and colored more vividly, indicating their recent prominent contributions. Nodes such as Germany and France, though smaller, remain influential. The Institutional Collaboration Network (Figure 4B) comprises 575 nodes and 964 connections, with core institutions including the Centers for Disease Control and Prevention (CDC)-USA, the University of California System, and Harvard University. The University of Colorado Anschutz Medical Campus occupies a central and large position, while nodes such as Johns Hopkins University and Baylor College of Medicine are large and new, indicating high recent activity. The author collaboration network (Figure 4C) shows Esposito, Susanna and Fry, Alicia M. as leading authors in publications. Nodes such as Fry, Alicia M. and Ambrose, Christopher S. are large and new, with significant recent contributions. Multiple collaborative clusters have formed among authors, such as the group comprising Fry, Alicia M. and Finelli, Lyn, and another group including Ambrose, Christopher S., Belongia, Edward A., and Monto, Arnold S., reflecting a trend toward clustered collaboration. Frequently collaborating countries, institutions, and authors are detailed in Table S1.

Variation of the most active topics
Subject category burst
From 2000 to 2024, citation surges occurred in 108 out of 121 relevant disciplines, indicating significant shifts in research focus. Figure 5 displays the top 50 disciplines by emergence intensity and their temporal distribution. “Allergy” exhibited the highest emergence intensity (8.23, 1997–2007), followed by “Medicine, General & Internal” (6.53, 1994–2009). Emergence patterns diversified across disciplines, exemplified by “Dermatology” (1999–2005), “Gastroenterology & Hepatology” (2007–2012), and “Biotechnology & Applied Microbiology” (2012–2013). Twenty emerging disciplines emerged in 2024 (see Table S2), with “Chemistry, Analytical”, “Environmental Sciences”, and “Electrochemistry” ranking as the top three, reflecting the growing influence of interdisciplinary research in the field of pediatric influenza.

Keywords burst
During the study period (1994–2024), a total of 720 keywords exhibited emergence activity, with the top 50 in emergence intensity shown in Figure 6. “Seasonal influenza” demonstrated the highest emergence intensity (32.47, 2016–2024), indicating sustained attention. Early keywords such as “virus” (21.46, 1994–2010) and “efficacy” (20.82, 1997–2007) also exhibited significant emergence. In 2024, 20 keywords remain in an emergent state, indicating potential research hotspots. These include “baloxavir marboxil” (11.03, 2019–2024), “influenza” (10.1, 2021–2024), and “China” (7.23, 2020–2024), with the latter potentially linked to the COVID-19 pandemic.

A total of 1,885 publications exhibiting citation salience were identified, with the 30 most frequently cited articles from 1994 to 2024 listed in Figure 7.

Belshe RB (1998) demonstrated the highest citation impact (40.84, 1999–2003), with this multicenter randomized controlled trial confirming the safety and efficacy of cold-adapted trivalent live attenuated influenza vaccine in young children, achieving 93% protection (21); Neuzil KM (2000) (40.56, 2000–2005) quantified the influenza disease burden in infants and young children using Medicaid data, revealing a negative correlation between age and hospitalization risk (50); Izurieta HS (2000) (35.56) emphasized significantly increased hospitalization risk during influenza season among healthy children under 2 years old, supporting routine immunization for this group (51).
In 2024, 98 new emerging literature pieces appeared, with the top 20 in emergence intensity (including 3 reviews and 17 research papers) listed in Table 3. These publications rapidly garnered attention in the year of publication or the following year, significantly advancing pediatric influenza prevention and control research and potentially indicating future research directions.
Table 3
| Title | Begin | End | Strength | Year | Type |
|---|---|---|---|---|---|
| Estimates of global seasonal influenza-associated respiratory mortality: a modelling study (52) | 2019 | 2024 | 34.79 | 2018 | Article |
| Baloxavir Marboxil for Uncomplicated Influenza in Adults and Adolescents (53) | 2019 | 2024 | 26.79 | 2018 | Article |
| Efficacy and Safety of Oseltamivir in Children: Systematic Review and Individual Patient Data Meta-analysis of Randomized Controlled Trials (54) | 2019 | 2024 | 20.87 | 2018 | Article |
| Estimates of the global, regional, and national morbidity, mortality, and etiologies of lower respiratory infections in 195 countries, 1990-2016: a systematic analysis for the Global Burden of Disease Study 2016 (55) | 2020 | 2024 | 7.86 | 2018 | Article |
| Clinical Practice Guidelines by the Infectious Diseases Society of America: 2018 Update on Diagnosis, Treatment, Chemoprophylaxis, and Institutional Outbreak Management of Seasonal Influenza (56) | 2020 | 2024 | 18.26 | 2019 | Article |
| Causes of severe pneumonia requiring hospital admission in children without HIV infection from Africa and Asia: the PERCH multi-country case-control study (57) | 2020 | 2024 | 15.52 | 2019 | Article |
| Global burden of respiratory infections associated with seasonal influenza in children under 5 years in 2018: a systematic review and modelling study (8) | 2020 | 2024 | 22.75 | 2020 | Review |
| Baloxavir Marboxil in Japanese Pediatric Patients With Influenza: Safety and Clinical and Virologic Outcomes (58) | 2020 | 2024 | 13.68 | 2020 | Article |
| Baloxavir Marboxil Single-dose Treatment in Influenza-infected Children A Randomized, Double-blind, Active Controlled Phase 3 Safety and Efficacy Trial (miniSTONE-2) (59) | 2020 | 2024 | 11.43 | 2020 | Article |
| Influenza-Associated Pediatric Deaths in the United States, 2010–2016 (60) | 2021 | 2024 | 9.86 | 2018 | Article |
| Prevention and Control of Seasonal Influenza with Vaccines: Recommendations of the Advisory Committee on Immunization Practices - United States, 2020-2021 Influenza Season (61) | 2021 | 2024 | 9.02 | 2019 | Article |
| Influenza vaccine effectiveness against influenza-associated hospitalization in children: A systematic review and meta-analysis (62) | 2021 | 2024 | 6.86 | 2019 | Review |
| Global patterns in monthly activity of influenza virus, respiratory syncytial virus, parainfluenza virus, and metapneumovirus: a systematic analysis (63) | 2021 | 2024 | 7.21 | 2020 | Article |
| Annual estimates of the burden of seasonal influenza in the United States: A tool for strengthening influenza surveillance and preparedness (64) | 2021 | 2024 | 11.63 | 2020 | Article |
| Impact of COVID-19 outbreaks and interventions on influenza in China and the United States (65) | 2022 | 2024 | 6.61 | 2019 | Article |
| Etiological and epidemiological features of acute respiratory infections in China (66) | 2022 | 2024 | 6.61 | 2019 | Article |
| Characterization of influenza virus variants induced by treatment with the endonuclease inhibitor baloxavir marboxil (67) | 2023 | 2024 | 10.25 | 2018 | Article |
| Treatment-Emergent Influenza Variant Viruses With Reduced Baloxavir Susceptibility: Impact on Clinical and Virologic Outcomes in Uncomplicated Influenza (68) | 2023 | 2024 | 7.83 | 2019 | Article |
| Influenza A(H3N2) virus exhibiting reduced susceptibility to baloxavir due to a polymerase acidic subunit I38T substitution detected from a hospitalised child without prior baloxavir treatment, Japan, January 2019 (69) | 2023 | 2024 | 6.86 | 2021 | Article |
| Clinical Practice Guidelines by the Infectious Diseases Society of America: 2018 Update on Diagnosis, Treatment, Chemoprophylaxis, and Institutional Outbreak Management of Seasonal Influenza (56) | 2023 | 2024 | 6.86 | 2021 | Review |
Begin, the year when the citation burst started. End, the year when the citation burst ended. Strength, the strength of the citation burst, the higher the value, the higher the citation growth during the period. Year, the year the document was published. The literature is first sorted by outbreak start year; if the outbreak start year is the same, it is then sorted by publication year.
Emerging trends and new developments
The temporal variation in keyword clusters
Keyword clustering analysis from 1994 to 2024 reveals the evolution of research themes (Figure 8). Studies were divided into five 6-year phases, with the following cluster structures for each: 1994–2000 (169 papers): 8 clusters, primarily comprising #0 “adults”, #1 “a virus,” and #2 “infection” (Figure 8A). 2001–2006 (328 articles): 8 clusters dominated by #0 “influenza vaccine,” #1 “zanamivir,” and #2 “infant” (Figure 8B). 2007–2012 (991 articles): 7 clusters, including #0 “live attenuated influenza vaccine”, #1 “zanamivir”, and #2 “tolerability” (Figure 8C). 2013–2018 (1,047 articles): 6 clusters, dominated by #0 “respiratory syncytial virus”, #1 “influenza A”, and #2 “infection” (Figure 8D). 2019–2024 (980 articles): 8 clusters, e.g., #0 “respiratory syncytial virus”, #1 “influenza A”, and #2 “immunization” (Figure 8E).

Compared to the preceding 15 years, classic research topics such as RSV and influenza vaccines remain prominent research hotspots. However, emerging research clusters—such as #1 “influenza A”, #2 “immunization”, #3 “oseltamivir”, #4 “antibody”, #6 “respiratory pathogens”, and #7 “retrospective cohort”—are increasingly attracting researchers’ attention. Cluster #1 “influenza A” contains 54 articles focused on this specific virus. Clusters #2 “immunization”, #3 “oseltamivir”, and #4 “antibody” comprise 46, 41, and 35 articles, respectively, detailing approaches to treating pediatric influenza. Cluster #6 “respiratory pathogens” includes 29 articles addressing influenza pathogenesis, while cluster #7 “retrospective cohort” contains 9 articles related to large-scale retrospective cohort studies. Detailed data for 2019–2024 can be found in Table S3. Representative keywords within these clusters help identify core research areas in pediatric influenza prevention and treatment during the recent period (2019–2024).
The keyword alluvial flow visualization
As illustrated in Figure 9, associated keywords dynamically reorganized into specific modules, with their formation and dissolution driving the emergence of new research domains. Keywords such as “immunization,” “influenza pandemic”, and “children’s health” remained consistently active during the early period (1994–2000), reflecting sustained attention to vaccination and public health. Meanwhile, terms like “bacteria” gradually receded, indicating a shift in focus toward more pressing issues such as viral infections and novel vaccines. While the visual extremes highlight the historical baseline and current frontiers, the intermediate transitional shifts are fully detailed in available online: https://cdn.amegroups.cn/static/public/tp-2026-1-0086-1.xlsx.

The six major keyword clusters in 2024 (Figure 10) further reveal research frontiers, carrying distinct clinical significance for pediatric practice. For instance, Module 1 (“young children”; Figure 10A), which encompasses keywords like “live attenuated influenza vaccine” and “neuraminidase”, clinically highlights the tailored preventive and therapeutic strategies strictly required for this highly vulnerable demographic, emphasizing age-specific immune responses and accurate antiviral dosing. In sharp contrast, Module 2 (“syncytial virus”; Figure 10B), featuring “clinical characteristics” and “epidemiology”, underscores the diagnostic challenges of viral co-circulation. Clinically, this module reflects the urgent real-world need for pediatricians to differentiate influenza from RSV and effectively manage complex co-infections during overlapping seasonal peaks. Building on these challenges, Module 3 (“COVID-19 pandemic”; Figure 10C), with keywords like “randomized trial” and “impact,” signifies the paradigm shift in managing pediatric respiratory infections post-2020. Clinically, it dictates the necessity to understand viral interference and rigorously evaluate the safety of co-administering influenza and severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) vaccines in children. Module 4 (“immunization”; Figure 10D) revolves around “cost-effectiveness”, “immunogenicity”, and “safety”, directing clinical attention toward the health-economic justification and biological optimization of novel pediatric vaccines, moving beyond basic efficacy to comprehensive clinical utility. Furthermore, Module 5 (“recommendations”; Figure 10E), featuring “vaccine effectiveness” and “advisory committee”, represents the crucial translation of real-world evidence into actionable clinical guidelines. It highlights how organizations [e.g., Advisory Committee on Immunization Practices (ACIP), World Health Organization (WHO)] continuously update annual pediatric vaccination strategies based on circulating drifted strains. Finally, Module 6 (“acute respiratory illness”; Figure 10F) includes “transmission”, “Hong Kong”, and “pandemic influenza”. Clinically, this underscores the vital importance of regional epidemiological surveillance hubs (e.g., Hong Kong as a historical sentinel) in predicting transmission dynamics, preparing pediatric hospital infrastructures, and optimizing infection control protocols to mitigate sudden pandemic surges.

The timeline visualization of references
Timeline visualization analysis identifies classic, emerging, and relatively outdated research themes by tracking citation trends. The visual map comprises 20 clusters (Figure 11A), with clusters #0 “pandemic”, #1 “immunogenicity”, #4 “asthma”, #5 “treatment”, #7 “oseltamivir”, #12 “transmission”, and #13 “adults” representing classic themes that remain closely linked to other studies and continue exerting significant academic influence. In contrast, clusters such as #8 “cost-effectiveness” and #10 “pneumococcal antibodies” exhibit lower activity and a declining research trend. Emerging themes include #2 “baloxavir”, #3 “vaccine effectiveness”, #6 “COVID-19”, #9 “pneumonia”, and #19 “beliefs”. These domains have maintained activity since their emergence, with increasing relevance, demonstrating potential to become future research hotspots. Table S4 provides more detailed information on these emerging clusters. To bridge these bibliometric findings with clinical practice, Table 4 provides a simplified translational summary of these 19 clusters. This table distills the dense bibliometric network into actionable clinical implications, offering pediatricians a clear historical context for current prevention and treatment guidelines.

Table 4
| Themes | Mean (year) | Cluster ID |
|---|---|---|
| Declining research themes | 1990–1997 | #8 cost-effectiveness, #10 pneumococcal antibodies, #14 transplantation, #16 combined paediatric vaccines, #17 covid-19 era, #18 national policies |
| Classic research themes | 1999–2009 | #0 pandemic, #1 immunogenicity, #4 asthma, #5 treatment, #7 oseltamivir, #11 respiratory tract infections, #12 transmission, #15 severe acute respiratory syndrome |
| Emerging research themes | 2013–2020 | #2 baloxavir, #3 vaccine effectiveness, #6 COVID-19, #9 pneumonia, #13 adults, #19 beliefs |
COVID-19, coronavirus disease 2019.
Furthermore, several seminal papers have played pivotal roles in advancing specific subfields (Figure 11B). For instance, Iuliano AD (2018) in cluster #2 (52) provided new global estimates of pediatric influenza mortality, offering critical evidence for optimizing prevention and control strategies; Grohskopf LA (2016) (70) in Cluster #3 summarized the latest vaccination guidelines from the ACIP. Olsen SJ (2020, Am J Transplant) (71) in Cluster #6 revealed that non-pharmaceutical interventions (NPIs) offer practical guidance for addressing vaccine hesitancy from a psychological perspective. In contrast, Lafond EK (2016) (72) in Cluster #9 estimated over 1.2 million annual hospitalizations among children under five due to influenza, with developing countries experiencing three times the hospitalization rate of industrialized nations, underscoring the need to expand vaccination coverage. Conversely, Brewer NT (2021) (73) in Cluster #19 explores psychological factors influencing vaccination behavior, offering guidance for addressing vaccine hesitancy. Citation distribution projections indicate (Figure 11C) that these key publications will maintain high academic attention in the coming years.
Discussion
Analysis of the fundamental characterization of publications on pediatric influenza prevention and treatment
Research in pediatric influenza prevention and control remains highly active, with publications having grown steadily and close international collaboration forming a complex partnership network. The diversity of research output highlights its multidisciplinary nature. Temporal analysis revealed three distinct phases in the development of this field. The initial phase (1994–1999) was characterized by a limited number of publications. From 2000 to 2011, there was a period of rapid growth, driven by multiple synergistic advancements. In 1999, the WHO released its first “Influenza Pandemic Preparedness Plan”, emphasizing the importance of strengthening influenza surveillance, enhancing emergency preparedness, and advancing the development of vaccines and antiviral therapies (74). The emergence of novel vaccines and antivirals, along with interdisciplinary collaborations, technological advancements, and growing awareness of the disease burden among high-risk groups such as children, catalyzed research efforts and resource investment in the field, leading to an increase in publications. Following the H1N1 pandemic in 2009, there was a sharp rise in pediatric mortality rates, prompting a global response with the development of guidelines, countermeasures, and extensive research. As a result, the volume of publications surged, peaking in 2011 (75,76). After 2012, although the publication rate slightly declined, the annual number of new articles consistently exceeded 123, reflecting the maturity of this field. Research continues to focus on antiviral therapies, vaccine efficacy, and public health strategies.
Persistent challenges and emerging topics in pediatric influenza prevention and treatment
Research on pediatric influenza prevention and treatment continues to evolve. While classic themes such as “live attenuated vaccine”, “neuraminidase”, and “immune response” remain crucial, emerging keywords like “seasonal influenza”, “baloxavir marboxil”, and “immunization practices” signal future research directions. Keyword clustering and timeline network analysis further reveal that “baloxavir”, “vaccine efficacy”, “COVID-19”, and “pneumonia” have emerged as new research hotspots. Increasing interdisciplinary participation highlights how this field is addressing persistent challenges while continuously generating novel research questions.
Exploration of hot topics
Public health control measures
The COVID-19 pandemic has reshaped the epidemiological patterns of childhood influenza. During the pandemic, widespread implementation of NPIs such as mask-wearing and social distancing not only effectively controlled SARS-CoV-2 transmission but also significantly reduced childhood influenza incidence (71,77,78). With the gradual lifting of NPIs, seasonal influenza activity has largely returned to pre-pandemic levels, except for the B/Yamagata lineage, though regional variations persist (79,80). This suggests that NPIs should be incorporated into routine influenza prevention and control strategies.
Vaccine hesitancy has led to low influenza vaccine coverage in children (81). As indicated by the keyword clustering module, “vaccine hesitancy”, “immunization”, and “coverage” have emerged as key research topics in this field. Despite the proven safety and efficacy of influenza vaccines in preventing childhood influenza, vaccination rates have been declining. The 2023–2024 season saw a child vaccination rate of only 55.4%, representing an 8.3% decrease compared to pre-pandemic levels (82). Vaccine hesitancy is listed by the WHO as a global health threat (83,84). Contributing factors include concerns about side effects/safety (85), low perceived disease severity, and limited confidence in vaccine effectiveness (86), socioeconomic factors (87), and limited public awareness (88). This issue is exacerbated in low-income regions due to inequitable resource distribution and limited healthcare access (12,89), necessitating urgent policy and immunization program interventions.
Mixed respiratory virus infections
In recent years, advances in multiplex respiratory pathogen detection technologies (90,91). have heightened attention on mixed infections in children. Keyword analysis reveals that “RSV”, “oseltamivir”, and “COVID-19 pandemic” emerged as prominent themes between 2019 and 2024, reflecting growing concern over viral co-infections. As a susceptible population, children face significantly higher risks of mixed infections compared to adults (92). Although NPIs reduced influenza and RSV transmission during the COVID-19 pandemic (80,93), these viruses have resurged following the relaxation of measures. Studies indicate that influenza incidence among children in northern China in 2023 has significantly increased compared to the previous 2 years (94), with disease severity approaching pre-pandemic levels (95), potentially linked to “immunological debt” (96).
IV shares similar transmission routes and seasonal activity overlap with RSV and SARS-CoV-2 (97), making co-infection difficult to avoid. However, their interactions are complex, potentially synergistic or antagonistic, and whether they exacerbate clinical severity remains controversial (98).
Regarding IV and RSV co-infection, existing conclusions are inconsistent: experimental studies indicate influenza A virus (IAV) can suppress RSV replication (99), resulting in a reduced viral load during subsequent RSV infection; however, this apparent virological suppression does not necessarily translate into clinical benefit, as IAV pre-infection induces pulmonary congestion and inflammatory injury, thereby compromising lung integrity and increasing susceptibility to secondary damage, ultimately leading to aggravated disease severity and increased mortality upon secondary RSV challenge (100). Epidemiological models suggest prior infection with one virus may provide short-term cross-protection (101). Conversely, evidence also exists that co-infection can generate hybrid virus particles (HVPs), enhancing viral spread and evolution while exacerbating lung injury and mortality (102,103). Infection sequence may influence outcomes, with RSV priming potentially conferring protection by stimulating interferon-γ and innate immunity (104).
Concurrent IV and SARS-CoV-2 infection exhibits marked synergistic effects, upregulating angiotensin-converting enzyme-2 (ACE2) expression and enhancing viral replication, leading to increased mechanical ventilation requirements and mortality risk (105-107). Although the global impact of COVID-19 is diminishing, the virus continues to circulate, with new variants emerging persistently. SARS-CoV-2 is likely to persist as a seasonal virus, similar to influenza, posing a long-term public health risk. Co-circulation of both viruses will exacerbate the healthcare burden and may influence vaccine and drug development strategies (108). Therefore, there is an urgent need for more in-depth epidemiological and mechanistic research, as well as the development of coordinated prevention and control strategies targeting multiple respiratory viruses.
Vaccine development and effectiveness
Vaccination is the core measure for preventing influenza in children. The primary seasonal vaccines used include intramuscularly administered inactivated vaccine (IIV), intranasally administered live attenuated influenza vaccine (LAIV), and intramuscularly administered recombinant vaccine (RIV) (109). LAIV induces both mucosal and systemic immunity, offering approximately 50% higher efficacy than IIV in protecting children aged 6–59 months (P<0.001), particularly against antigenically matched or drifted viruses (110). This effect is associated with local IgA responses and compartmentalized immunity (111). Painless vaccination also improves compliance, but is contraindicated in immunocompromised or asthmatic children (112).
To address declining protection due to antigenic drift and viral subtype diversity (11), current research focuses on antigen engineering, nanoparticle formulations, dose escalation, novel adjuvants, and multi-target strategies to broaden immune responses (113). Although the quadrivalent high-dose inactivated vaccine (IIV4-HD) is not approved for pediatric use, Phase II trials indicate that the 60 µg dose demonstrates superior immunogenicity and good safety in children (1,114).
Adjuvants are critical for enhancing vaccine efficacy. Those approved for human use include aluminum adjuvants, MF59, and AS03 (81). MF59, an oil-in-water adjuvant, is widely used in elderly populations and infants in some countries (115). Systematic reviews indicate it enhances immunogenicity and protective efficacy in unvaccinated children but provides limited benefit for previously vaccinated individuals (116). Novel adjuvants such as saponins and Toll-like receptor (TLR) agonists remain in clinical development (113).
Universal influenza vaccines represent an ideal solution. The NIAID 2018 initiative aims to achieve ≥75% symptomatic protection, immunity lasting over one year, and applicability across all age groups (117,118). Research strategies include targeting conserved regions [e.g., hemagglutinin (HA) stem, matrix protein 2 ectodomain (M2e), NA], eliciting T-cell responses [nucleoprotein (NP), matrix protein 1 (M1)], nanoparticle/virus-like particle (VLP) vaccines, nucleic acid vaccines, and viral vector vaccines (119). However, their immunological persistence and efficacy require further experimental validation.
Novel antiviral drugs
Keyword clustering analysis for 2019–2024 indicates that “oseltamivir” remains a prominent topic of interest, while keyword citation bursts reveal “baloxavir” as an emerging focus of research.
Clinical use and resistance of oseltamivir
Oseltamivir is the first-line antiviral drug for influenza in children aged 2 weeks and older (5). Systematic reviews indicate that prompt administration of oseltamivir shortens the illness duration by approximately 18 hours in children and reduces the risk of complications such as otitis media, with no significant adverse effects beyond mild gastrointestinal reactions (54). The AAP recommends early initiation of treatment to optimize efficacy. A study confirm that initiating treatment within 24 hours of hospitalization shortens hospital stays and reduces risks of intensive care unit (ICU) transfer, extracorporeal membrane oxygenation (ECMO) use, readmission, and mortality (14). However, widespread use has led to the emergence of resistant strains, particularly in H1N1 viruses. Resistance primarily arises from mutations in the NA active site, reducing binding affinity with the inhibitor. Immunocompromised patients are more susceptible to developing resistant mutations. This susceptibility is associated with prolonged viral replication and incomplete viral clearance under drug pressure, which increases the risk of severe illness and death (120). Furthermore, resistance rates are higher in patients with influenza-associated acute respiratory distress syndrome (ARDS). A French study showed that approximately 23% of H1N1pdm09-associated ARDS patients exhibited the NA-H275Y mutation, and the 28-day mortality rate was significantly higher in the group infected with the mutant strain (121).
Baloxavir marboxil: applications and limitations
Baloxavir, a novel polymerase acid endonuclease inhibitor, demonstrates broad-spectrum antiviral activity against influenza and is approved for children aged ≥5 years in multiple countries (5). It acts by inhibiting the cap-dependent endonuclease activity of the PA subunit of the viral polymerase complex, thereby blocking viral mRNA transcription at an early stage of replication (122). Clinical studies indicate that in uncomplicated influenza patients aged 12–64 years, a single dose demonstrates favorable safety, provides more effective symptom relief than placebo, and reduces viral load more potently than oseltamivir (53). Pediatric studies further confirm its good oral tolerability, rapid reduction in viral titers, and relief of acute symptoms, with adverse event rates comparable to oseltamivir (58,59). However, its application is constrained by emerging resistance, with reports of reduced IV susceptibility and resistant strains (53,68), particularly those associated with substitutions in the polymerase acidic protein (PA) subunit (e.g., I38 variants), which significantly reduce drug susceptibility (67,123), underscoring the urgent need to develop novel antiviral agents with lower resistance risks.
Exploration of novel antiviral drugs
To combat resistance and enhance therapeutic efficacy, the development of novel antiviral drugs is actively advancing, broadly classified into two categories: virus-directed drugs targeting viral components at different stages of the replication cycle, and host-directed drugs targeting host cell factors involved in IV replication and pathogenesis. HA inhibitors act at the early stage of infection by preventing receptor binding or inhibiting the low-Ph-induced conformational changes required for membrane fusion, thereby blocking viral entry into host cells. Additionally, receptor-modulating agents such as DAS181 (Fludase) cleave sialic acid receptors on host cells to prevent viral attachment. Polymerase complex inhibitors, including PA endonuclease inhibitors (e.g., baloxavir marboxil), PB1 nucleoside analogs (e.g., favipiravir), and PB2 cap-binding inhibitors (e.g., pimodivir), interfere with the cap-dependent transcription (“cap-snatching”) mechanism and RNA chain elongation, which are essential for viral mRNA synthesis in the nucleus. NP-targeting agents such as naproxen disrupt viral ribonucleoprotein (vRNP) complex assembly, nuclear trafficking, and genome replication, thereby impairing efficient virion production. In addition, host cell-directed therapies target critical host factors involved in viral replication, such as endosomal acidification, intracellular trafficking pathways, and host proteases required for HA activation, offering a complementary strategy with a potentially higher barrier to resistance. Beyond traditional inhibitors, proteolysis-targeting chimeras (PROTACs) represent a cutting-edge approach by inducing the ubiquitination and degradation of viral proteins (e.g., NA, HA, or PA), providing a more sustained antiviral effect. Notably, compared with viral-targeted therapies, host-directed strategies may confer a higher genetic barrier to resistance, although they require careful evaluation of safety and potential off-target effects (124,125). Future research should focus on optimizing mechanisms, improving pharmacokinetics, reducing resistance risks, and validating safety and efficacy through large-scale clinical trials to develop more effective pediatric influenza prevention and treatment strategies.
Future directions
Optimization of public health control strategies
During the COVID-19 pandemic, the widespread adoption of NPIs led to a significant decline in influenza incidence, highlighting the potential of NPIs for controlling influenza in a SARS-CoV-2 co-circulation context. Moderate community interventions can effectively slow influenza transmission, but further research is needed to balance NPIs intensity with socioeconomic activity to develop more scientifically grounded control strategies (126).
Furthermore, enhancing vaccination coverage and improving healthcare infrastructure remain critical priorities for influenza control. This requires increased public health investment to reduce vaccine hesitancy and improve access to antiviral medications, particularly in low- and middle-income countries. Annual increases in healthcare resources for children, alongside strengthened primary healthcare services and equitable resource distribution, are also essential to reduce disease burden and enhance pandemic preparedness (3).
Research on IV co-infection with multiple respiratory viruses
Influenza and other respiratory viruses share high similarity in transmission routes, susceptible populations, and seasonal patterns, making them prone to co-infection, though the underlying mechanisms remain unclear. The impact of such co-infections on disease severity and transmission dynamics remains controversial, and effective prevention and control strategies are lacking. The persistent circulation of SARS-CoV-2 in the post-pandemic era has further intensified the mixed transmission trend of influenza and its co-infections, complicating diagnosis, treatment, and prognosis while significantly increasing the public health burden (127). Future research should systematically conduct epidemiological investigations of co-infections to clarify their epidemiological characteristics and risk factors, deeply explore the mechanisms of viral interactions, and progressively establish a comprehensive prevention and control system for “multiple disease co-prevention”. This will provide a scientific basis for developing coordinated intervention strategies.
Advancing the development of novel vaccines and antiviral drugs
Development of next-generation vaccines
Efforts should be accelerated to develop universal influenza vaccines offering broad-spectrum and long-lasting protection, leveraging multiple vaccine platforms and technologies such as next-generation sequencing. Advances in virology, immunology, and vaccinology in recent years have brought this goal closer to reality. Future endeavors should focus on integrating multidisciplinary research, optimizing vaccine design and manufacturing processes, and advancing the clinical translation of vaccines with broad-spectrum and durable protective efficacy (117).
Antiviral drug development and combination therapy
To address drug resistance to existing antiviral drugs, research and development of new antiviral drugs targeting different mechanisms should be prioritized to avoid the limitations of single-target drugs. Several promising new antiviral candidates are currently undergoing clinical trials with the potential for future application. In addition to drug development, combination therapy represents a crucial direction for future influenza treatment (15). Personalized treatment strategies for critically ill and immunocompromised patients also enhance therapeutic efficacy. Future efforts should focus on further evaluating the efficacy and safety of combination strategies to advance their clinical translation and application.
Enhancing influenza surveillance with artificial intelligence (AI) and big data platforms
AI and big data analytics have significantly enhanced the accuracy and efficiency of influenza surveillance. By integrating multi-source data and modeling techniques, it is possible to systematically evaluate the impact of viral evolution, population immunity, and behavioral patterns on transmission, providing scientific evidence for vaccine deployment, NPIs, and resource allocation (126). AI also enables real-time monitoring of viral mutations and drug resistance changes, predicting epidemic trends and optimizing control strategies. Future efforts should strengthen multidisciplinary collaboration and enhance data platform development and application to support precision influenza prevention and control.
Translational outlook for pediatric clinical practice
To directly bridge our bibliometric findings with frontline clinical applications, Table 5 summarizes the top five emerging trends in pediatric influenza research and outlines their immediate translational implications for pediatricians. As the landscape of pediatric infectious diseases rapidly evolves, adapting clinical practices to encompass novel antivirals, multiplex diagnostics, and proactive communication strategies will be paramount for optimizing patient outcomes.
Table 5
| # | Emerging trend | Description | Immediate implications for pediatric clinical practice |
|---|---|---|---|
| 1 | Routine integration of non-pharmaceutical interventions | The pandemic has demonstrated that measures such as mask-wearing and social distancing are effective not only against COVID-19 but also in significantly reducing childhood influenza incidence | During peak influenza seasons or when facing threats from novel respiratory viruses, NPIs should be actively reconsidered and promoted in pediatric clinics, schools, and other congregate settings. They serve as a crucial complement to vaccination |
| 2 | Increasing complexity of mixed respiratory virus infections | The co-circulation of influenza, RSV, and SARS-CoV-2 increases the likelihood of co-infections. The interactions between these viruses can be synergistic or antagonistic, and their impact on disease severity remains unclear |
Indications for multiplex pathogen testing should be broadened, especially for severely ill children or those unresponsive to standard treatment. Detecting a single pathogen should not be the endpoint; clinicians must remain vigilant for the potential of co-infections to exacerbate illness |
| 3 | Vaccine hesitancy and declining coverage | Despite proven vaccine effectiveness, childhood influenza vaccination rates are declining globally, highlighted as a key research topic. This decline poses a significant public health threat | Pediatricians should proactively address “vaccine hesitancy” by engaging in patient, evidence-based communication with caregivers regarding vaccine safety and efficacy. Targeted educational interventions are needed to counter concerns about side effects and build trust |
| 4 | Application and challenges of novel antivirals | Newer antivirals like baloxavir, approved for pediatric use, offer potent viral load reduction. However, their clinical utility is constrained by the emergence of resistant strains, mirroring the challenge seen with oseltamivir | Clinicians must understand the indications, benefits, and limitations of both oseltamivir and Baloxavir. Treatment decisions should balance efficacy against resistance risks, avoiding overuse. Enhanced resistance monitoring is critical, particularly in high-risk groups like immunocompromised children |
| 5 | AI-enhanced surveillance and precision prevention | Integrating big data and Artificial Intelligence enables real-time monitoring of viral mutations, prediction of epidemic trends, and evaluation of control strategies | Public health decisions and clinical alerts will become more precise. Clinicians should leverage AI-driven surveillance data to understand locally circulating strains and resistance patterns, guiding empirical treatment choices and pandemic preparedness efforts |
AI, artificial intelligence; COVID-19, coronavirus disease 2019; NPI, non-pharmaceutical intervention; RSV, respiratory syncytial virus; SARS-CoV-2, severe acute respiratory syndrome coronavirus 2.
Although this study provides valuable insights into pediatric influenza, the following limitations of bibliometric analysis should be noted. First, reliance solely on the WoSCC database inherently introduces a profound English-language bias. While WoSCC captures high-impact international research, it often under-represents localized clinical guidelines, regional epidemiological reports, and public health policies published in native languages. Consequently, our bibliometric findings may disproportionately reflect Western or Anglophone paradigms. This bias specifically limits our ability to fully capture the diverse and highly effective regional public health strategies employed in Asia and non-English-speaking Europe. For instance, localized NPIs, specific national childhood immunization schedules, and the integration of traditional medicine are extensively documented in regional databases but are likely overlooked in this study. Therefore, while our study outlines global megatrends, the generalizability of these findings to culturally and regionally specific public health practices should be interpreted with caution. Second, bibliometric analysis mainly uses quantitative metrics, which can highlight research trends and collaboration networks but offer limited ability to assess the academic value and specific contributions of individual studies. Additionally, due to time lags, some recent studies may have low citation counts, potentially underestimating their impact. The inclusion of only published literature may also introduce publication bias. Lastly, while collaboration network analysis shows partnerships, it does not reflect their depth or quality, which could overstate the influence of certain authors or institutions.
To address these limitations, future studies should incorporate a wider range of data sources, such as PubMed, Scopus, and China National Knowledge Infrastructure (CNKI), and combine quantitative and qualitative methods. This approach will help provide a more complete understanding of research in pediatric influenza prevention and control, thereby supporting evidence-based policy and practice in this important public health field.
Conclusions
Research on pediatric influenza prevention and control has undergone three distinct phases, with current studies focusing on interactions with COVID-19, vaccine hesitancy, influenza vaccine efficacy, and antiviral drug resistance mechanisms. Future research should prioritize elucidating influenza mixed infection pathways, accelerating broad-spectrum vaccine development, and innovating antiviral treatment regimens. Concurrently, strengthening public health interventions and health communication mechanisms is crucial for enhancing global influenza prevention and control efficacy among children.
Acknowledgments
We would like to express our gratitude to the Web of Science Core Collection (WoSCC) database for providing the bibliometric data essential to this study. We also acknowledge the developers of CiteSpace software for creating the tool that enabled the visual analysis of the research. Additionally, we thank all peer reviewers and editorial staff for their valuable comments and guidance during the manuscript revision process.
Footnote
Reporting Checklist: The authors have completed the BIBLIO reporting checklist. Available at https://tp.amegroups.com/article/view/10.21037/tp-2026-1-0086/rc
Peer Review File: Available at https://tp.amegroups.com/article/view/10.21037/tp-2026-1-0086/prf
Funding: This study was supported by
Conflicts of Interest: All authors have completed the ICMJE uniform disclosure form (available at https://tp.amegroups.com/article/view/10.21037/tp-2026-1-0086/coif). The authors have no conflicts of interest to declare.
Ethical Statement: The authors are accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Open Access Statement: This is an Open Access article distributed in accordance with the Creative Commons Attribution-NonCommercial-NoDerivs 4.0 International License (CC BY-NC-ND 4.0), which permits the non-commercial replication and distribution of the article with the strict proviso that no changes or edits are made and the original work is properly cited (including links to both the formal publication through the relevant DOI and the license). See: https://creativecommons.org/licenses/by-nc-nd/4.0/.
References
- Uyeki TM. Influenza. Ann Intern Med 2017;167:ITC33-ITC48. [Crossref] [PubMed]
- Influenza (Seasonal). WHO fact sheet on influenza: includes key facts, definition, symptoms, transmission, seasonal epidemics, effects, prevention, WHO response. Available online: https://www.who.int/news-room/fact-sheets/detail/influenza-(seasonal)
- Ratre YK, Vishvakarma NK, Bhaskar LVKS, et al. Dynamic Propagation and Impact of Pandemic Influenza A (2009 H1N1) in Children: A Detailed Review. Curr Microbiol 2020;77:3809-20. [Crossref] [PubMed]
- PLOS Medicine Staff. Correction: Global Role and Burden of Influenza in Pediatric Respiratory Hospitalizations, 1982-2012: A Systematic Analysis. PLoS Med 2016;13:e1002060. [Crossref] [PubMed]
- Recommendations for Prevention and Control of Influenza in Children, 2024-2025: Policy Statement. Pediatrics. 2024;154:e2024068507.
- Villani L, D'Ambrosio F, Ricciardi R, et al. Seasonal influenza in children: Costs for the health system and society in Europe. Influenza Other Respir Viruses 2022;16:820-31. [Crossref] [PubMed]
- Zhang F, Wu X, Yu N, et al. Global burden of influenza lower respiratory tract infections in children younger than 5 years from 1990 to 2021. J Thorac Dis 2026;18:71. [Crossref] [PubMed]
- Wang X, Li Y, O'Brien KL, et al. Global burden of respiratory infections associated with seasonal influenza in children under 5 years in 2018: a systematic review and modelling study. Lancet Glob Health 2020;8:e497-510. [Crossref] [PubMed]
- Zeno EE, Nogareda F, Regan A, et al. Interim Effectiveness Estimates of 2024 Southern Hemisphere Influenza Vaccines in Preventing Influenza-Associated Hospitalization - REVELAC-i Network, Five South American Countries, March-July 2024. MMWR Morb Mortal Wkly Rep 2024;73:861-8. [Crossref] [PubMed]
- Liang Y. Pathogenicity and virulence of influenza. Virulence 2023;14:2223057. [Crossref] [PubMed]
- Tenforde MW, Kondor RJG, Chung JR, et al. Effect of Antigenic Drift on Influenza Vaccine Effectiveness in the United States-2019-2020. Clin Infect Dis 2021;73:e4244-50. [Crossref] [PubMed]
- Palache A, Rockman S, Taylor B, et al. Vaccine complacency and dose distribution inequities limit the benefits of seasonal influenza vaccination, despite a positive trend in use. Vaccine 2021;39:6081-7. [Crossref] [PubMed]
- Shapiro Ben David S, Vered S, Adler L, et al. Family matters: influenza vaccination uptake and associated factors in children aged 6 months-6 years. Front Public Health 2025;13:1704497. [Crossref] [PubMed]
- Walsh PS, Schnadower D, Zhang Y, et al. Association of Early Oseltamivir With Improved Outcomes in Hospitalized Children With Influenza, 2007-2020. JAMA Pediatr 2022;176:e223261. [Crossref] [PubMed]
- Sarker A, Gu Z, Mao L, et al. Influenza-existing drugs and treatment prospects. Eur J Med Chem 2022;232:114189. [Crossref] [PubMed]
- Kleinberg J. Bursty and hierarchical structure in streams. DATA Min Knowl Disc 2003;7:373-97.
- Chen C, Ibekwe-SanJuan F, Hou J. The Structure and Dynamics of Cocitation Clusters: A Multiple-Perspective Cocitation Analysis. J Am Soc Inf Sci Tec 2010;61:1386-409.
- Chen C, Dubin R, Kim MC. Emerging trends and new developments in regenerative medicine: a scientometric update (2000 - 2014). Expert Opin Biol Ther 2014;14:1295-317. [Crossref] [PubMed]
- Rosvall M, Bergstrom CT. Mapping change in large networks. PLoS One 2010;5:e8694. [Crossref] [PubMed]
- Thompson WW, Shay DK, Weintraub E, et al. Mortality associated with influenza and respiratory syncytial virus in the United States. JAMA 2003;289:179-86. [Crossref] [PubMed]
- Belshe RB, Mendelman PM, Treanor J, et al. The efficacy of live attenuated, cold-adapted, trivalent, intranasal influenzavirus vaccine in children. N Engl J Med 1998;338:1405-12. [Crossref] [PubMed]
- Thompson WW, Shay DK, Weintraub E, et al. Influenza-associated hospitalizations in the United States. JAMA 2004;292:1333-40. [Crossref] [PubMed]
- Whitley RJ, Hayden FG, Reisinger KS, et al. Oral oseltamivir treatment of influenza in children. Pediatr Infect Dis J 2001;20:127-33. [Crossref] [PubMed]
- Bhat N, Wright JG, Broder KR, et al. Influenza-associated deaths among children in the United States, 2003-2004. N Engl J Med 2005;353:2559-67. [Crossref] [PubMed]
- Reichert TA, Sugaya N, Fedson DS, et al. The Japanese experience with vaccinating schoolchildren against influenza. N Engl J Med 2001;344:889-96. [Crossref] [PubMed]
- Jain S, Kamimoto L, Bramley AM, et al. Hospitalized patients with 2009 H1N1 influenza in the United States, April-June 2009. N Engl J Med 2009;361:1935-44. [Crossref] [PubMed]
- Neuzil KM, Zhu Y, Griffin MR, et al. Burden of interpandemic influenza in children younger than 5 years: a 25-year prospective study. J Infect Dis 2002;185:147-52. [Crossref] [PubMed]
- Li X, Chen Z, Liang H, et al. Efficacy and Safety of Huashi Baidu Granules in the Treatment of Children Suffering from Influenza with Exterior-cold and Interior-heat Syndrome: A Multi-center, Randomized Controlled Trial Protocol. Altern Ther Health Med 2024;30:185-91.
- Osterholm MT, Kelley NS, Sommer A, et al. Efficacy and effectiveness of influenza vaccines: a systematic review and meta-analysis. Lancet Infect Dis 2012;12:36-44. [Crossref] [PubMed]
- Belshe RB, Gruber WC, Mendelman PM, et al. Efficacy of vaccination with live attenuated, cold-adapted, trivalent, intranasal influenza virus vaccine against a variant (A/Sydney) not contained in the vaccine. J Pediatr 2000;136:168-75. [Crossref] [PubMed]
- Kiso M, Mitamura K, Sakai-Tagawa Y, et al. Resistant influenza A viruses in children treated with oseltamivir: descriptive study. Lancet 2004;364:759-65. [Crossref] [PubMed]
- Hurwitz ES, Haber M, Chang A, et al. Effectiveness of influenza vaccination of day care children in reducing influenza-related morbidity among household contacts. JAMA 2000;284:1677-82. [Crossref] [PubMed]
- Hoberman A, Greenberg DP, Paradise JL, et al. Effectiveness of inactivated influenza vaccine in preventing acute otitis media in young children: a randomized controlled trial. JAMA 2003;290:1608-16. [Crossref] [PubMed]
- Edwards KM, Dupont WD, Westrich MK, et al. A randomized controlled trial of cold-adapted and inactivated vaccines for the prevention of influenza A disease. J Infect Dis 1994;169:68-76. [Crossref] [PubMed]
- Loeb M, Russell ML, Moss L, et al. Effect of influenza vaccination of children on infection rates in Hutterite communities: a randomized trial. JAMA 2010;303:943-50. [Crossref] [PubMed]
- Hedrick JA, Barzilai A, Behre U, et al. Zanamivir for treatment of symptomatic influenza A and B infection in children five to twelve years of age: a randomized controlled trial. Pediatr Infect Dis J 2000;19:410-7. [Crossref] [PubMed]
- Fleming DM, Crovari P, Wahn U, et al. Comparison of the efficacy and safety of live attenuated cold-adapted influenza vaccine, trivalent, with trivalent inactivated influenza virus vaccine in children and adolescents with asthma. Pediatr Infect Dis J 2006;25:860-9. [Crossref] [PubMed]
- Kaiser L, Wat C, Mills T, et al. Impact of oseltamivir treatment on influenza-related lower respiratory tract complications and hospitalizations. Arch Intern Med 2003;163:1667-72. [Crossref] [PubMed]
- Welliver R, Monto AS, Carewicz O, et al. Effectiveness of oseltamivir in preventing influenza in household contacts: a randomized controlled trial. JAMA 2001;285:748-54. [Crossref] [PubMed]
- Harper SA, Bradley JS, Englund JA, et al. Seasonal influenza in adults and children--diagnosis, treatment, chemoprophylaxis, and institutional outbreak management: clinical practice guidelines of the Infectious Diseases Society of America. Clin Infect Dis 2009;48:1003-32. [Crossref] [PubMed]
- King JC Jr, Stoddard JJ, Gaglani MJ, et al. Effectiveness of school-based influenza vaccination. N Engl J Med 2006;355:2523-32. [Crossref] [PubMed]
- Clements DA, Langdon L, Bland C, et al. Influenza A vaccine decreases the incidence of otitis media in 6- to 30-month-old children in day care. Arch Pediatr Adolesc Med 1995;149:1113-7. [Crossref] [PubMed]
- Neuzil KM, Dupont WD, Wright PF, et al. Efficacy of inactivated and cold-adapted vaccines against influenza A infection, 1985 to 1990: the pediatric experience. Pediatr Infect Dis J 2001;20:733-40. [Crossref] [PubMed]
- Heinonen S, Silvennoinen H, Lehtinen P, et al. Early oseltamivir treatment of influenza in children 1-3 years of age: a randomized controlled trial. Clin Infect Dis 2010;51:887-94. [Crossref] [PubMed]
- Vesikari T, Fleming DM, Aristegui JF, et al. Safety, efficacy, and effectiveness of cold-adapted influenza vaccine-trivalent against community-acquired, culture-confirmed influenza in young children attending day care. Pediatrics 2006;118:2298-312. [Crossref] [PubMed]
- Louie JK, Acosta M, Winter K, et al. Factors associated with death or hospitalization due to pandemic 2009 influenza A(H1N1) infection in California. JAMA 2009;302:1896-902. [Crossref] [PubMed]
- Hayden FG, Gubareva LV, Monto AS, et al. Inhaled zanamivir for the prevention of influenza in families. Zanamivir Family Study Group. N Engl J Med 2000;343:1282-9.
- Ritzwoller DP, Bridges CB, Shetterly S, et al. Effectiveness of the 2003-2004 influenza vaccine among children 6 months to 8 years of age, with 1 vs 2 doses. Pediatrics 2005;116:153-9. [Crossref] [PubMed]
- Principi N, Esposito S, Marchisio P, et al. Socioeconomic impact of influenza on healthy children and their families. Pediatr Infect Dis J 2003;22:S207-10. [Crossref] [PubMed]
- Neuzil KM, Mellen BG, Wright PF, et al. The effect of influenza on hospitalizations, outpatient visits, and courses of antibiotics in children. N Engl J Med 2000;342:225-31. [Crossref] [PubMed]
- Izurieta HS, Thompson WW, Kramarz P, et al. Influenza and the rates of hospitalization for respiratory disease among infants and young children. N Engl J Med 2000;342:232-9. [Crossref] [PubMed]
- Iuliano AD, Roguski KM, Chang HH, et al. Estimates of global seasonal influenza-associated respiratory mortality: a modelling study. Lancet 2018;391:1285-300. [Crossref] [PubMed]
- Hayden FG, Sugaya N, Hirotsu N, et al. Baloxavir Marboxil for Uncomplicated Influenza in Adults and Adolescents. N Engl J Med 2018;379:913-23. [Crossref] [PubMed]
- Malosh RE, Martin ET, Heikkinen T, et al. Efficacy and Safety of Oseltamivir in Children: Systematic Review and Individual Patient Data Meta-analysis of Randomized Controlled Trials. Clin Infect Dis 2018;66:1492-500. [Crossref] [PubMed]
- Estimates of the global, regional, and national morbidity, mortality, and aetiologies of lower respiratory infections in 195 countries, 1990-2016: a systematic analysis for the Global Burden of Disease Study 2016. Lancet Infect Dis. 2018;18:1191-210.
- Uyeki TM, Bernstein HH, Bradley JS, et al. Clinical Practice Guidelines by the Infectious Diseases Society of America: 2018 Update on Diagnosis, Treatment, Chemoprophylaxis, and Institutional Outbreak Management of Seasonal Influenzaa. Clin Infect Dis 2019;68:895-902. [Crossref] [PubMed]
- Pneumonia Etiology Research for Child Health (PERCH) Study Group. Causes of severe pneumonia requiring hospital admission in children without HIV infection from Africa and Asia: the PERCH multi-country case-control study. Lancet 2019;394:757-79. [Crossref] [PubMed]
- Hirotsu N, Sakaguchi H, Sato C, et al. Baloxavir Marboxil in Japanese Pediatric Patients With Influenza: Safety and Clinical and Virologic Outcomes. Clin Infect Dis 2020;71:971-81. [Crossref] [PubMed]
- Baker J, Block SL, Matharu B, et al. Baloxavir Marboxil Single-dose Treatment in Influenza-infected Children: A Randomized, Double-blind, Active Controlled Phase 3 Safety and Efficacy Trial (miniSTONE-2). Pediatr Infect Dis J 2020;39:700-5. [Crossref] [PubMed]
- Shang M, Blanton L, Brammer L, et al. Influenza-Associated Pediatric Deaths in the United States, 2010-2016. Pediatrics 2018;141:e20172918. [Crossref] [PubMed]
- Grohskopf LA, Alyanak E, Ferdinands JM, et al. Prevention and Control of Seasonal Influenza with Vaccines: Recommendations of the Advisory Committee on Immunization Practices, United States, 2021-22 Influenza Season. MMWR Recomm Rep 2021;70:1-28. [Crossref] [PubMed]
- Kalligeros M, Shehadeh F, Mylona EK, et al. Influenza vaccine effectiveness against influenza-associated hospitalization in children: A systematic review and meta-analysis. Vaccine 2020;38:2893-903. [Crossref] [PubMed]
- Li Y, Reeves RM, Wang X, et al. Global patterns in monthly activity of influenza virus, respiratory syncytial virus, parainfluenza virus, and metapneumovirus: a systematic analysis. Lancet Glob Health 2019;7:e1031-45. [Crossref] [PubMed]
- Rolfes MA, Foppa IM, Garg S, et al. Annual estimates of the burden of seasonal influenza in the United States: A tool for strengthening influenza surveillance and preparedness. Influenza Other Respir Viruses 2018;12:132-7. [Crossref] [PubMed]
- Feng L, Zhang T, Wang Q, et al. Impact of COVID-19 outbreaks and interventions on influenza in China and the United States. Nat Commun 2021;12:3249. [Crossref] [PubMed]
- Li ZJ, Zhang HY, Ren LL, et al. Etiological and epidemiological features of acute respiratory infections in China. Nat Commun 2021;12:5026. [Crossref] [PubMed]
- Omoto S, Speranzini V, Hashimoto T, et al. Characterization of influenza virus variants induced by treatment with the endonuclease inhibitor baloxavir marboxil. Sci Rep 2018;8:9633. [Crossref] [PubMed]
- Uehara T, Hayden FG, Kawaguchi K, et al. Treatment-Emergent Influenza Variant Viruses With Reduced Baloxavir Susceptibility: Impact on Clinical and Virologic Outcomes in Uncomplicated Influenza. J Infect Dis 2020;221:346-55. [Crossref] [PubMed]
- Takashita E, Kawakami C, Ogawa R, et al. Influenza A(H3N2) virus exhibiting reduced susceptibility to baloxavir due to a polymerase acidic subunit I38T substitution detected from a hospitalised child without prior baloxavir treatment, Japan, January 2019. Euro Surveill 2019;24:1900170. [Crossref] [PubMed]
- Grohskopf LA, Sokolow LZ, Broder KR, et al. Prevention and Control of Seasonal Influenza with Vaccines. MMWR Recomm Rep 2016;65:1-54. [Crossref] [PubMed]
- Olsen SJ, Azziz-Baumgartner E, Budd AP, et al. Decreased influenza activity during the COVID-19 pandemic-United States, Australia, Chile, and South Africa, 2020. Am J Transplant 2020;20:3681-5. [Crossref] [PubMed]
- Lafond KE, Nair H, Rasooly MH, et al. Global Role and Burden of Influenza in Pediatric Respiratory Hospitalizations, 1982-2012: A Systematic Analysis. PLoS Med 2016;13:e1001977. [Crossref] [PubMed]
- Brewer NT, Chapman GB, Rothman AJ, et al. Increasing Vaccination: Putting Psychological Science Into Action. Psychol Sci Public Interest 2017;18:149-207. [Crossref] [PubMed]
- World Health Organization. Influenza pandemic preparedness plan: the role of WHO and guidelines for national and regional planning. Geneva: World Health Organization; 1999.
- Shrestha SS, Swerdlow DL, Borse RH, et al. Estimating the burden of 2009 pandemic influenza A (H1N1) in the United States (April 2009-April 2010). Clin Infect Dis 2011;52.
- Baden LR, Drazen JM, Kritek PA, et al. H1N1 influenza A disease--information for health professionals. N Engl J Med 2009;360:2666-7. [Crossref] [PubMed]
- Yu X, Xu C, Huang W, et al. The incidence of influenza in children was decreased in the first flu season after COVID-19 pandemic in Wuhan. J Infect Public Health 2021;14:1279-81. [Crossref] [PubMed]
- Cowling BJ, Ali ST, Ng TWY, et al. Impact assessment of non-pharmaceutical interventions against coronavirus disease 2019 and influenza in Hong Kong: an observational study. Lancet Public Health 2020;5:e279-88. [Crossref] [PubMed]
- Chen Z, Tsui JL, Gutierrez B, et al. COVID-19 pandemic interventions reshaped the global dispersal of seasonal influenza viruses. Science 2024;386:eadq3003. [Crossref] [PubMed]
- Melidou A, Ködmön C, Nahapetyan K, et al. Influenza returns with a season dominated by clade 3C.2a1b.2a.2 A(H3N2) viruses, WHO European Region, 2021/22. Euro Surveill 2022;27:2200255. [Crossref] [PubMed]
- Nuzhath T, Yang Y, Couture MC, et al. Modification and validation of the teen vaccine hesitancy scale toward vaccines for adolescents. Vaccine 2026;77:128385. [Crossref] [PubMed]
- Disease Control and Prevention. Flu Vaccination Coverage, United States, 2023–24 Influenza Season. 2024. Available online: https://www.cdc.gov/fluvaxview/coverage-by-season/2023-2024.html
- MacDonald NESAGE Working Group on Vaccine Hesitancy. Vaccine hesitancy: Definition, scope and determinants. Vaccine 2015;33:4161-4. [Crossref] [PubMed]
- Dubé È, Ward JK, Verger P, et al. Vaccine Hesitancy, Acceptance, and Anti-Vaccination: Trends and Future Prospects for Public Health. Annu Rev Public Health 2021;42:175-91. [Crossref] [PubMed]
- Gayle T, Webber K, Finstuen-Magro K, et al. Parental Attitudes Toward Inpatient Pediatric Vaccination. Hosp Pediatr 2026;16:e279-84. [Crossref] [PubMed]
- Kahn KE, Santibanez TA, Jain A, et al. Parental reasons for non-receipt of influenza vaccination among children 6 months-17 years and changes over time, 2015-2024. Vaccine 2025;61:127415. [Crossref] [PubMed]
- Salo-Tuominen K, Teros-Jaakkola T, Toivonen L, et al. Parental socioeconomic and psychological determinants of the 2009 pandemic influenza A(H1N1) vaccine uptake in children. Vaccine 2022;40:3684-9. [Crossref] [PubMed]
- Wei Z, Sun X, Yang Y, et al. Seasonal influenza vaccine hesitancy profiles and determinants among Chinese children's guardians and the elderly. Expert Rev Vaccines 2021;20:601-10. [Crossref] [PubMed]
- Stuetzle SCW, Willis M, Barnowska EJ, et al. Factors influencing vaccine hesitancy toward non-covid vaccines in South Asia: a systematic review. BMC Public Health 2025;25:1246. [Crossref] [PubMed]
- Gauthier NPG, Chorlton SD, Krajden M, et al. Agnostic Sequencing for Detection of Viral Pathogens. Clin Microbiol Rev 2023;36:e0011922. [Crossref] [PubMed]
- Zeng D, Jiao J, Mo T. Combination of nucleic acid amplification and CRISPR/Cas technology in pathogen detection. Front Microbiol 2024;15:1355234. [Crossref] [PubMed]
- Yan X, Li K, Lei Z, et al. Prevalence and associated outcomes of coinfection between SARS-CoV-2 and influenza: a systematic review and meta-analysis. Int J Infect Dis 2023;136:29-36. [Crossref] [PubMed]
- Mott JA, Fry AM, Kondor R, et al. Re-emergence of influenza virus circulation during 2020 in parts of tropical Asia: Implications for other countries. Influenza Other Respir Viruses 2021;15:415-8. [Crossref] [PubMed]
- Wang K, Lv Z, Li S, et al. Post-pandemic respiratory infection trends among Northern Chinese children: Challenges and responses. Pulmonology 2024;30:419-21. [Crossref] [PubMed]
- Hoy G, Maier HE, Kuan G, et al. Increased influenza severity in children in the wake of SARS-CoV-2. Influenza Other Respir Viruses 2023;17:e13178. [Crossref] [PubMed]
- Yang MC, Su YT, Chen PH, et al. Changing patterns of infectious diseases in children during the COVID-19 pandemic. Front Cell Infect Microbiol 2023;13:1200617. [Crossref] [PubMed]
- Neumann G, Kawaoka Y. Seasonality of influenza and other respiratory viruses. EMBO Mol Med 2022;14:e15352. [Crossref] [PubMed]
- Trepat K, Gibeaud A, Trouillet-Assant S, et al. Exploring viral respiratory coinfections: Shedding light on pathogen interactions. PLoS Pathog 2024;20:e1012556. [Crossref] [PubMed]
- Drori Y, Jacob-Hirsch J, Pando R, et al. Influenza A Virus Inhibits RSV Infection via a Two-Wave Expression of IFIT Proteins. Viruses 2020;12:1171. [Crossref] [PubMed]
- Gilbert-Girard S, Piret J, Rhéaume C, et al. Influenza A virus interferes with respiratory syncytial virus in mice and reconstituted human airway epithelium. Microbiol Spectr 2025;13:e0318724. [Crossref] [PubMed]
- Kramer SC, Pirikahu S, Casalegno JS, et al. Characterizing the interactions between influenza and respiratory syncytial viruses and their implications for epidemic control. Nat Commun 2024;15:10066. [Crossref] [PubMed]
- Haney J, Vijayakrishnan S, Streetley J, et al. Coinfection by influenza A virus and respiratory syncytial virus produces hybrid virus particles. Nat Microbiol 2022;7:1879-90. [Crossref] [PubMed]
- George JA, AlShamsi SH, Alhammadi MH, et al. Exacerbation of Influenza A Virus Disease Severity by Respiratory Syncytial Virus Co-Infection in a Mouse Model. Viruses 2021;13:1630. [Crossref] [PubMed]
- Hartwig SM, Miller AM, Varga SM. Respiratory Syncytial Virus Provides Protection against a Subsequent Influenza A Virus Infection. J Immunol 2022;208:720-31. [Crossref] [PubMed]
- Swets MC, Russell CD, Harrison EM, et al. SARS-CoV-2 co-infection with influenza viruses, respiratory syncytial virus, or adenoviruses. Lancet 2022;399:1463-4. [Crossref] [PubMed]
- Bai L, Zhao Y, Dong J, et al. Coinfection with influenza A virus enhances SARS-CoV-2 infectivity. Cell Res 2021;31:395-403. [Crossref] [PubMed]
- Ziegler CGK, Allon SJ, Nyquist SK, et al. SARS-CoV-2 Receptor ACE2 Is an Interferon-Stimulated Gene in Human Airway Epithelial Cells and Is Detected in Specific Cell Subsets across Tissues. Cell 2020;181:1016-1035.
- COVID is here to stay: countries must decide how to adapt. Nature. 2022;601:165.
- Kennedy RB, Ovsyannikova IG, Poland GA. Update on Influenza Vaccines: Needs and Progress. J Allergy Clin Immunol Pract 2021;9:3599-603. [Crossref] [PubMed]
- Belshe RB, Edwards KM, Vesikari T, et al. Live attenuated versus inactivated influenza vaccine in infants and young children. N Engl J Med 2007;356:685-96. [Crossref] [PubMed]
- Thwaites RS, Uruchurtu ASS, Negri VA, et al. Early mucosal events promote distinct mucosal and systemic antibody responses to live attenuated influenza vaccine. Nat Commun 2023;14:8053. [Crossref] [PubMed]
- De Gioia ER, Porqueddu A, Nebiaj O, et al. The Role of Needle Fear in Pediatric Flu Vaccine Hesitancy: A Cross-Sectional Study in Bologna Metropolitan Area. Vaccines (Basel) 2022;10:1388. [Crossref] [PubMed]
- Wei CJ, Crank MC, Shiver J, et al. Next-generation influenza vaccines: opportunities and challenges. Nat Rev Drug Discov 2020;19:239-52. [Crossref] [PubMed]
- Chang LJ, Anderson EJ, Jeanfreau R, et al. Safety and immunogenicity of high doses of quadrivalent influenza vaccine in children 6 months through <18 years of age: A randomized controlled phase II dose-finding trial. Vaccine 2021;39:1572-82. [Crossref] [PubMed]
- Zhang W, Cui H, Xu J, et al. Biodistribution and mechanisms of action of MF59 and MF59-like adjuvants. J Control Release 2025;378:573-87. [Crossref] [PubMed]
- Lin YJ, Wen CN, Lin YY, et al. Oil-in-water emulsion adjuvants for pediatric influenza vaccines: a systematic review and meta-analysis. Nat Commun 2020;11:315. [Crossref] [PubMed]
- Erbelding EJ, Post DJ, Stemmy EJ, et al. A Universal Influenza Vaccine: The Strategic Plan for the National Institute of Allergy and Infectious Diseases. J Infect Dis 2018;218:347-54. [Crossref] [PubMed]
- Cowling BJ, Okoli GN. Influenza Vaccine Effectiveness and Progress Towards a Universal Influenza Vaccine. Drugs 2024;84:1013-23. [Crossref] [PubMed]
- Taaffe J, Ostrowsky JT, Mott J, et al. Advancing influenza vaccines: A review of next-generation candidates and their potential for global health impact. Vaccine 2024;42:126408. [Crossref] [PubMed]
- Xu J, Luo Q, Huang Y, et al. Influenza neuraminidase mutations and resistance to neuraminidase inhibitors. Emerg Microbes Infect 2024;13:2429627. [Crossref] [PubMed]
- Behillil S, May F, Fourati S, et al. Oseltamivir Resistance in Severe Influenza A(H1N1)pdm09 Pneumonia and Acute Respiratory Distress Syndrome: A French Multicenter Observational Cohort Study. Clin Infect Dis 2020;71:1089-91. [Crossref] [PubMed]
- Todd B, Tchesnokov EP, Götte M. The active form of the influenza cap-snatching endonuclease inhibitor baloxavir marboxil is a tight binding inhibitor. J Biol Chem 2021;296:100486. [Crossref] [PubMed]
- Kohlbrand AJ, Stokes RW, Sankaran B, et al. Structural Studies of Inhibitors with Clinically Relevant Influenza Endonuclease Variants. Biochemistry 2024;63:264-72. [Crossref] [PubMed]
- Bonomini A, Mercorelli B, Loregian A. Antiviral strategies against influenza virus: an update on approved and innovative therapeutic approaches. Cell Mol Life Sci 2025;82:75. [Crossref] [PubMed]
- Tao S, Zhou Z, Li X, et al. Recent Advances of Non-Nucleoside Polymerase Inhibitors With Broad-Spectrum Antiviral Activities. Med Res Rev 2026; Epub ahead of print. [Crossref]
- Chen Z, Yu H. Importance of integrating epidemiological and genomic surveillance of seasonal influenza viruses to monitor global circulation. Clin Transl Med 2024;14:e70126. [Crossref] [PubMed]
- Yang J, Gong Y, Zhang C, et al. Co-existence and co-infection of influenza A viruses and coronaviruses: Public health challenges. Innovation (Camb) 2022;3:100306. [Crossref] [PubMed]